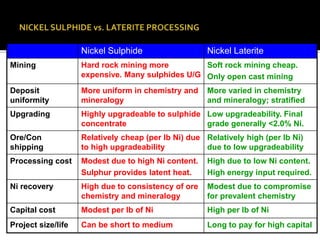
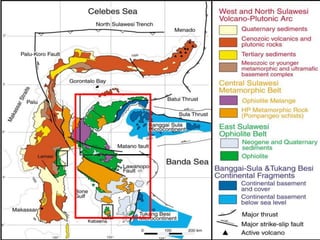
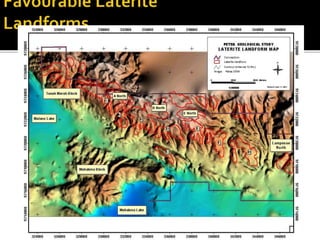
This document provides an overview of nickel laterite deposits, including:
1. Laterites form through chemical weathering of ultramafic rocks under tropical conditions, which leaches silica and magnesium and concentrates iron and other elements like nickel.
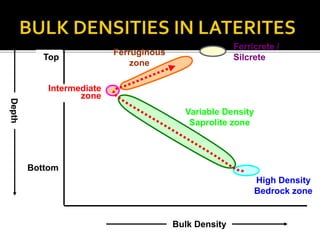
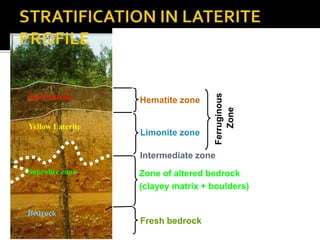
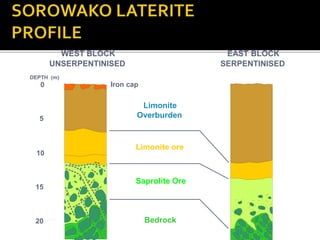
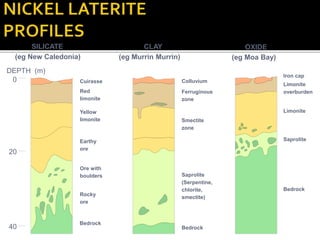
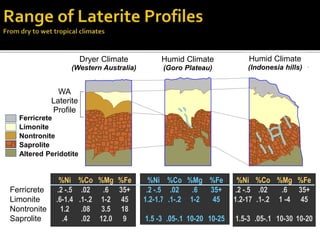
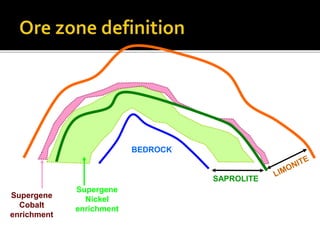
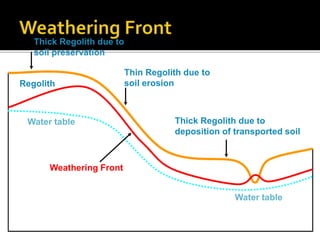
2. A laterite profile develops distinct zones from top to bottom with varying iron, nickel, magnesium, and other element concentrations based on the degree of weathering.
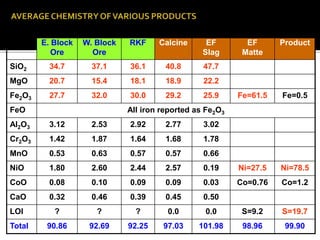
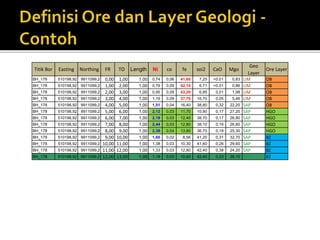
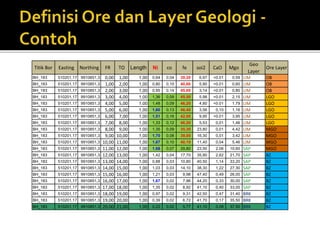
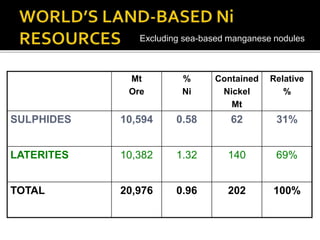
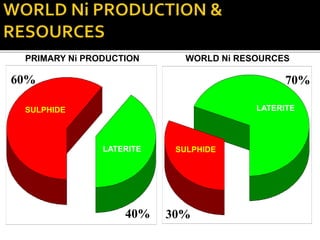
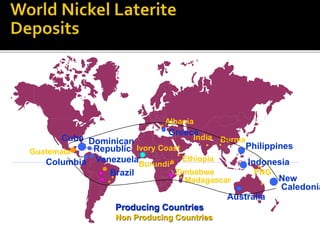
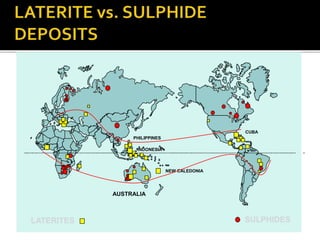
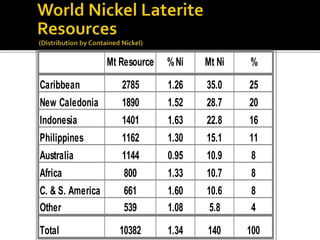
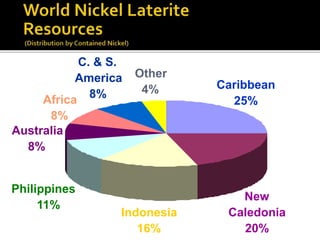
3. Major nickel resources are found in laterites, which make up 69% of the world's nickel resources compared to 31% in sulphide deposits. The top five countries by contained nickel in laterites are New Caledonia, Indonesia, Australia, Philippines, and Cuba.




![ Major Nickel producing companies:
1. Norilsk 243,000 t (19%)
2. CVRD Inco 221,000 t (17%)
3. BHP-Billiton 146,000 t (11%)
4. Falconbridge 114,000 t (9%) [Now Exstrata]
5. Jinchuan 93,000 t (7%)
6. Eramet 59,000 t (5%)
7. Sumitomo 51,000 t (4%)
Major Nickel producing countries:
1. Russia (Norilsk Nickel group)
2. Canada (CVRD Inco, Falconbridge, Sheritt)
3. Australia (BHP-B, Minara, Cawse, LionOre)](https://image.slidesharecdn.com/nickellateritedeposit-230216025703-5351cd97/85/Nickel-Laterite-Deposit-pptx-5-320.jpg)



















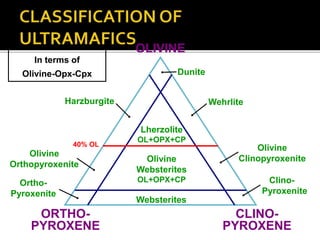


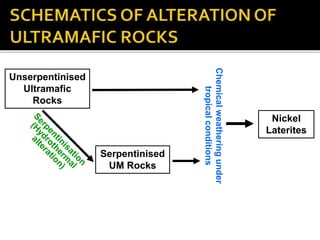








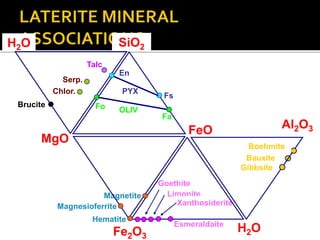
![ Serpentine: 3MgO.2SiO2.2H2O [H4Mg3Si2O9]
Magnesia is leached out first, leaving behind a silica
enriched phase or montmorillonite and chlorite
Ni can replace the magnesium being leached.This results
in the formation of:
Nickeliferous serpentine
Through a similar process, nickel is also fixed inTalc,
Chlorite, and Smectite
Eventually, montmorillonite and chlorite also break down,
releasing remaining magnesia and silica and forming iron
sesquioxides](https://image.slidesharecdn.com/nickellateritedeposit-230216025703-5351cd97/85/Nickel-Laterite-Deposit-pptx-54-320.jpg)
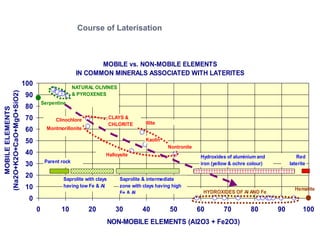


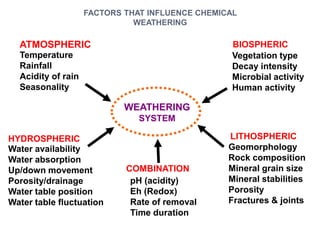
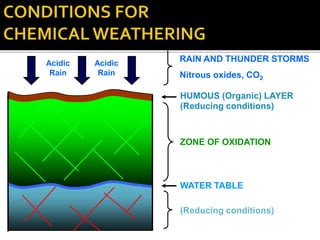
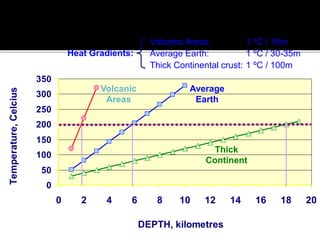
![ TEMPERATURE
Each 10 C change increases weathering speed by 2-3 times
Chemical weathering in tropics is 20-40 times higher than in
temperate regions
RAINFALL
Acidity of rain (dissolved CO2, Nitrous oxides)
Amount of precipitation (higher rainfall = higher leaching)
Seasonality:Constant humid vs.Wet/dry seasonal
EFFECT OF CLIMATE
Hematitic soils develop in hot and dryer climate
Goethitic/limonitic soils develop in hot and wet climate
Hot and humid climate leads to complete leaching of SiO2 & MgO
Seasonal wet/dry climate leads to formation of smectites
Climate can vary considerably over time [fossil laterites]](https://image.slidesharecdn.com/nickellateritedeposit-230216025703-5351cd97/85/Nickel-Laterite-Deposit-pptx-62-320.jpg)
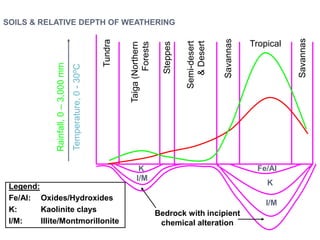




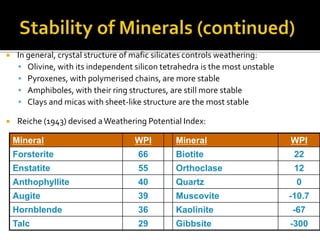
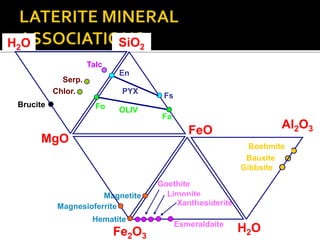
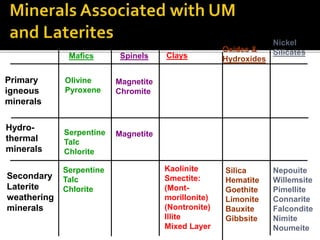
![Ultimate
stable
residuum
Hematite [Fe2O3]
Goethite [Fe2O3.H2O]
Limonite [Fe2O3.3H2O]
Boehmite [Al2O3.H2O]
Bauxite [Al2O3.2H2O]
Gibbsite [Al2O3.3H2O]
Secondary
minerals
Kaolinite
Smectites
Kaolinite
Primary
minerals
Olivine
Pyroxene
Plagioclase
Alkali Feldspars
Increased
Leaching](https://image.slidesharecdn.com/nickellateritedeposit-230216025703-5351cd97/85/Nickel-Laterite-Deposit-pptx-71-320.jpg)
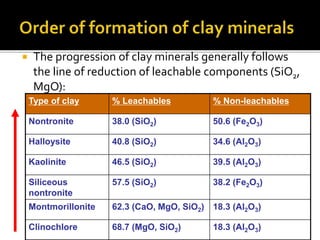
















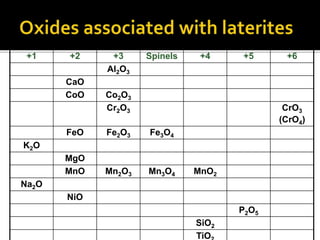





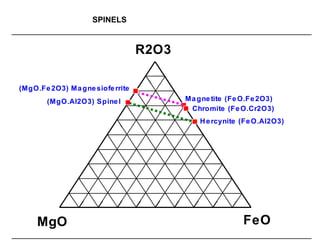
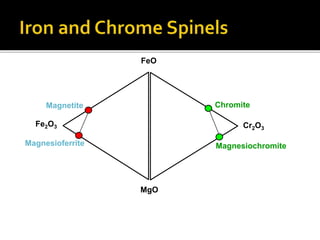
![SPINELS: RO.R2O3 [R=Fe, Mg, Mn, Ni, Zn]
[R2=Al, Fe, Cr, Mn]
Magnetite: Fe3O4 (FeO.Fe2O3) [Fe=72.3%]
Chromite: FeCr2O4 (FeO.Cr2O3)
Common spinel: MgAl2O4 (MgO.Al2O3)
OXIDES:
Hematite: Fe2O3
[Fe=69.9%]
Maghemite: Fe2.66 O4 [Fe=69.9%]
Silica: SiO2](https://image.slidesharecdn.com/nickellateritedeposit-230216025703-5351cd97/85/Nickel-Laterite-Deposit-pptx-112-320.jpg)