This newsletter summarizes guidelines for diagnosing and treating chronic kidney disease (CKD) and lupus nephritis (LN). CKD is classified based on cause, glomerular filtration rate (GFR) category, and albuminuria category. For LN, a kidney biopsy can confirm diagnosis and assess activity/chronicity to inform treatment and prognosis. Treatment recommendations are provided for different classes of LN, including immunosuppressive regimens for initial therapy and maintenance. Factors like infection risk, cardiovascular complications, bone health, pregnancy, and special populations are also addressed.

![N e p h r o l o g y
n e w s l e t t e r
Class I or Class
II lupus
nephritis
Approach to immunosuppressive
treatment for patients with Class I or
Class II LN
Immunosuppressive
treatment for patients with
Class I or Class II lupus
nephritis
these patients are similar to
those with minimal change
disease (MCD) or focal
segmental glomerulosclerosis
(FSGS), often showing a good
response to glucocorticoid
treatment.81-83
Although there
have been no RCTs,
observational data showed that
over 90% of patients given
glucocorticoid monotherapy
achieved remission within a
median time of 4 weeks.81, 84-88
Data on relapse are even more
limited, but there appears to be
a significant risk of relapse
after glucocorticoids are
tapered.89
Although optimal
duration is not known,
maintenance with low-dose
glucocorticoid plus an
additional agent such as
mycophenolic acid analogues
(MPAA), azathioprine, or a
CNI is suggested, especially in
patients with a history of
relapse.
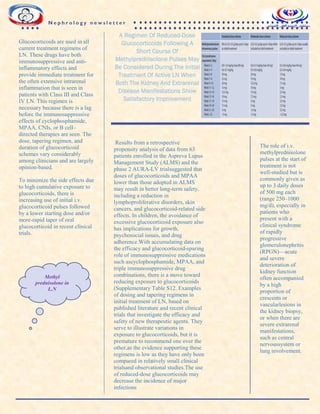
Class III or Class IV lupus
nephritis ,Initial therapy of active
Class III/IV lupus nephritis
recommend that patients with
active Class III or IV LN, with
or without a membranous
component, be treated initially
with glucocorticoids plus
either one of the following:
i. mycophenolic acid analogues
(MPAA) (1X); or
ii. low-dose intravenous
cyclophosphamide (1X)
iii. belimumab and either MPAA or low-
dose intravenous cyclophosphamide (X);
or
iv. MPAA and a calcineurin inhibitor
(CNI) when kidney function is not
severely impaired (for example
estimated glomerular filtration rate
[eGFR] ≤45 ml/min per 1.73 m2) (1X).
A regimen of reduced-dose
glucocorticoids following a short
course of methylprednisolone pulses
may be considered during the initial
treatment of active LN when both the
kidney and extrarenal disease
manifestations show satisfactory
improvement.
Example of glucocorticoid
regimens for lupus
nephritis.](https://image.slidesharecdn.com/nephronewsletterfinal-230714135018-e4abe770/85/nephro-newsletter-final-pdf-2-320.jpg)




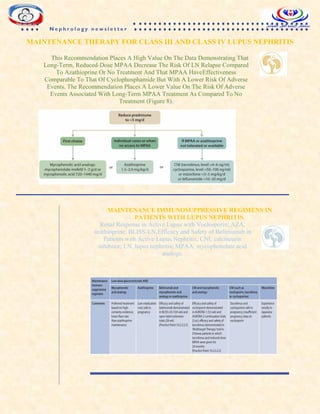
![INFECTIONS IN PATIENTS
WITH LN
Infection is a leading cause of death in
patients with LN, and infection-related deaths are
more common during the initial phase of
management following exposure to intensive
immunosuppressive therapy.
trimethoprim/sulfamethoxazole (TMP-SMX) was
effective in preventing pneumocystis pneumonia.
Herpes zoster is 2–10 times higher in patients with
SLE than in healthy controls, but the role of
antiviral prophylaxis is uncertain. Available zoster
vaccine preparations include the live-attenuated
vaccine Zostavax® and the adjuvanted
recombinant vaccine Shingrix. In general, live
vaccines should be avoided in immunosuppressed
subjects
. There is also concern that the polio vaccination
has been associated with lupus flares, whereas the
data on influenza vaccination are conflicting.
Response to vaccination is reduced following
exposure to high-dose immunosuppression
CARDIOVASCULAR COMPLICATIONS
IN PATIENTS WITH LN
Patients with SLE have both traditional
(dyslipidemia, smoking, obesity,
etc.) and non-traditional
(proteinuria, inflammation, etc.) CV
risk factors. A patient often has
multiple risk factors, which can be
secondary to disease-related organ damage
(especially CKD, hypertension, proteinuria) or
treatment (such as glucocorticoids and calcineurin
inhibitors [CNIs]). Regular evaluation of various
risk factors and timely treatment are essential to
prevent premature CV complications.
TREATMENT OF LUPUS NEPHRITIS IN
CHILDREN
Treat pediatric patients with LN using
immunosuppression regimens similar to those used
in adults, but consider issues relevant to
this population, such as dose
adjustment, growth, fertility, and
psychosocial factors, when devising
the therapy plan.
BONE HEALTH
Glucocorticoid therapy,
especially when high
doses are used
for long
durations,
increases bone
loss
glucocorticoid dose, and
the Fracture Risk
Assessment Tool (FRAX)
score.Calcium (optimal
intake 1000–1200 mg/d)
and vitamin D
supplementation are
recommended for patients
with LN, as well as
consideration for oral
bisphosphonates according
to individual risk
assessment
Malignancies in patients
with LN
Patients with SLE have
increased risk of malignant
tumors, including non-
Hodgkin’s lymphoma,
lung, liver, vulvar/vaginal,
thyroid, nonmelanoma
skin cancer, and the risk
(especially with bladder
cancer) is increased in
patients with a history of
exposure to
cyclophosphamide.
LUPUS PATIENTS
WITH KIDNEY
FAILURE
Patients with LN who
develop kidney failure
may be treated with
hemodialysis, peritoneal
dialysis, or kidney
transplantation; and
kidney transplantation is
preferred to
long-term
dialysis.
SPECIAL
POPULATIONS
N e p h r o l o g y n e w s l e t t e r](https://image.slidesharecdn.com/nephronewsletterfinal-230714135018-e4abe770/85/nephro-newsletter-final-pdf-7-320.jpg)