The document discusses a case of myelodysplastic syndrome (MDS) presenting as anemia, detailing its clinical presentation, diagnostic workup, and management approaches, including treatment options like azacitidine and decitabine. It highlights the importance of supportive care and the use of immunosuppressive therapies, along with the challenges faced in the Indian context, such as limited access to allogeneic stem cell transplantation. The document concludes with recommendations from various guidelines emphasizing azacitidine as the preferred treatment for high-risk MDS due to its survival benefits and overall improved patient outcomes.







![Evaluation
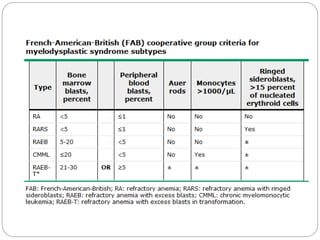
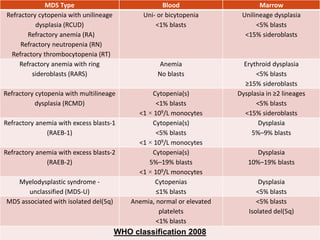
Myelodysplastic syndrome (MDS) is characterized by abnormal
cell morphology (dysplasia) and quantitative changes in one or
more of the blood and bone marrow elements (ie, red cells,
granulocytes, platelets)
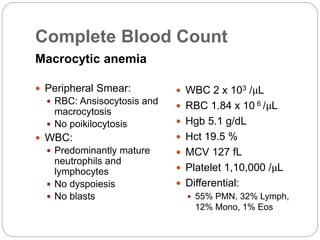
Complete blood count
Anemia
Leukopenia
Thrombocytopenia
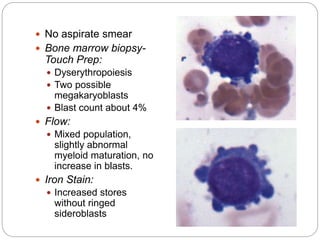
Periphreral blood smear
Dysplastic blood cells
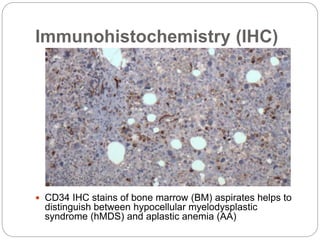
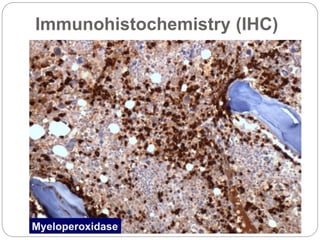
Bone marrow aspirate/ biopsy
Evaluation of the blasts and other cells
Fibrosis of marrow
Genetic features [del(7q), del(5q), del(13q),del(11q),
del(12p),del(9q)]
Distinguishes between MDS and acute myeloid leukemia (AML)
Prognostic](https://image.slidesharecdn.com/refractoryanemiacasereportmds-150925050126-lva1-app6892/85/Refractory-anemia-case-report-mds-14-320.jpg)
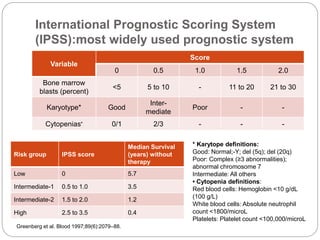
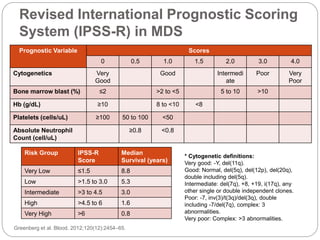



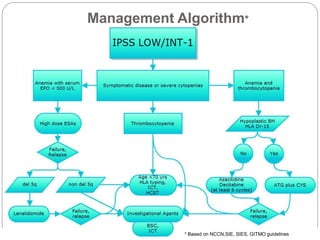
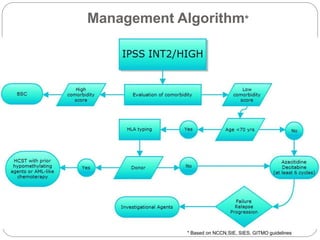





![Comparison between Decitabine and
Azacitidine for MDS: A Meta-analysis with
1,392 Subjects
Azacitidine vs BSC
Significantly improved overall survival (hazard ratio [HR],
0.69; 95% CI, 0.54-0.87)
Significantly delayed and time to acute myeloid leukemia
transformation (HR, 0.51; 95% CI, 0.35-0.74).
But these benefits were not found with decitabine.
Among patients with higher risk (IPSS >=3) or older >75
years, treatment with azacitidine was a favorable factor,
whereas decitabine showed no advantage.
Therefore, with higher overall response rates and better
survival benefits, azacitidine is recommended as the
first-line hypomethylating agent for MDS, especially in
elderly patients or those with high risk.
Mixue et al Clinical Lymphoma,Myeloma and Leukemia (2014).](https://image.slidesharecdn.com/refractoryanemiacasereportmds-150925050126-lva1-app6892/85/Refractory-anemia-case-report-mds-27-320.jpg)





