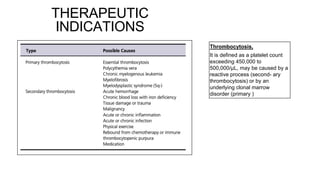
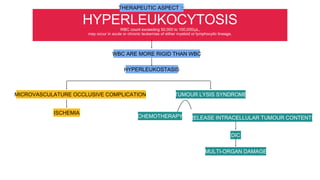
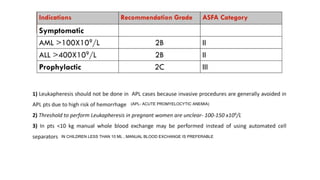
This document discusses various types of apheresis procedures including plateletpheresis, erythrocytopheresis, and leukapheresis. It provides definitions and principles of plateletpheresis including donor selection criteria, product requirements, advantages of single donor platelets, and complications. Therapeutic indications for plateletpheresis and leukapheresis are mentioned. Guidelines for donor selection, product requirements, and procedures for erythrocytopheresis are summarized as well.






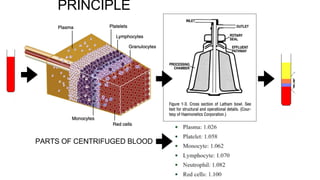





![PLATELET PRODUCT REQUIREMENT
•Standards for Blood Banks and Transfusion Services require that the mini- mum
number of platelets in an apheresis product should be 3.0 × 1011 in 90% of units tested
(Standards Committee of the AABB, 2018).
•The U.S. Food and Drug Administration (FDA) requires a minimum of 3.0 × 1011 in 75%
of units tested (Code of Federal Regulations [CFR], 2014).
•The result of these requirements, however, is that an apheresis platelet product
contains at least as many platelets as a pool of platelets from six whole- blood
donations.](https://image.slidesharecdn.com/plateletphresisppt-230211064001-71fc9baf/85/PLATELETPHRESIS-pptx-17-320.jpg)