Embed presentation
Download to read offline


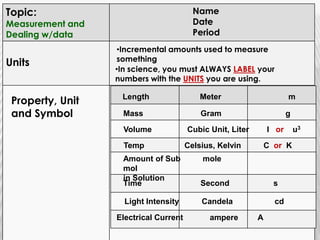
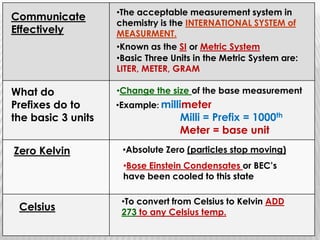
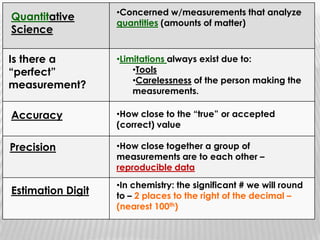
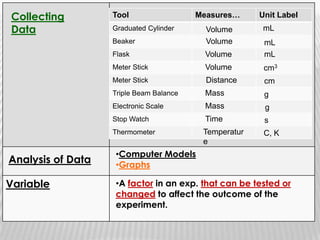
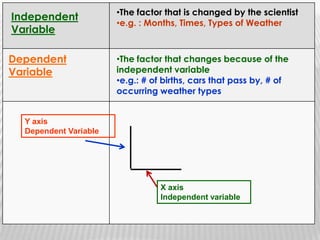

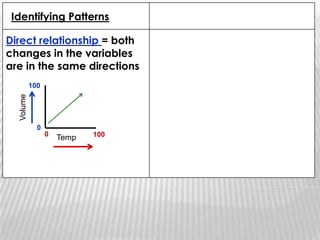
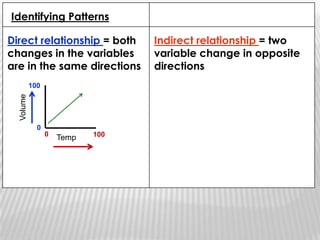
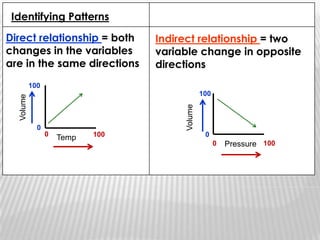

You will need a pencil and colored pencils or highlighters for today's chemistry lesson. The lesson will cover measurements and dealing with data, including the metric system units of liters, meters, and grams. Key concepts are that measurements must include units, and the acceptable measurement system is the International System of Units (SI) or metric system. Measurements have limitations related to tools, carelessness, and how close they are to the true or accepted value in terms of accuracy and precision.











