Report
Share
Download to read offline
Recommended
Recommended
More Related Content
Similar to phases.pdf
Similar to phases.pdf (20)
1670327194_887_Research_with_human_participation.pptx

1670327194_887_Research_with_human_participation.pptx
Stages of drug development by Dr Joseph Oyepata Simeon (Ph.D)

Stages of drug development by Dr Joseph Oyepata Simeon (Ph.D)
The Four Phases of Clinical Trials Before Market Approval

The Four Phases of Clinical Trials Before Market Approval
Clinical trial phases, requirements and regulations

Clinical trial phases, requirements and regulations
process for discovery and development of new drug and issues related to testi...

process for discovery and development of new drug and issues related to testi...
Lecture # 03 New Drug Approval and Development Process,

Lecture # 03 New Drug Approval and Development Process,
Clinical study emphasizing on phases of clinical trials.

Clinical study emphasizing on phases of clinical trials.
More from Bindu238662
More from Bindu238662 (20)
polarimetry-110527042535-phpap hghygytrfgfhghghghghghghghp01.pptx

polarimetry-110527042535-phpap hghygytrfgfhghghghghghghghp01.pptx
EXACT PROJECT PRESENTATION DM.pgefuggsggegeggehgegptx

EXACT PROJECT PRESENTATION DM.pgefuggsggegeggehgegptx
MI (4).ppt myocardial infarction is a type of heart disease

MI (4).ppt myocardial infarction is a type of heart disease
CASE PRESENTATION ON EPILEPSY.pptx epilepsy is a disorder

CASE PRESENTATION ON EPILEPSY.pptx epilepsy is a disorder
alcohol depenDENCE IS A DISORDER WHERE A ERSON IS ADDICTED TO ALCOHOL

alcohol depenDENCE IS A DISORDER WHERE A ERSON IS ADDICTED TO ALCOHOL
MI is cardiovascular disorder with infarction in cardiac muscles which leads ...

MI is cardiovascular disorder with infarction in cardiac muscles which leads ...
COPD is a lung disorder which involves the lung obstruction for airway and br...

COPD is a lung disorder which involves the lung obstruction for airway and br...
minimized asthma project is a project related to asthma treatment through GIN...

minimized asthma project is a project related to asthma treatment through GIN...
Recently uploaded
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Recently uploaded (20)
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Seal of Good Local Governance (SGLG) 2024Final.pptx

Seal of Good Local Governance (SGLG) 2024Final.pptx
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II

Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
phases.pdf
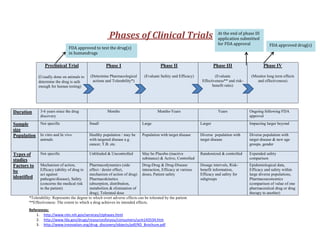
- 1. References: 1. http://www.nlm.nih.gov/services/ctphases.html 2. http://www.fda.gov/drugs/resourcesforyou/consumers/ucm143534.htm 3. http://www.innovation.org/drug_discovery/objects/pdf/RD_Brochure.pdf Phases of Clinical Trials Preclinical Trial (Usually done on animals to determine the drug is safe enough for human testing) Phase I (Determine Pharmacological actions and Tolerability*) Phase II (Evaluate Safety and Efficacy) Phase III (Evaluate Effectiveness** and risk- benefit ratio) Phase IV (Monitor long term effects and effectiveness) Duration 3-6 years since the drug discovery Months Months-Years Years Ongoing following FDA approval Sample size Not specific Small Large Larger Impacting larger beyond Population In vitro and In vivo animals Healthy population / may be with targeted disease e.g. cancer, T.B. etc. Population with target disease Diverse population with target disease Diverse population with target disease & new age groups, gender Types of studies Not specific Unblinded & Uncontrolled May be Placebo (inactive substance) & Active, Controlled Randomized & controlled Expended safety comparison Factors to be identified Mechanism of action, Efficacy (ability of drug to act against pathogen/disease), Safety (concerns the medical risk to the patient) Pharmacodynamics (side effect / desire effect, mechanism of action of drug) Pharmacokinetics (absorption, distribution, metabolism & elimination of drug), Tolerated dose Drug-Drug & Drug-Disease interaction, Efficacy at various doses, Patient safety Dosage intervals, Risk- benefit information, Efficacy and safety for subgroups Epidemiological data, Efficacy and safety within large diverse populations, Pharmacoeconomics (comparison of value of one pharmaceutical drug or drug therapy to another) *Tolerability: Represents the degree to which overt adverse effects can be tolerated by the patient. **Effectiveness: The extent to which a drug achieves its intended effects. FDA approved drug(s) FDA approved to test the drug(s) in humandrugs At the end of phase III application submitted for FDA approval