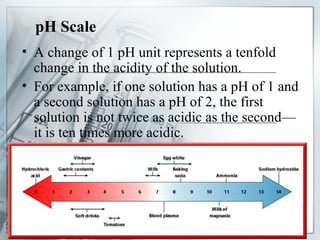
This document discusses acids and bases. It defines acids as solutions with an excess of H+ ions, noting that they have sour tastes, can corrode substances, and turn litmus paper red. Common acids include acetic acid in vinegar and citric acid in fruits. Bases are defined as solutions with an excess of OH- ions, noting they have bitter tastes and turn litmus paper blue. Common bases are found in soaps, ammonia, and cleaning products. The document also explains the pH scale for measuring acidity and alkalinity and how acid-base reactions result in neutralization and salt formation.