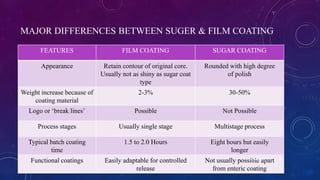
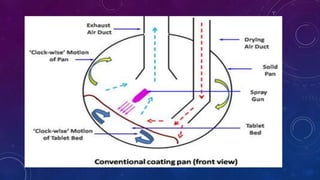
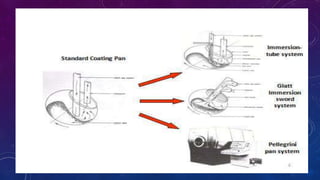
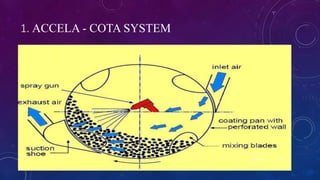
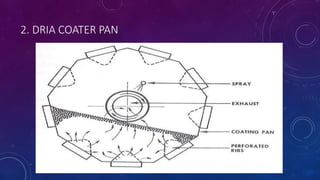
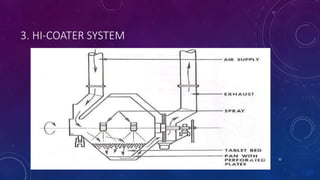
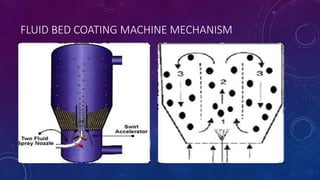
Tablet coating is the application of a coating material to the exterior of a tablet to confer benefits over uncoated tablets. Common purposes are to mask taste/odor, protect drugs from environmental factors or gastric acid, and control drug release. Major types are sugar coating, film coating, enteric coating, and press coating. Film coating involves spraying a polymer solution onto tablets while sugar coating is a multistage process including sealing, subcoating, smoothing, coloring and polishing. Standard pans and perforated pans are commonly used coating equipment.