This document defines and discusses several key concepts in food science and technology:
1. It defines density and different types of density measurements including true, material, particle, apparent, and bulk densities.
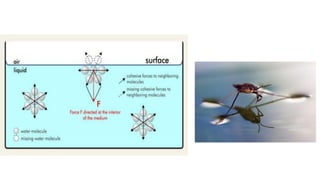
2. It explains phase changes, pH, osmosis, reverse osmosis, colloidal systems, surface tension, and specific gravity.
3. It discusses physical attributes of foods such as size, shape, volume, density, and porosity which are important for processing, screening, grading, and evaluating foods.