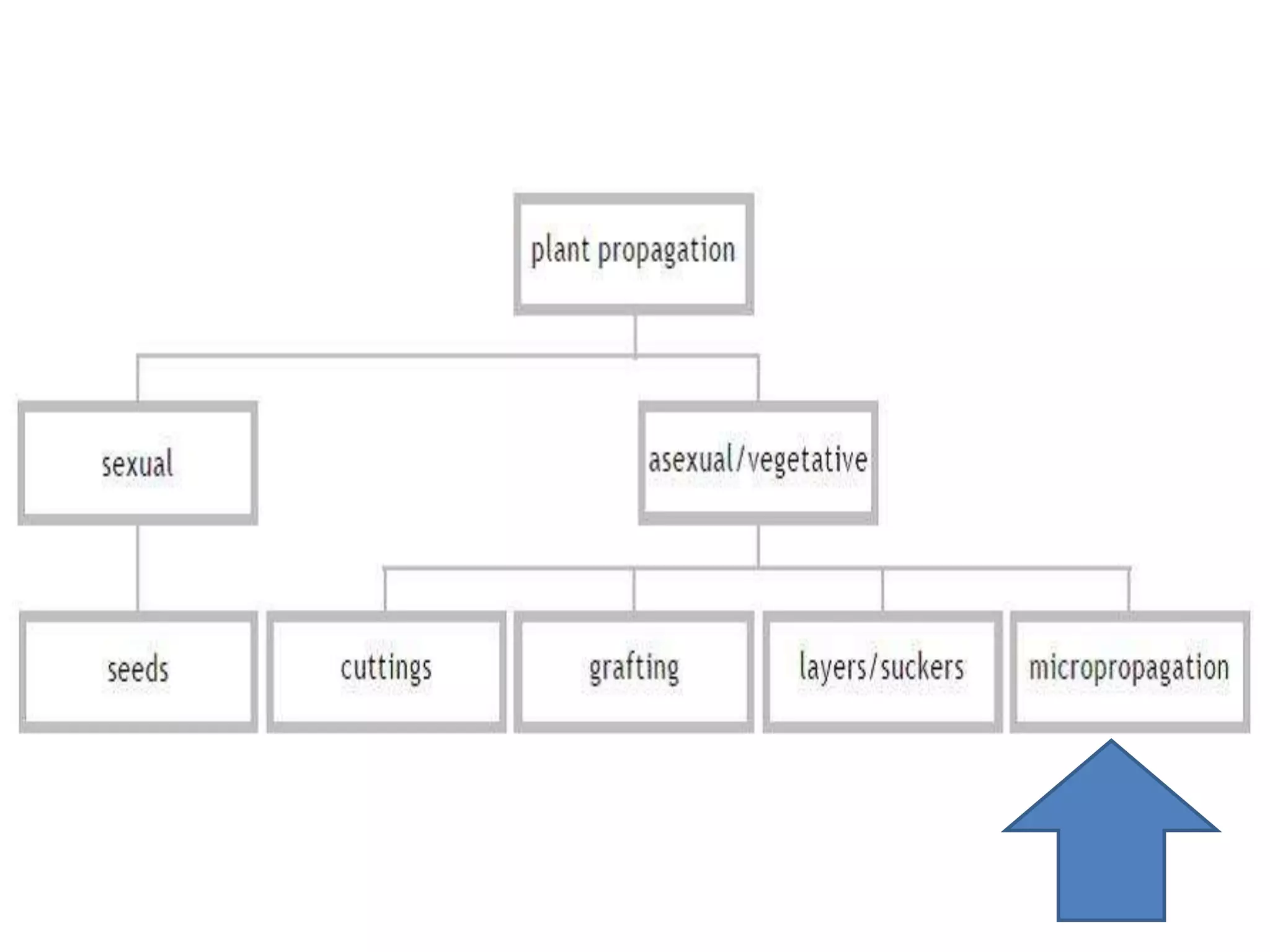
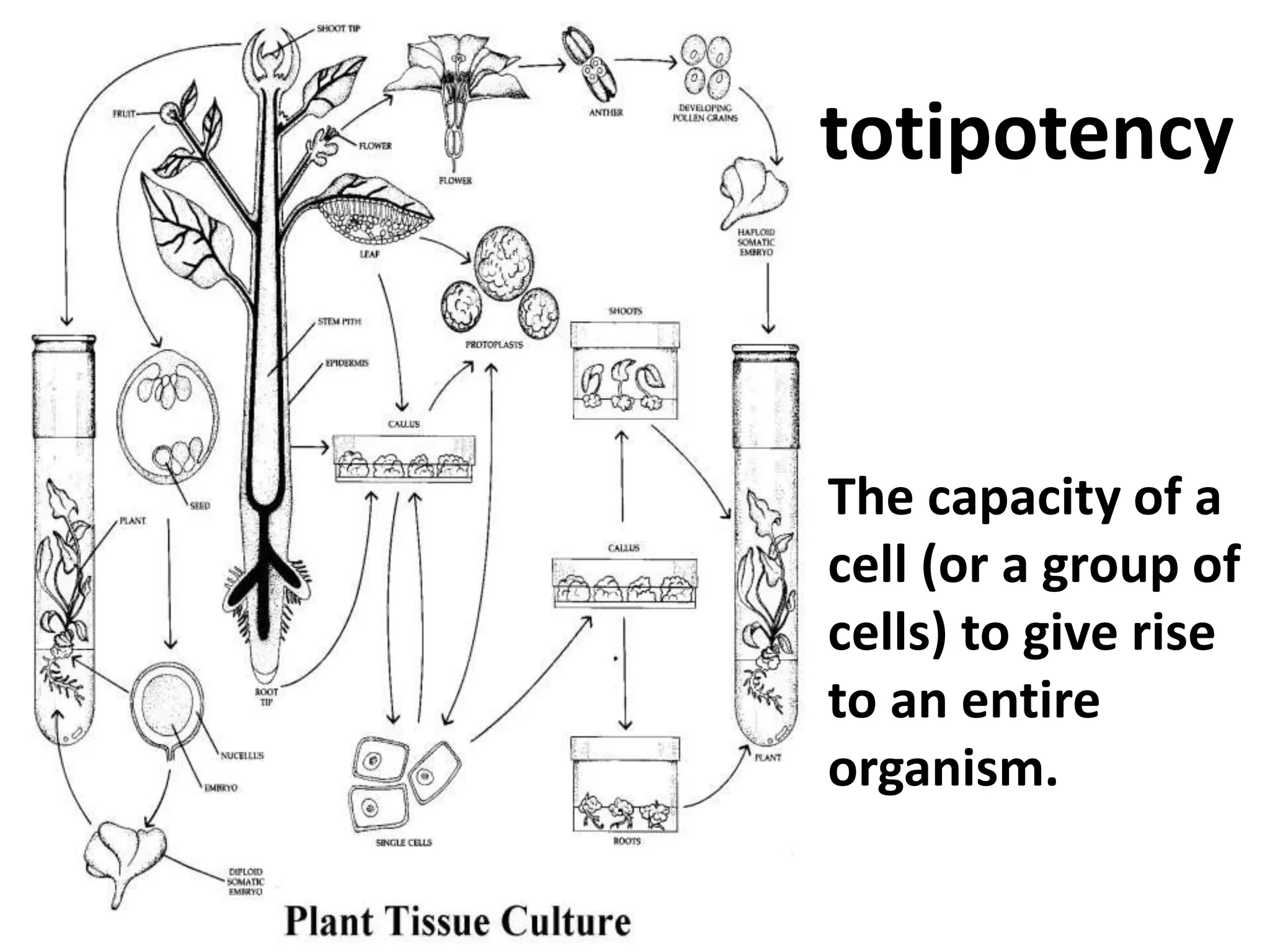
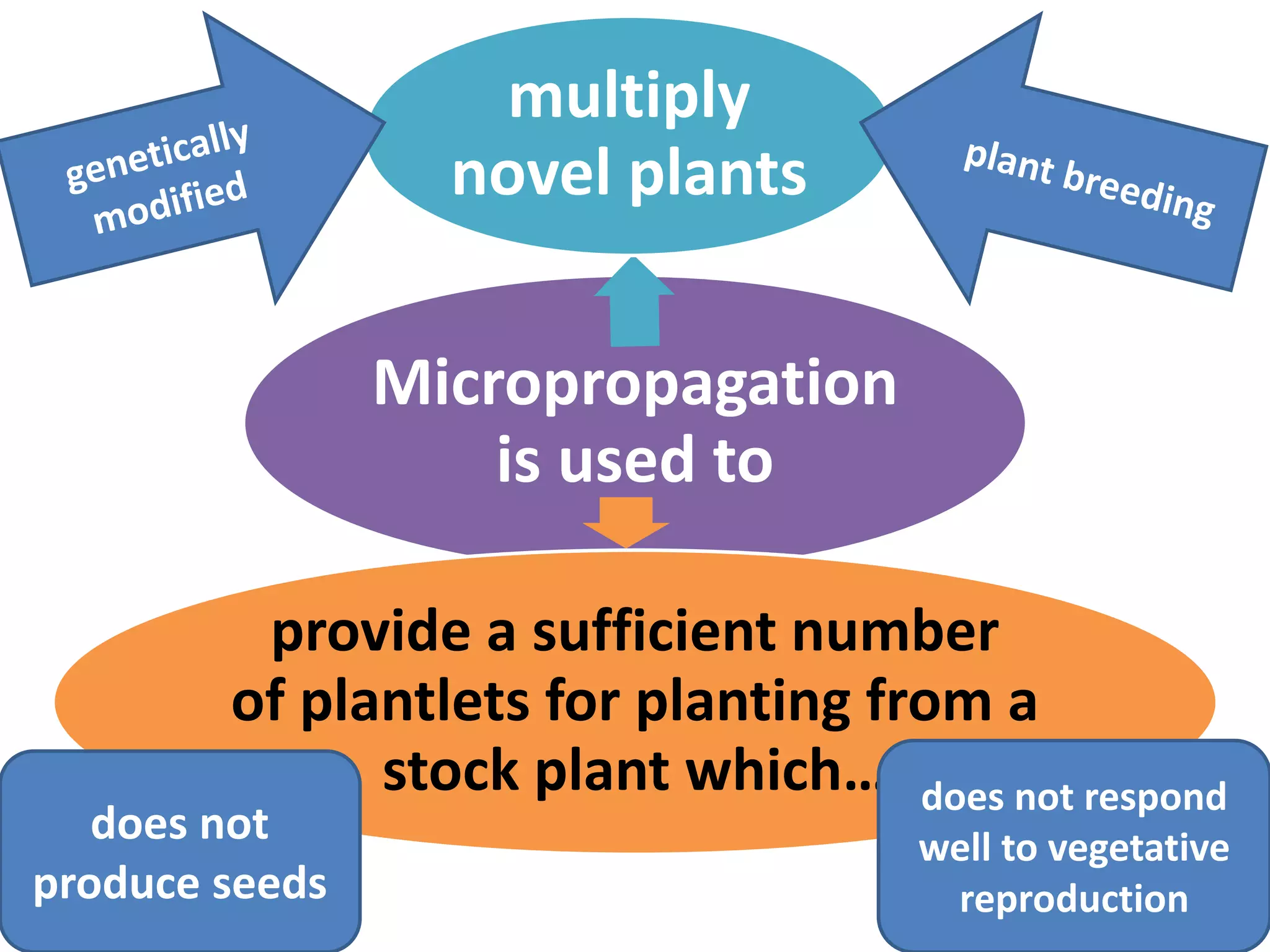
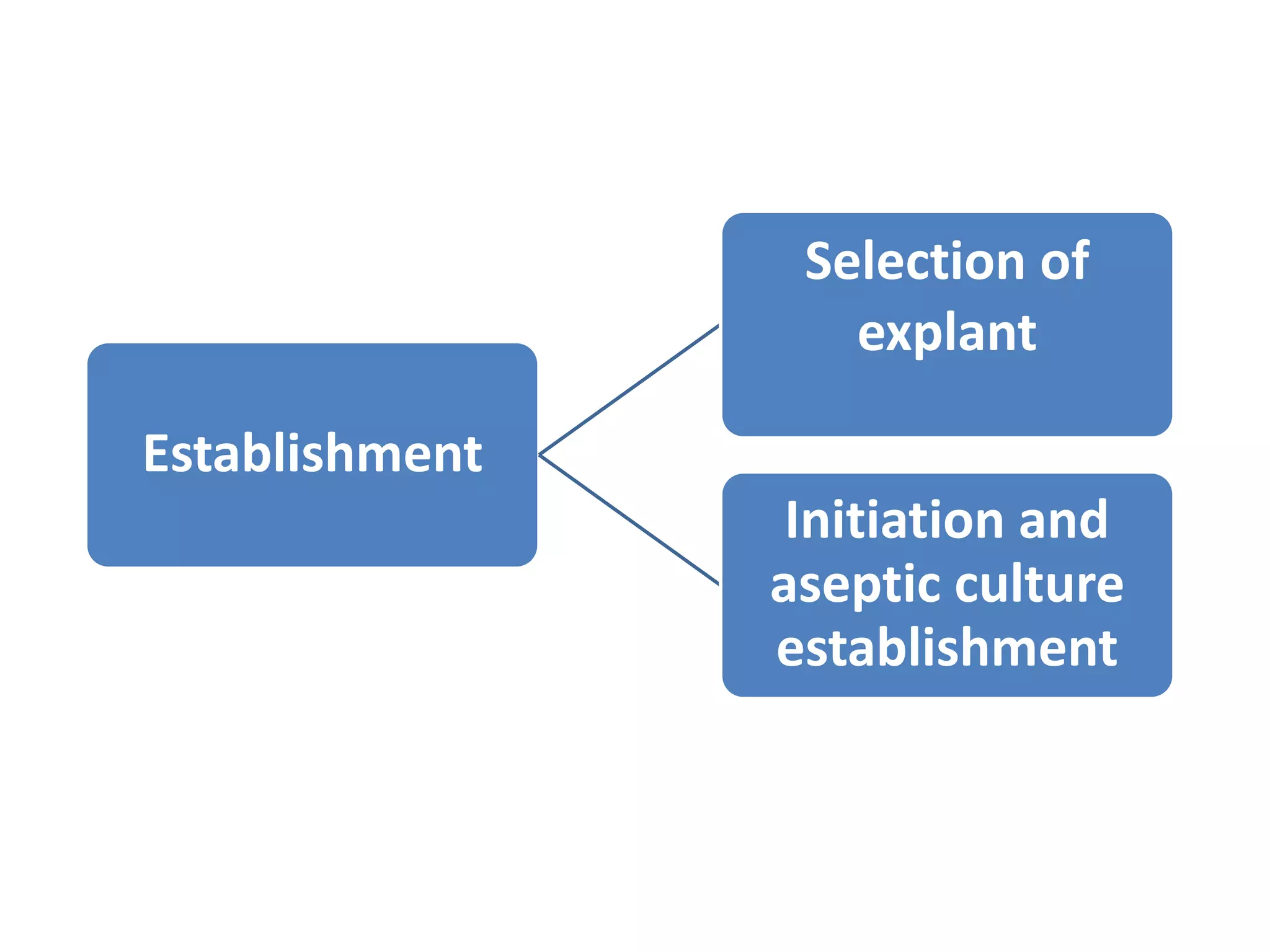
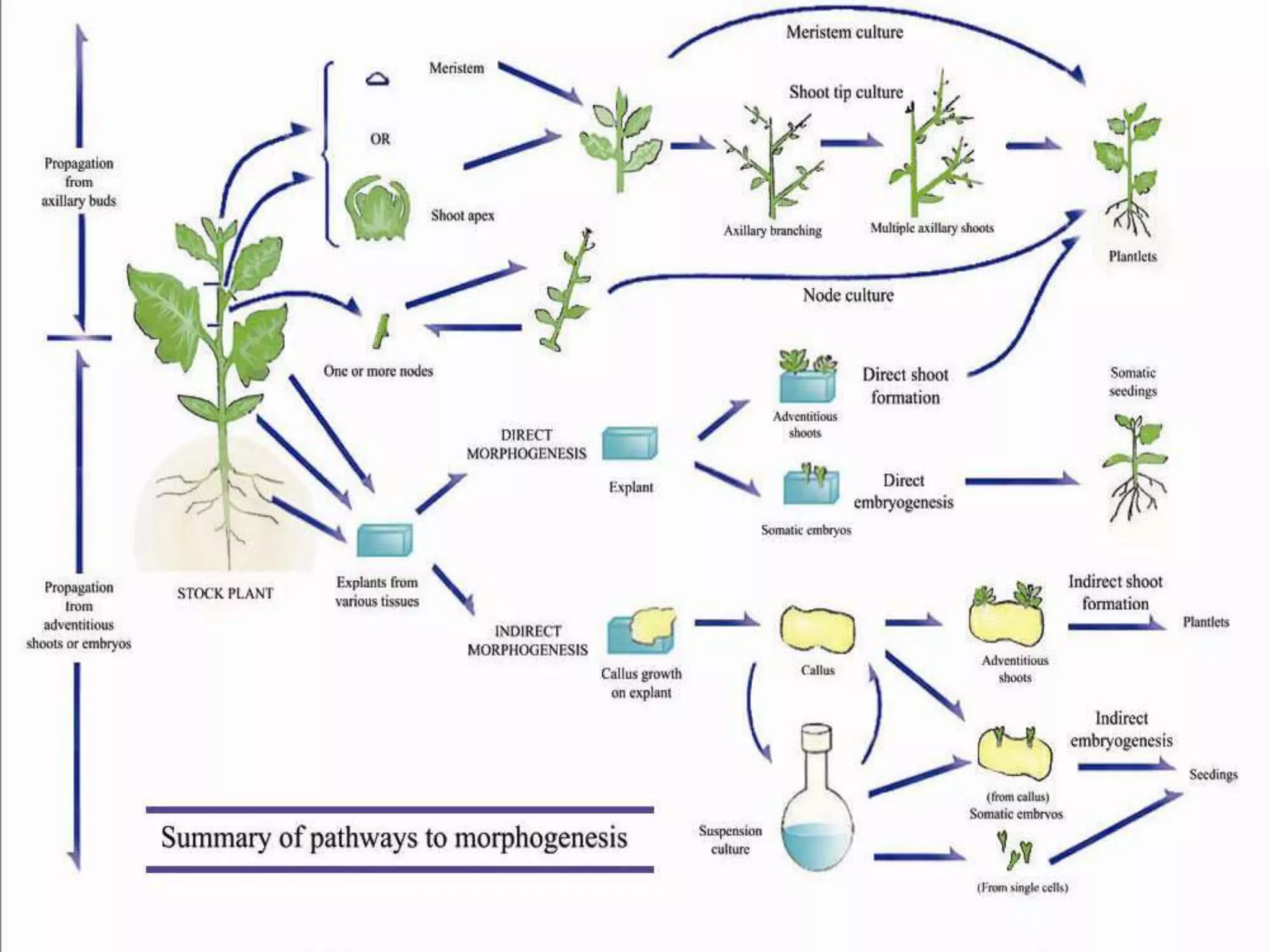
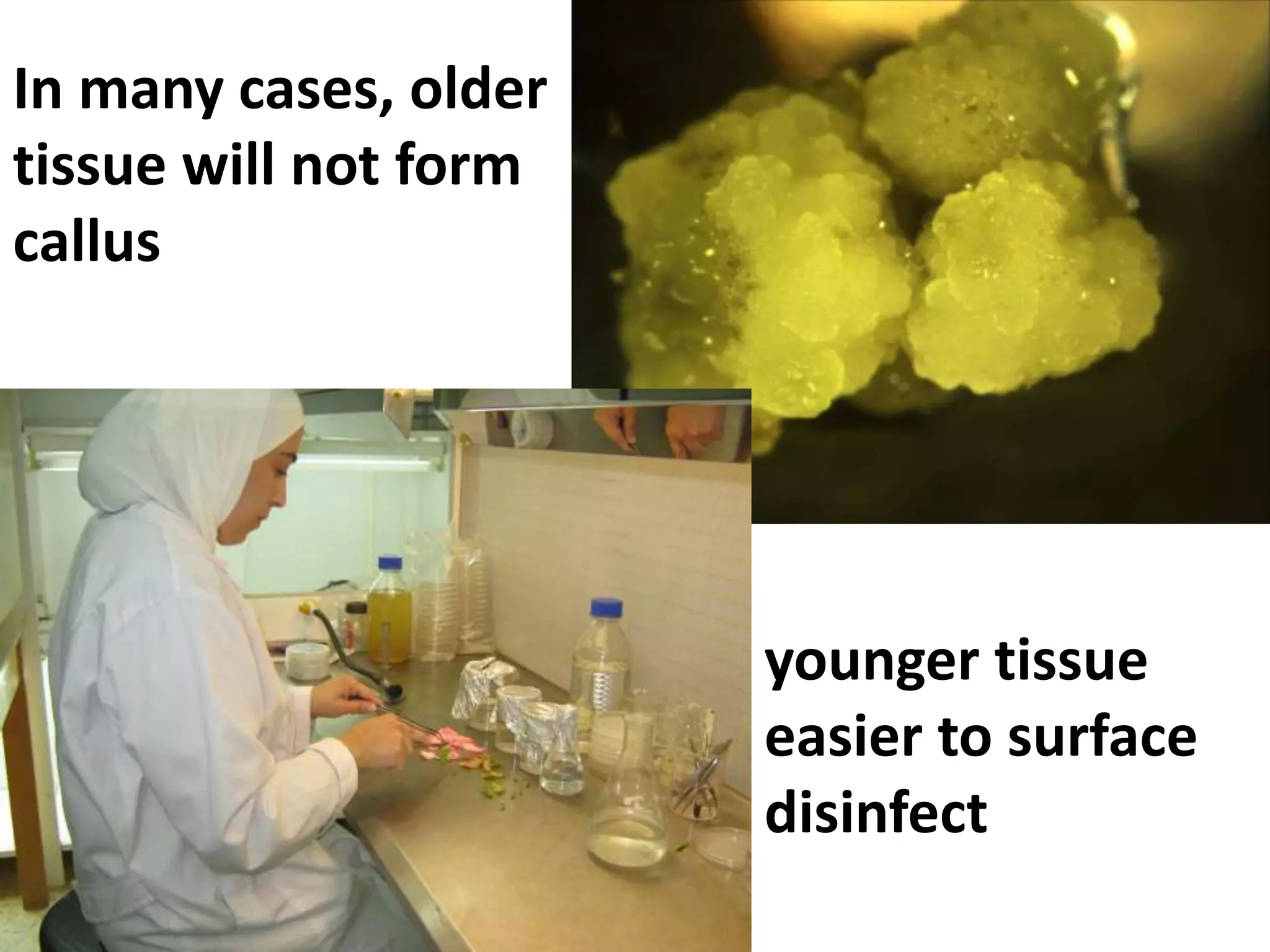
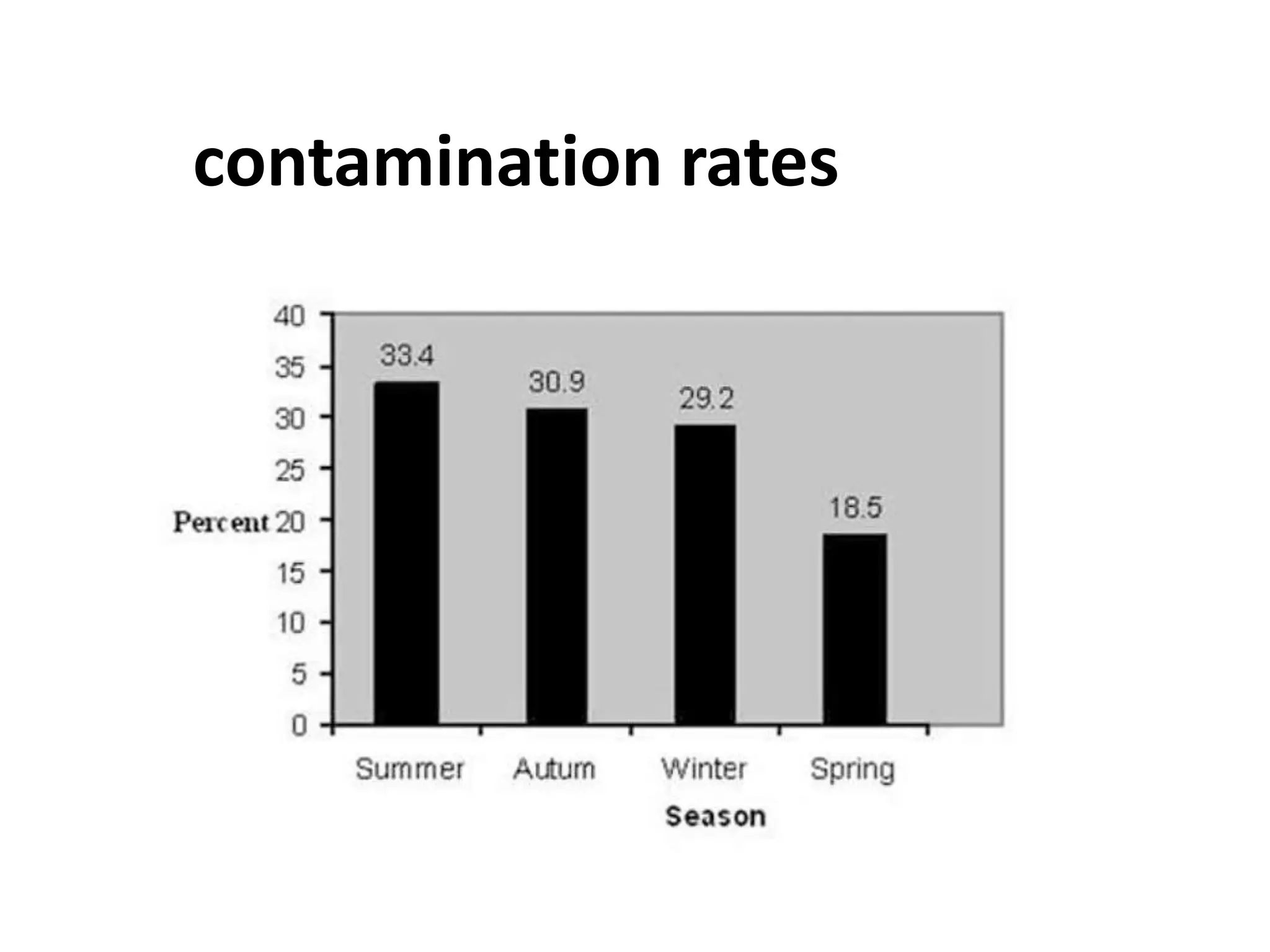
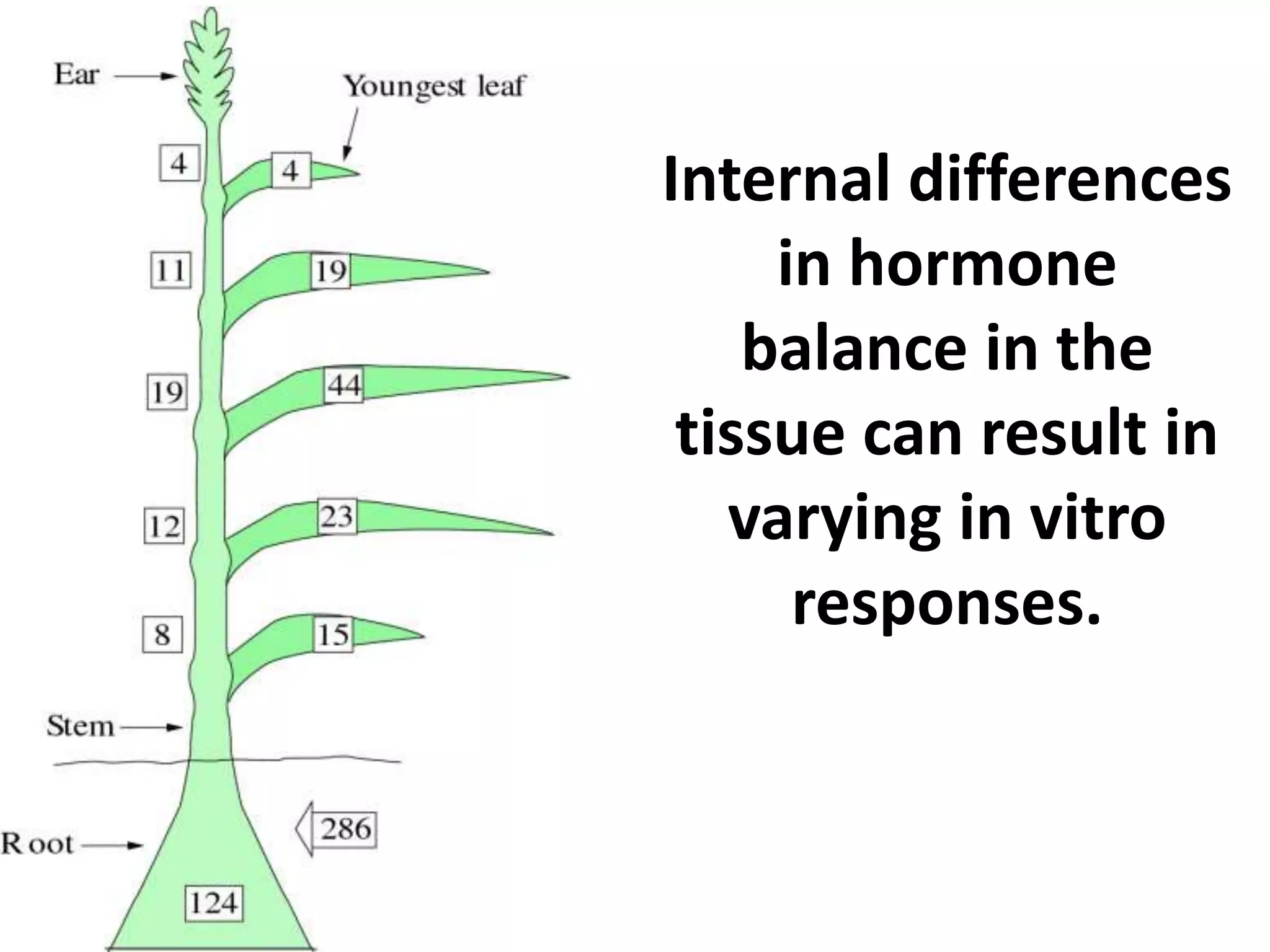
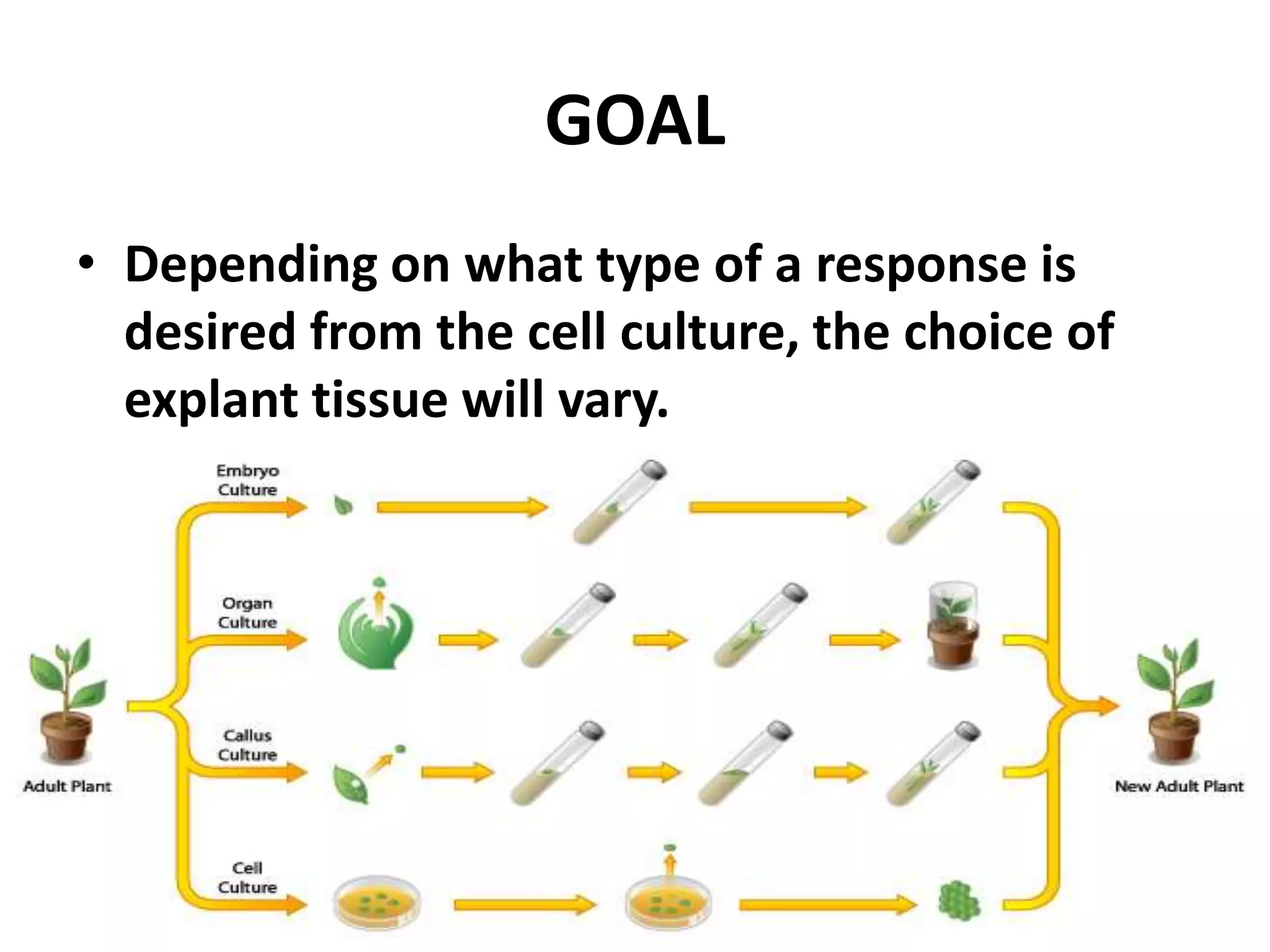
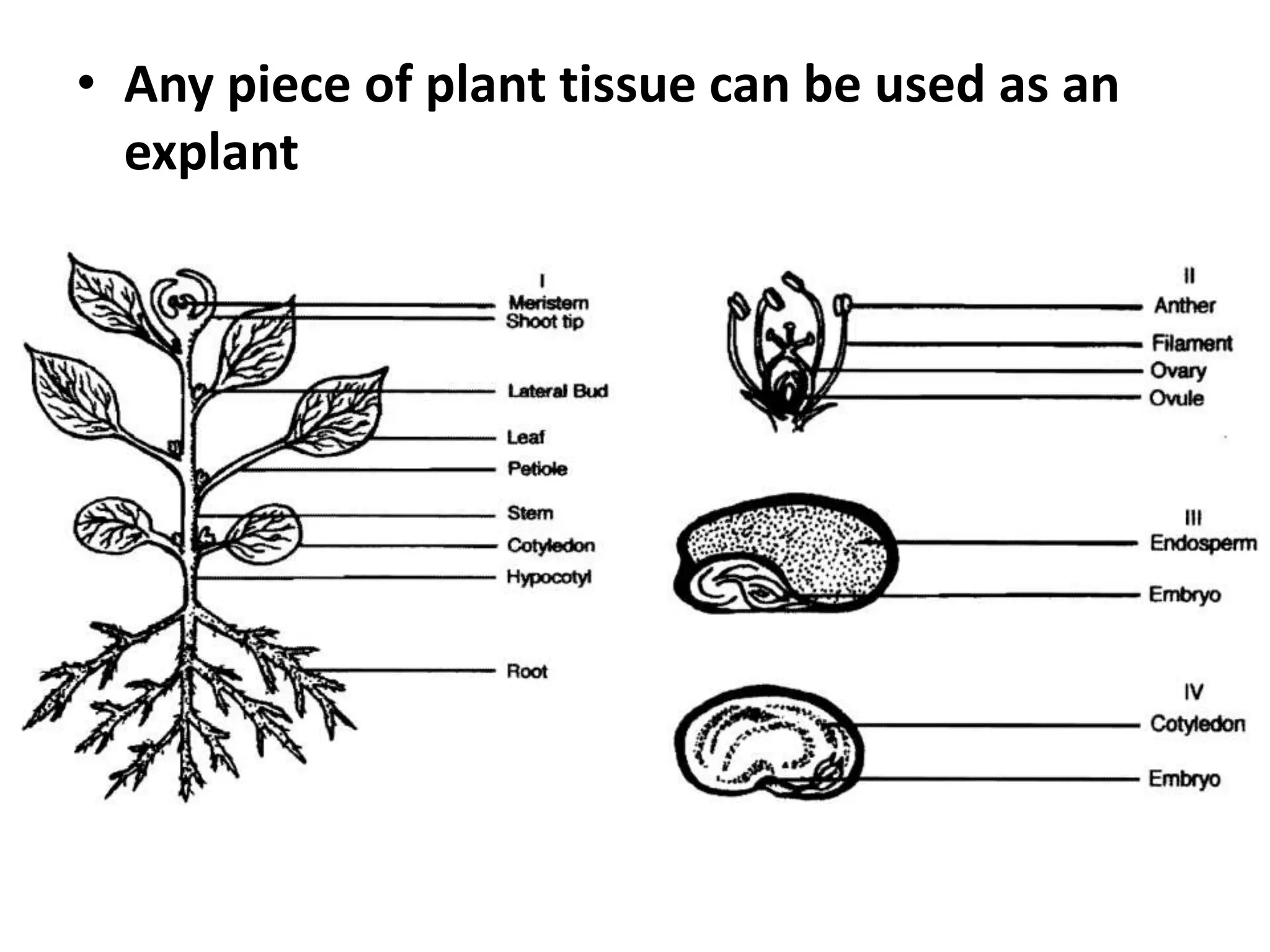
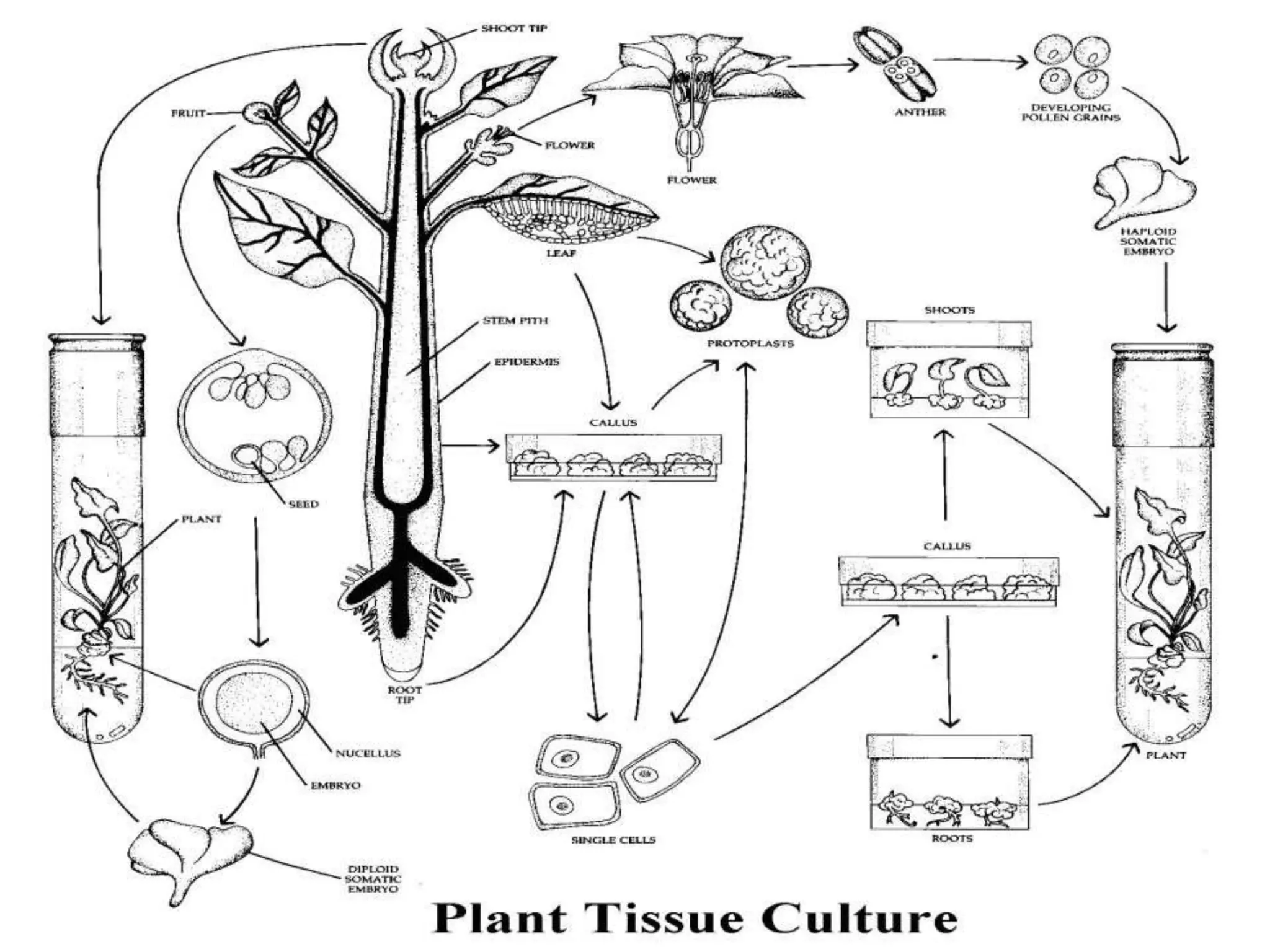
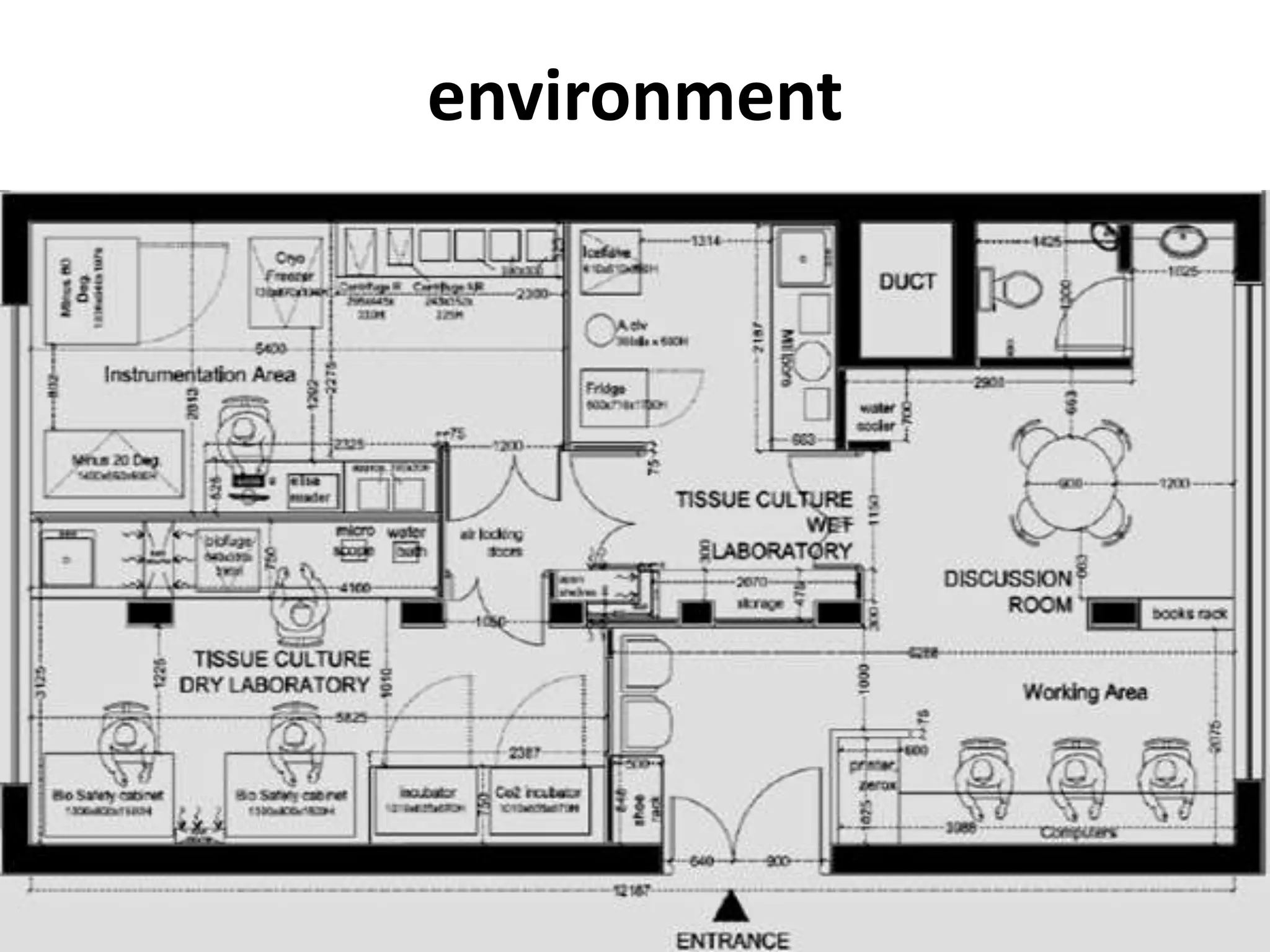
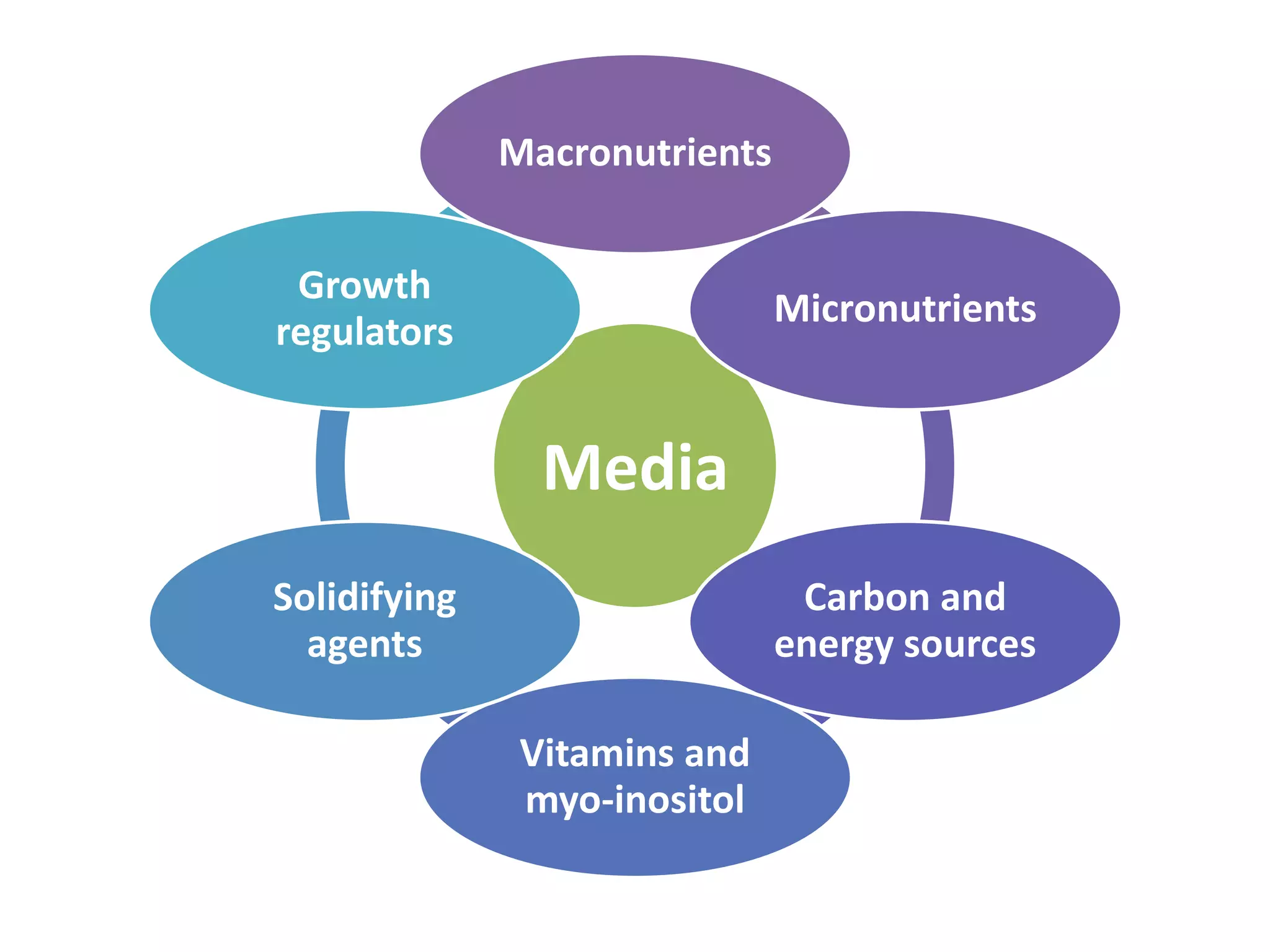
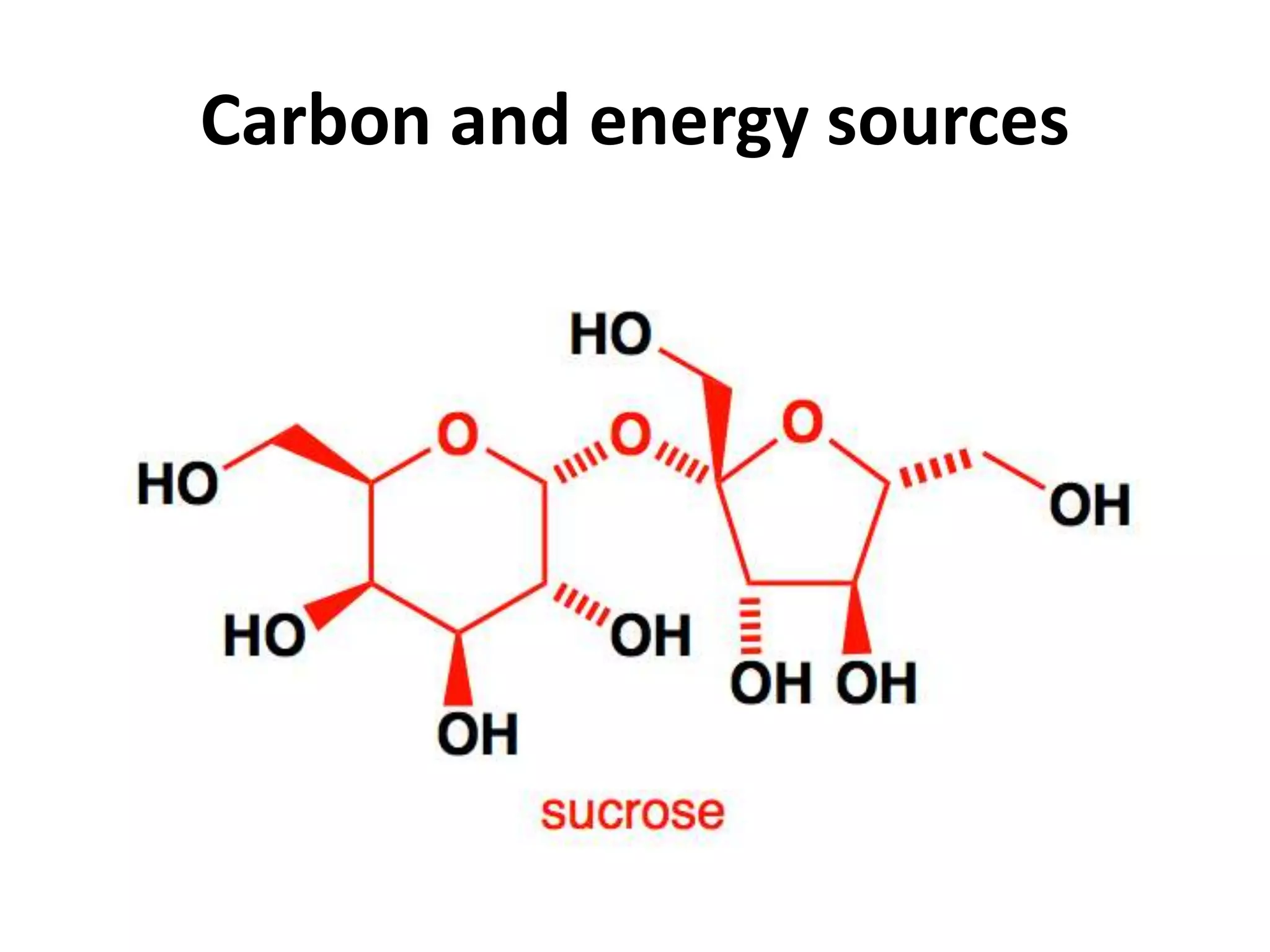
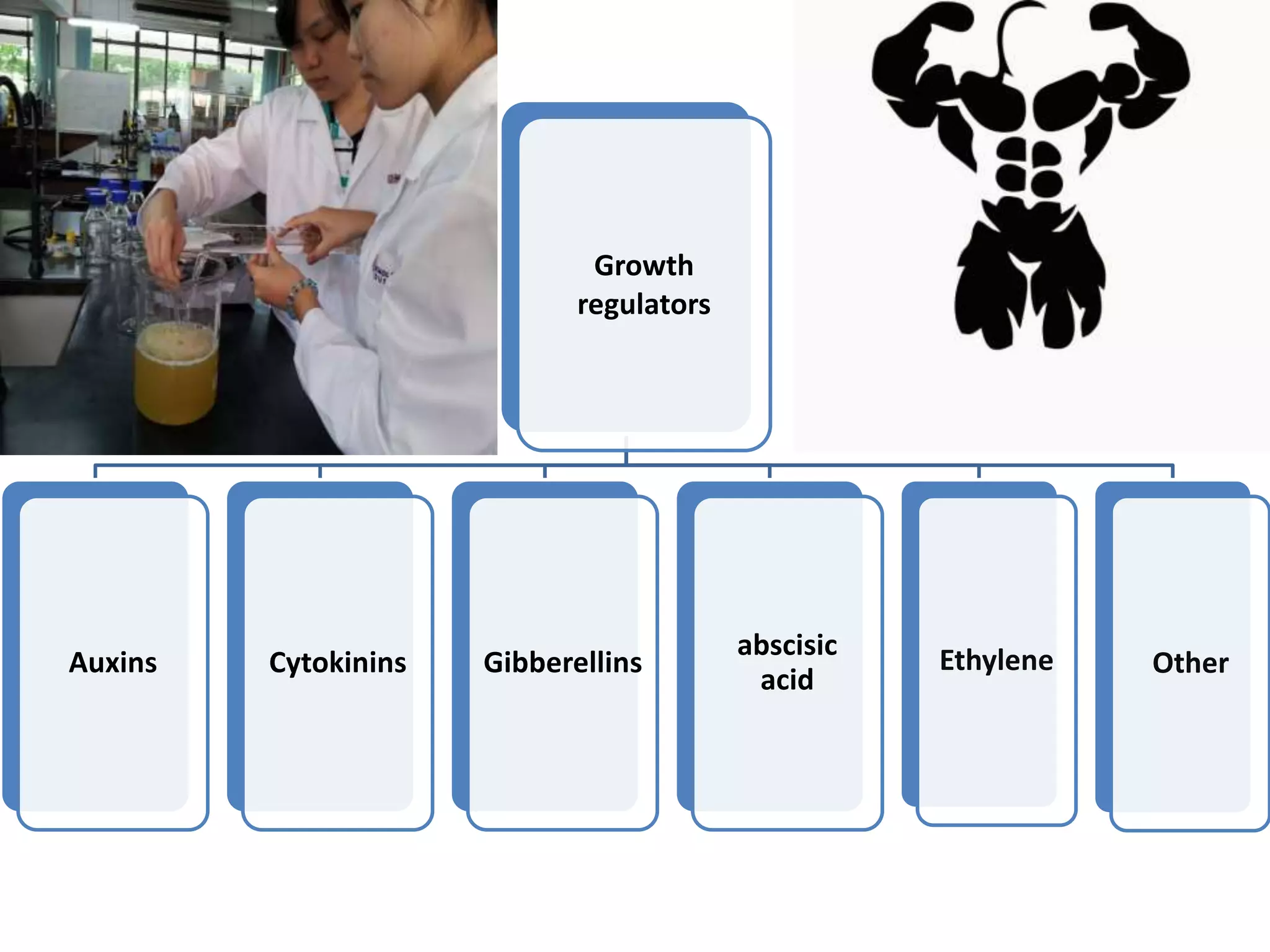
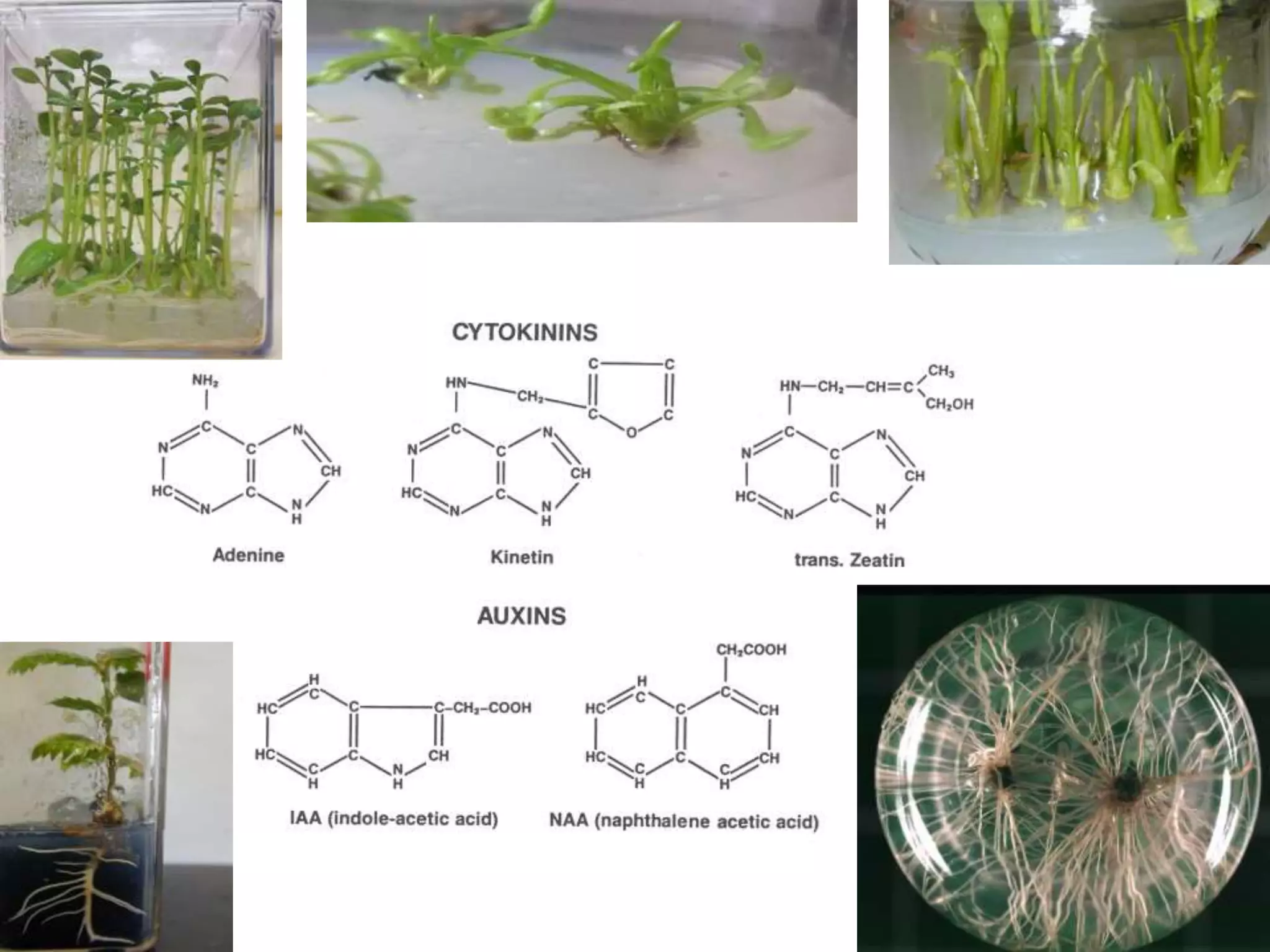
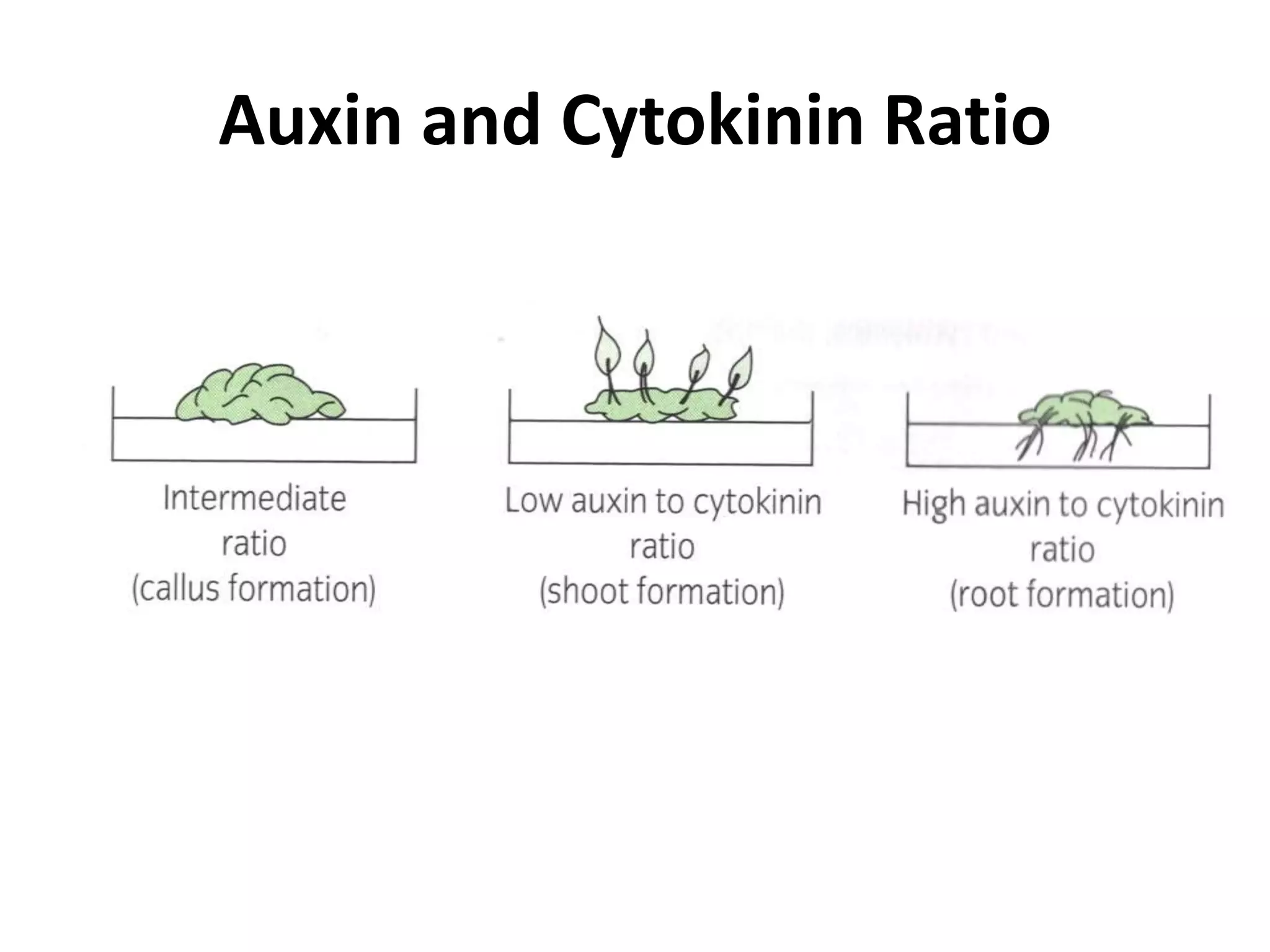
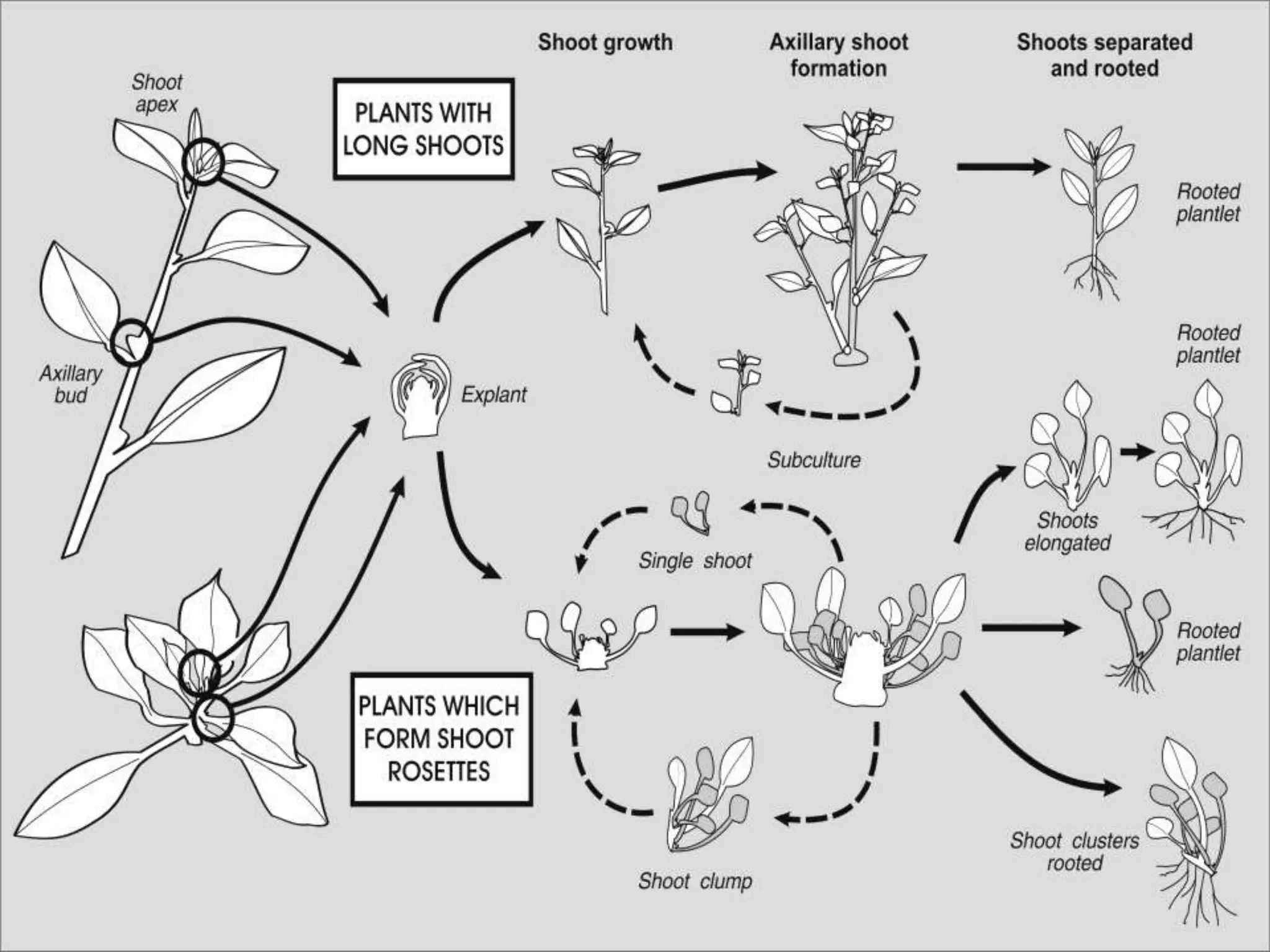
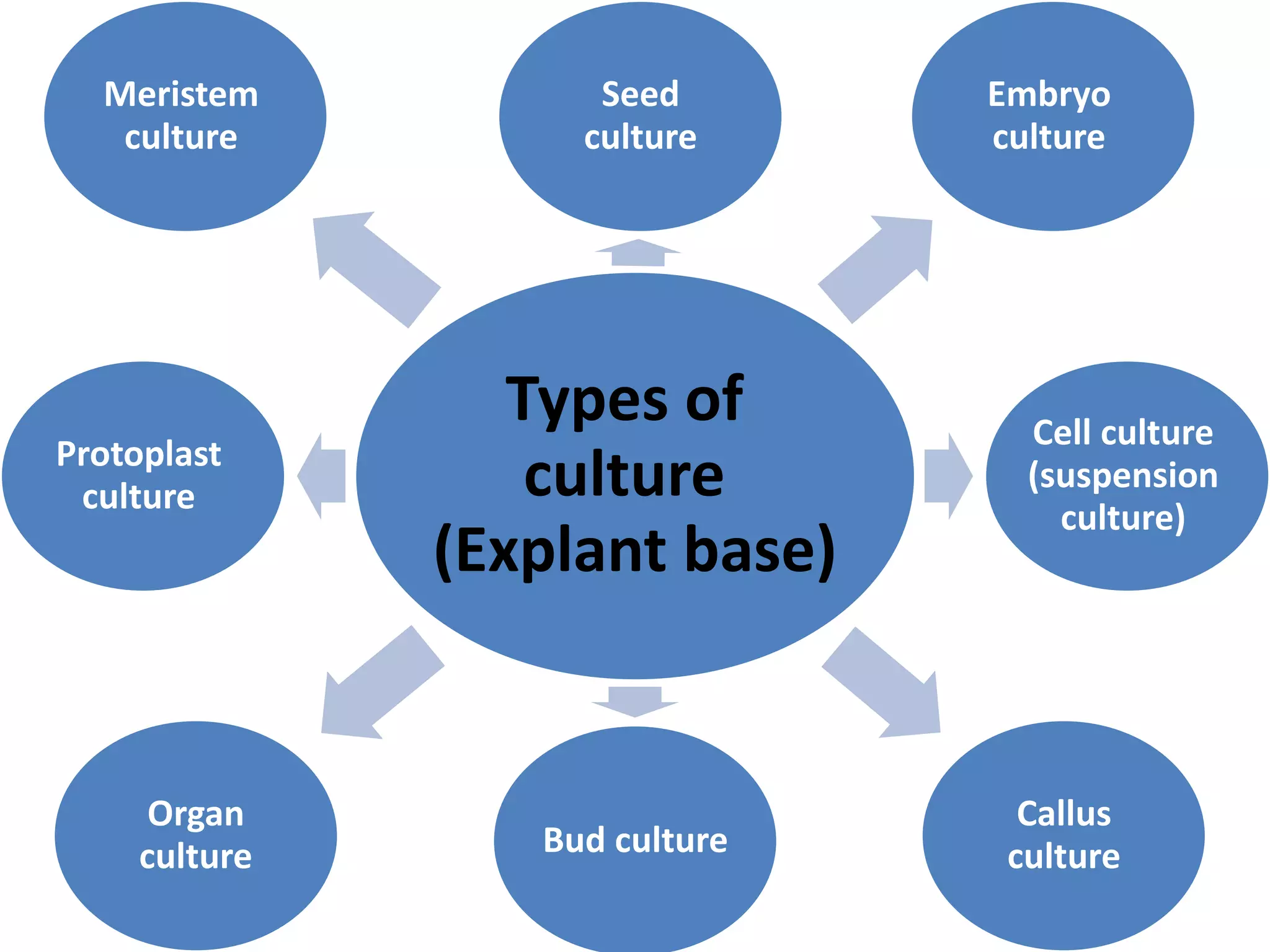
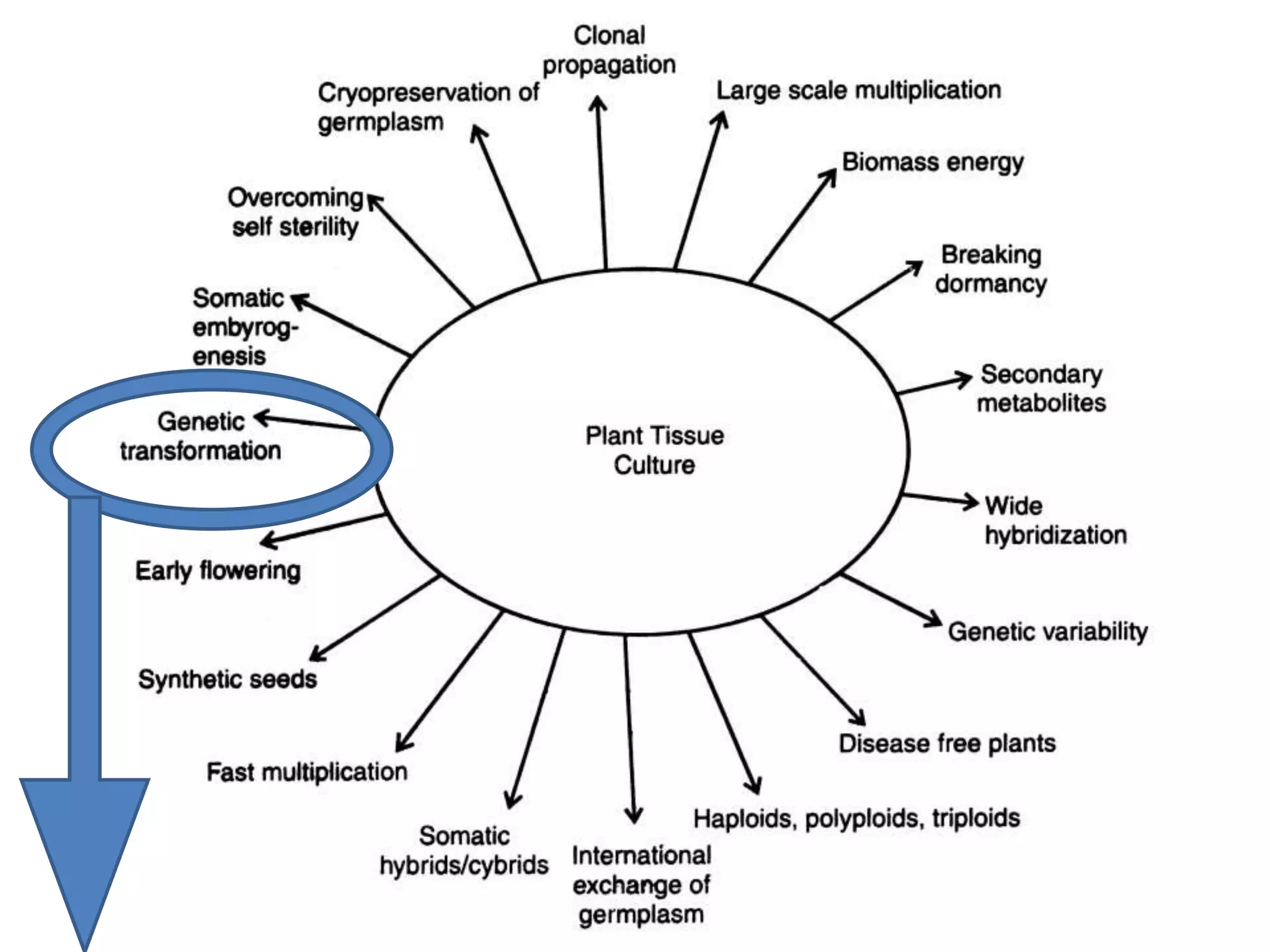
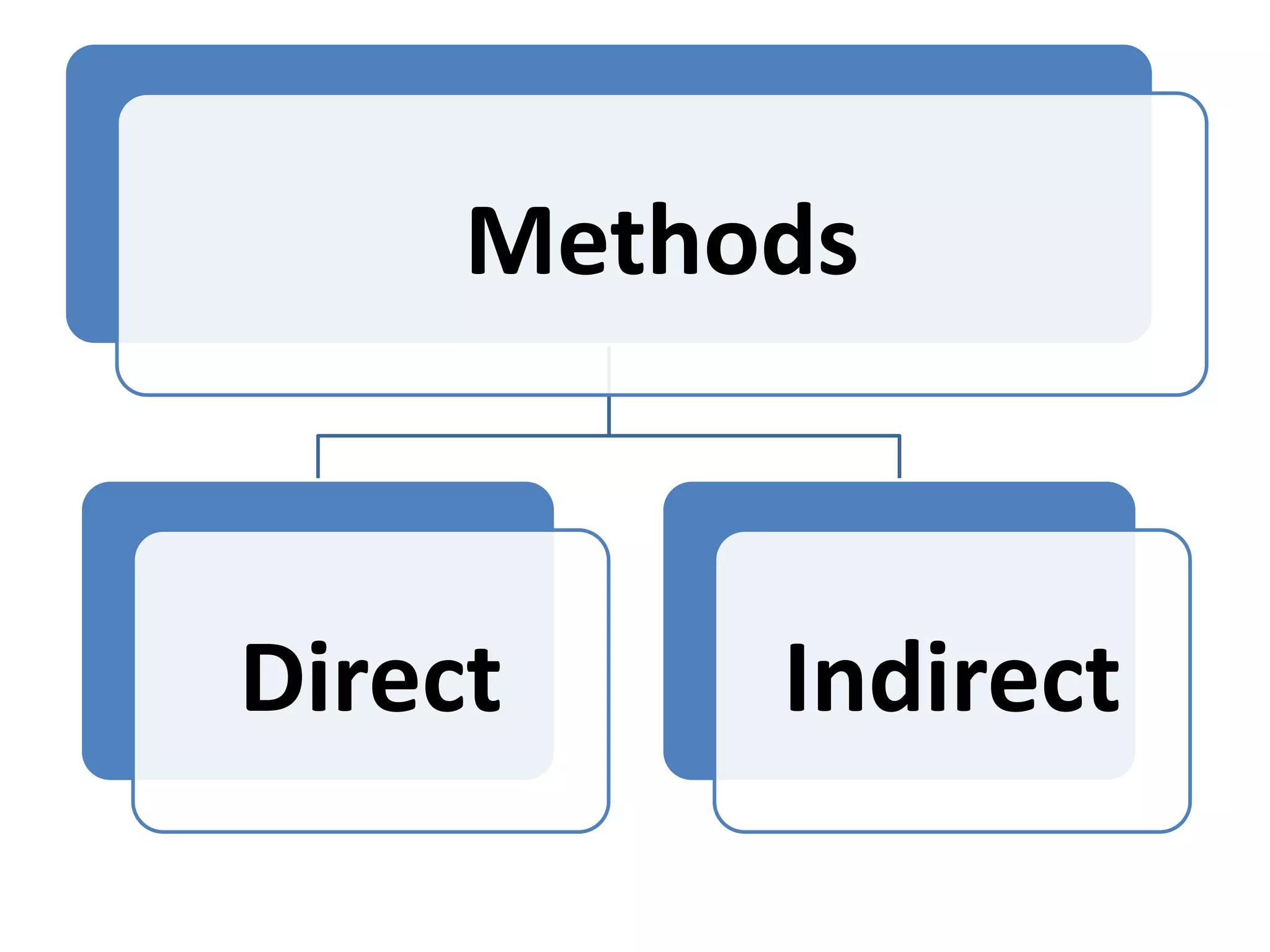
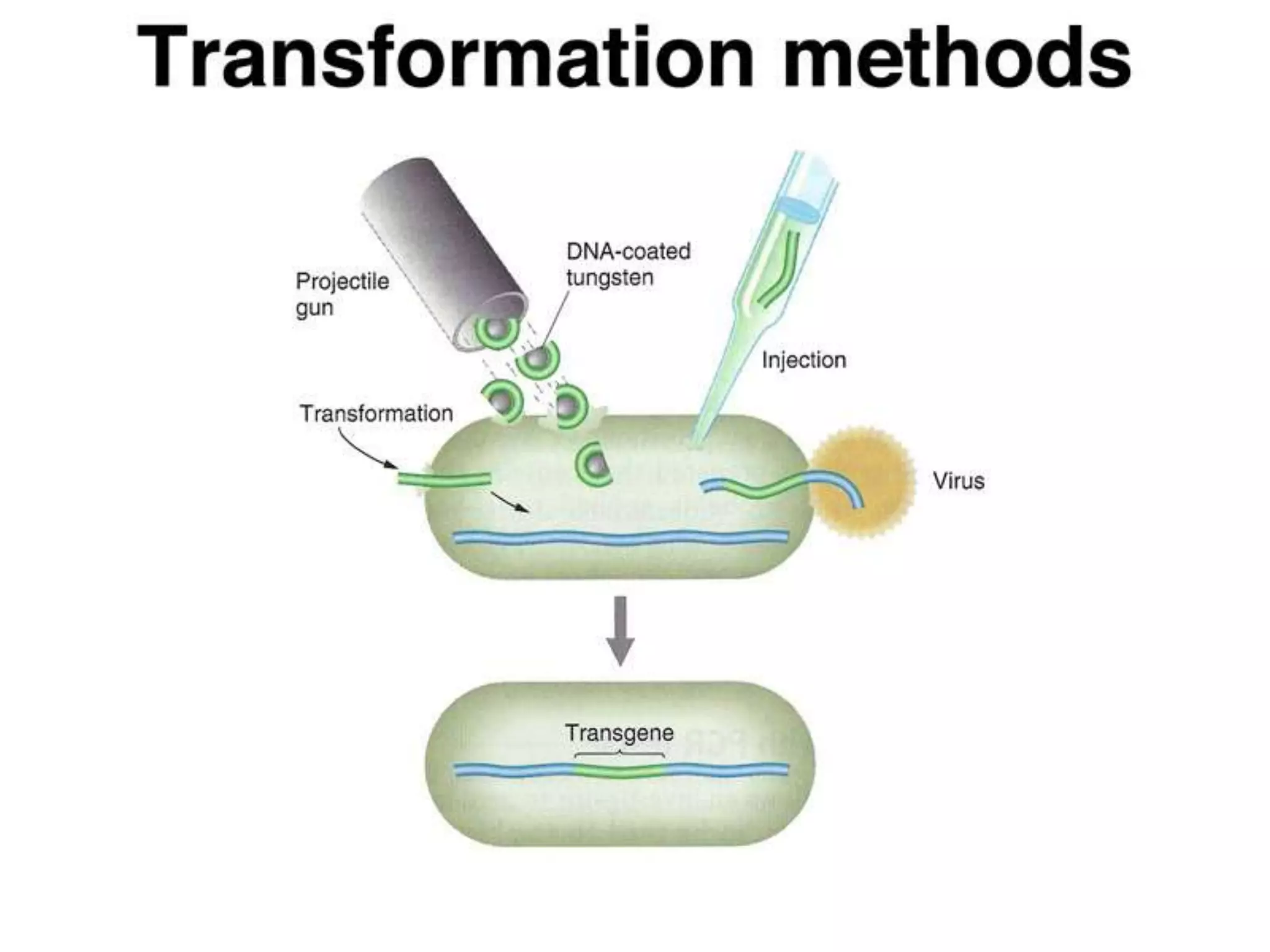
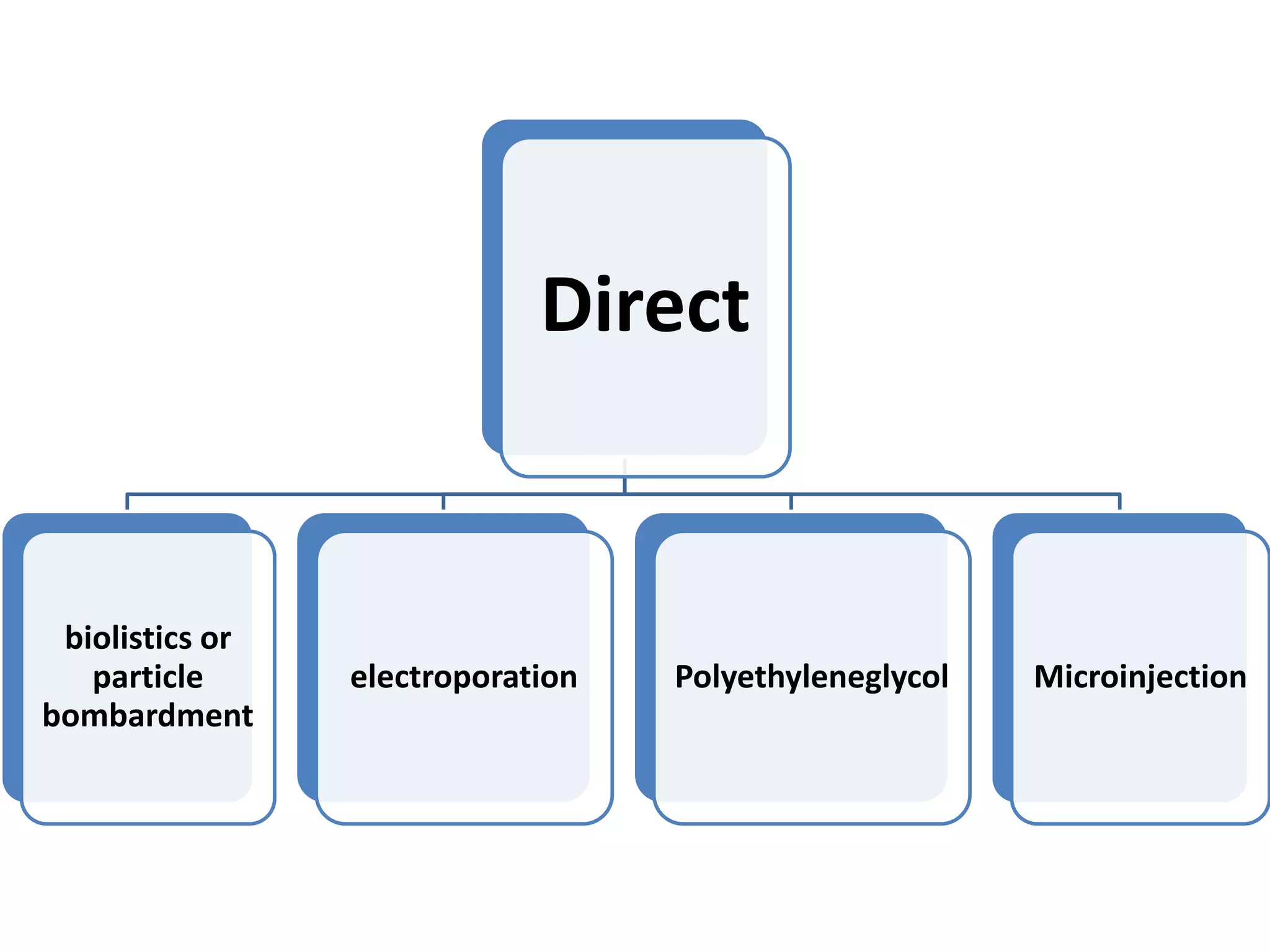
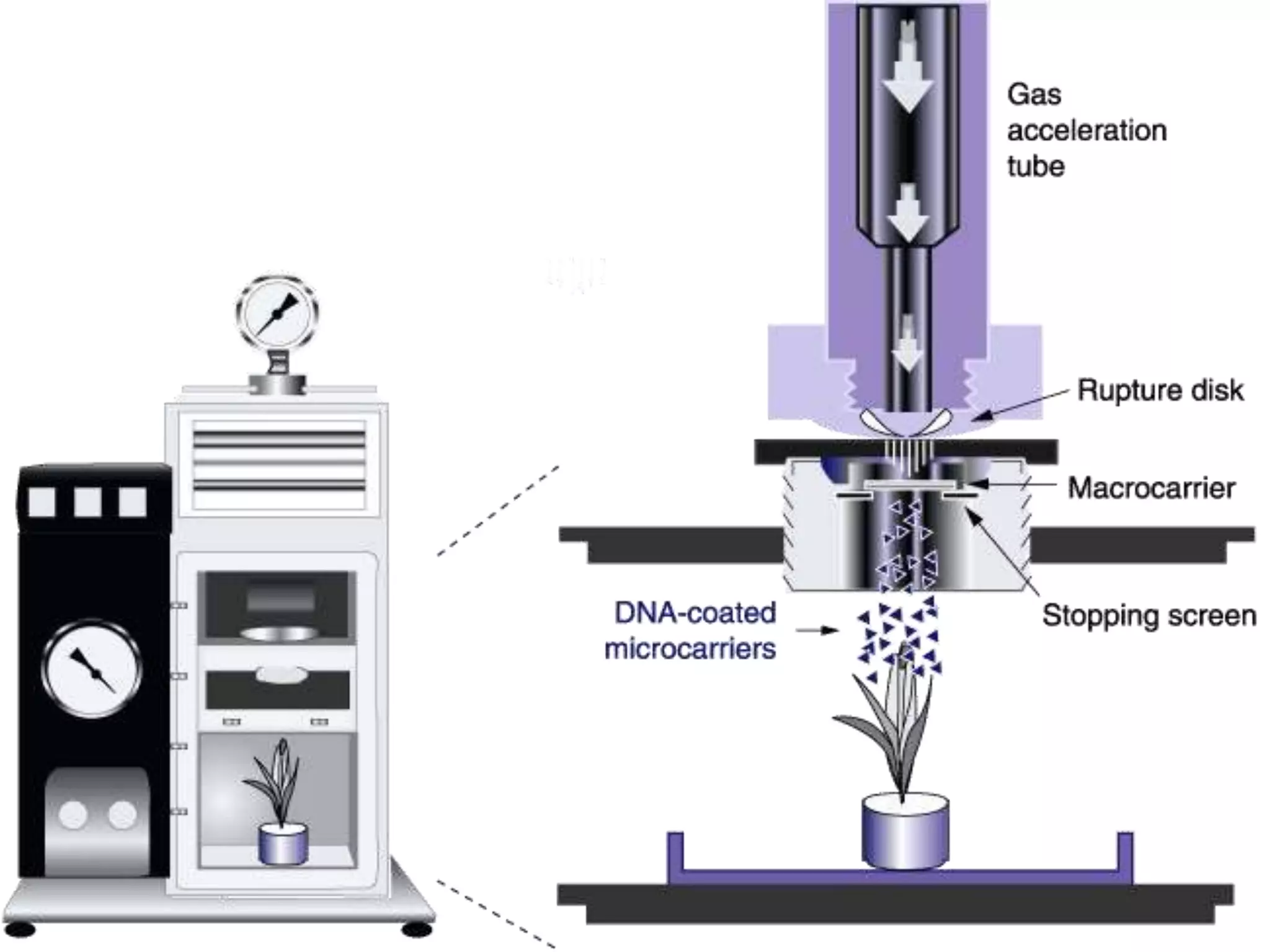
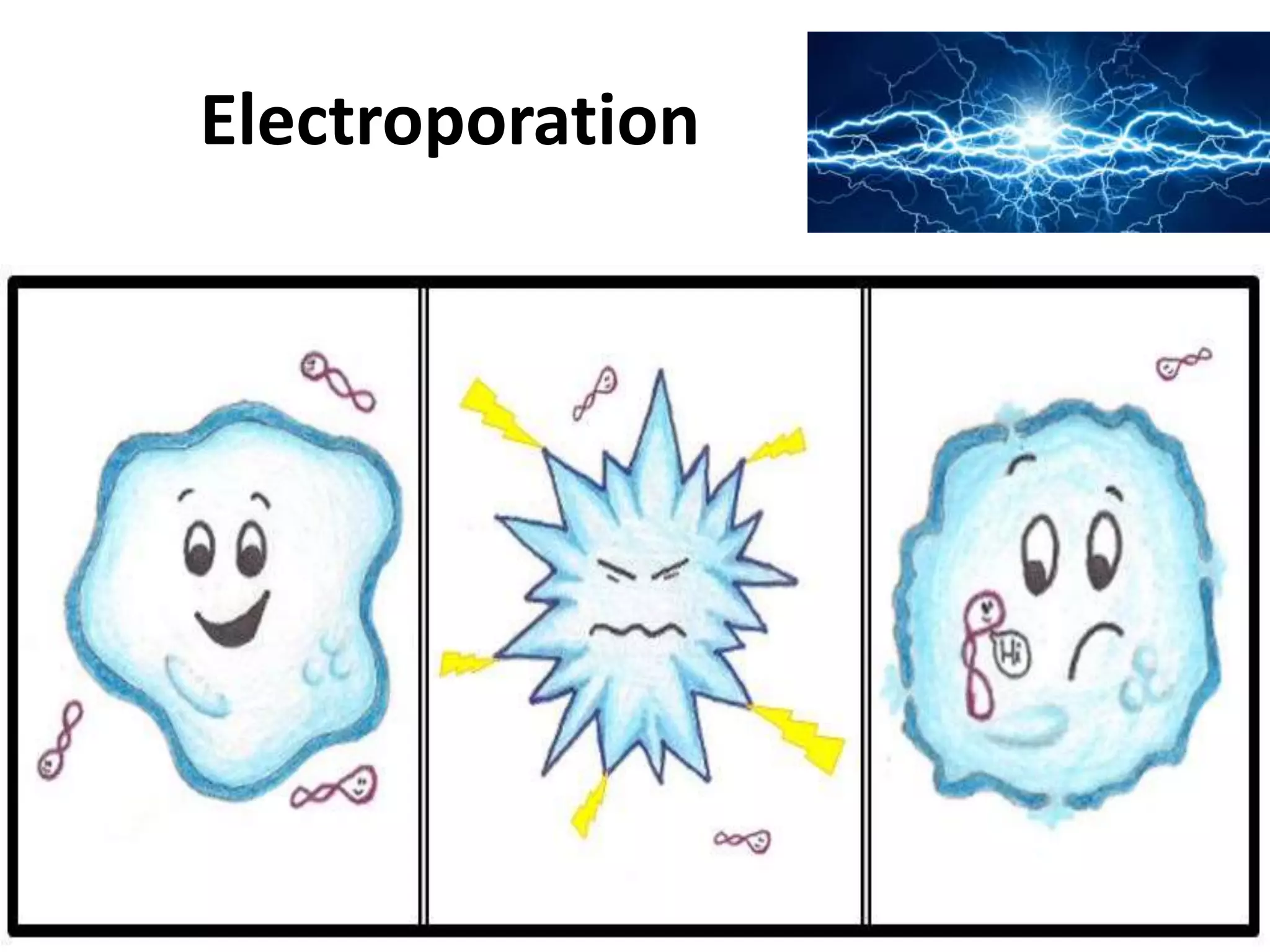
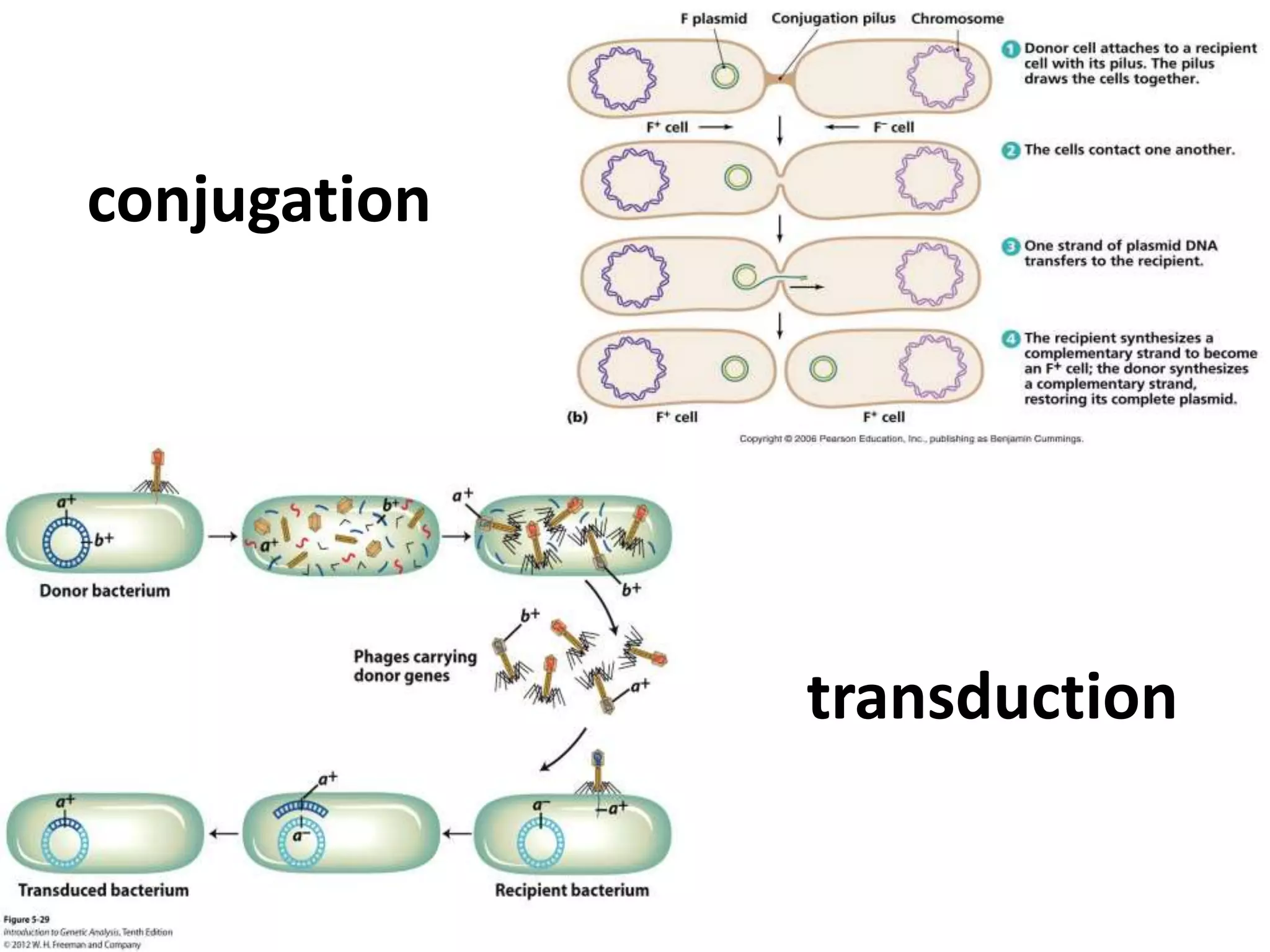
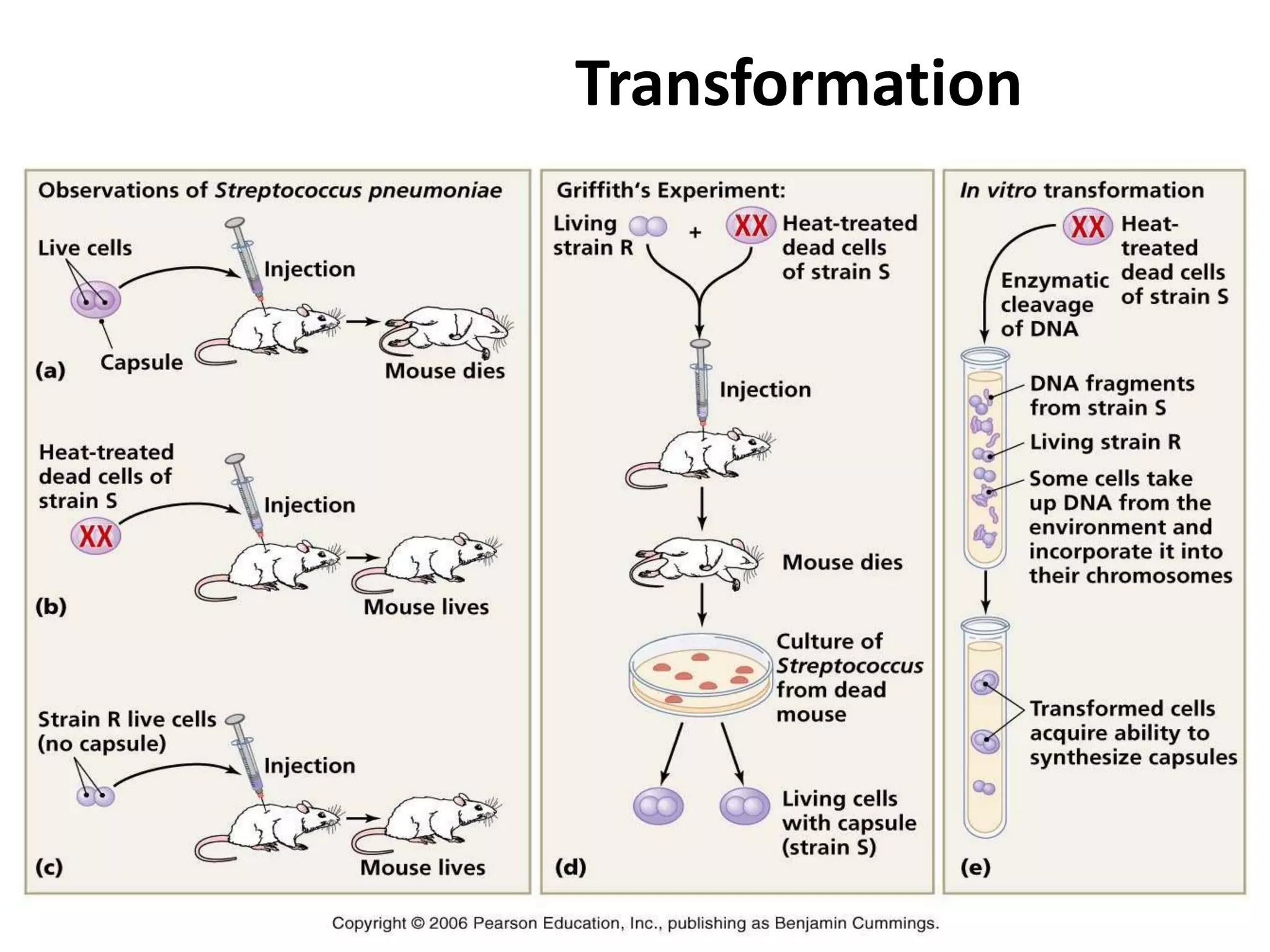
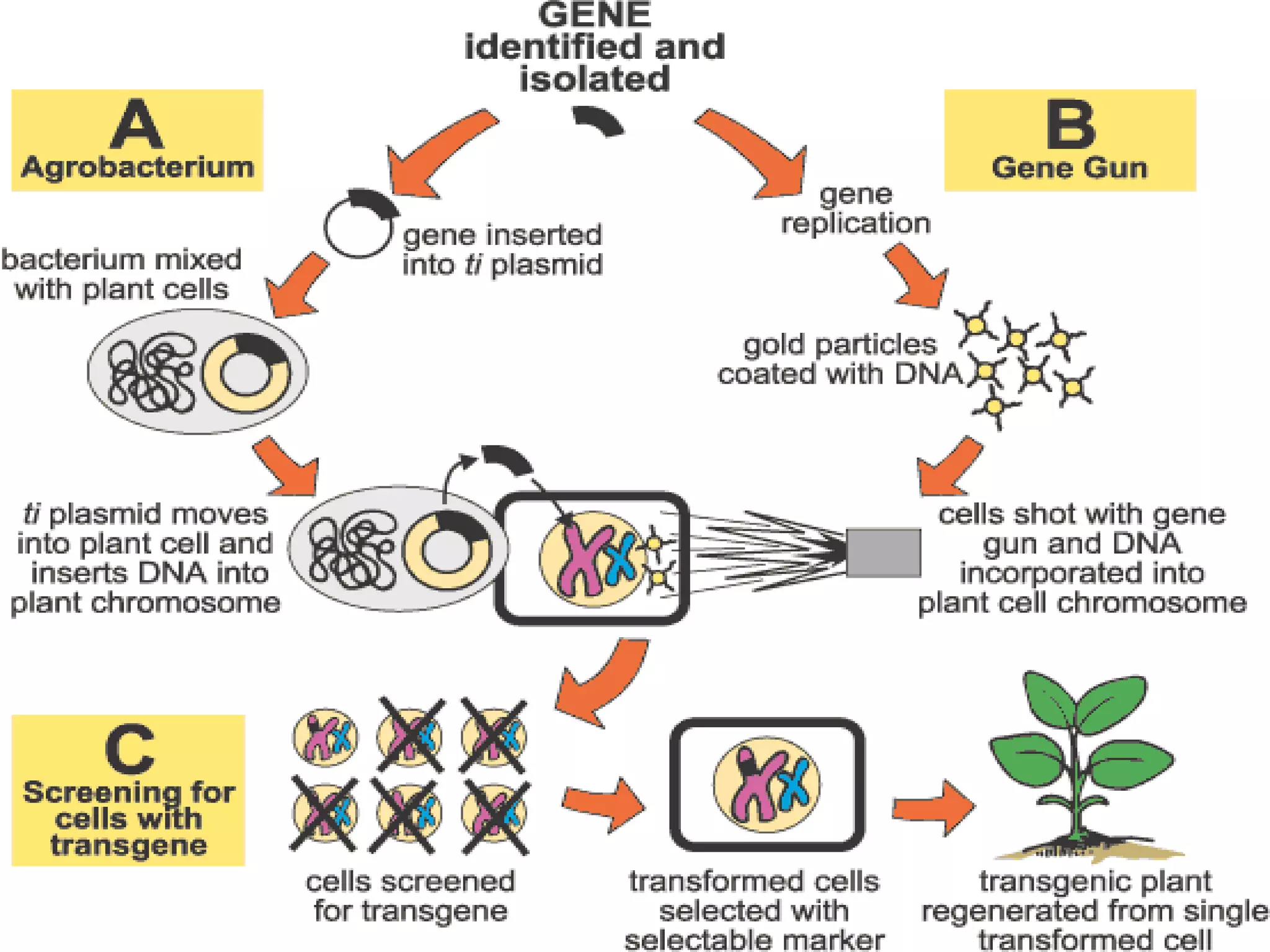
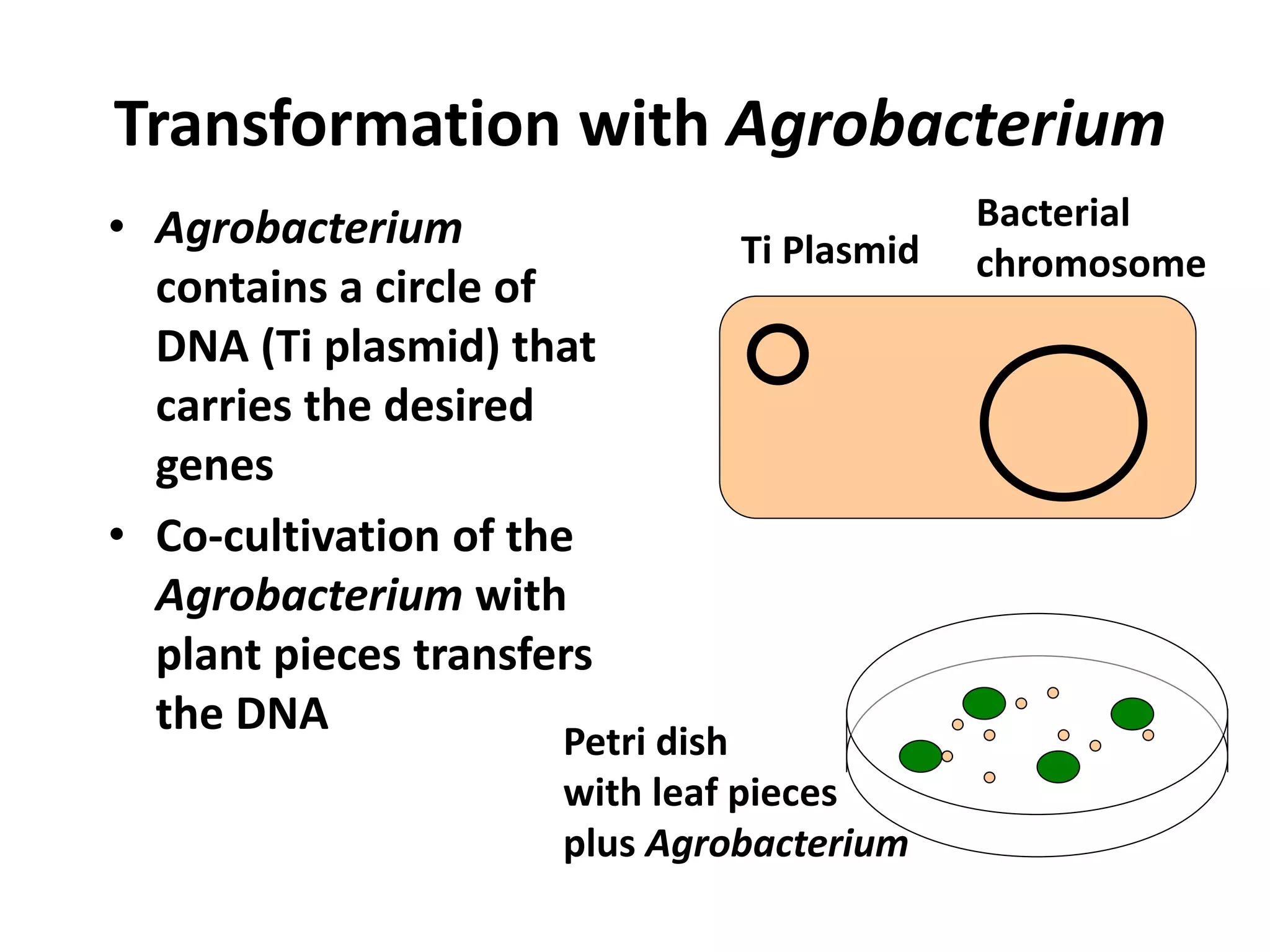
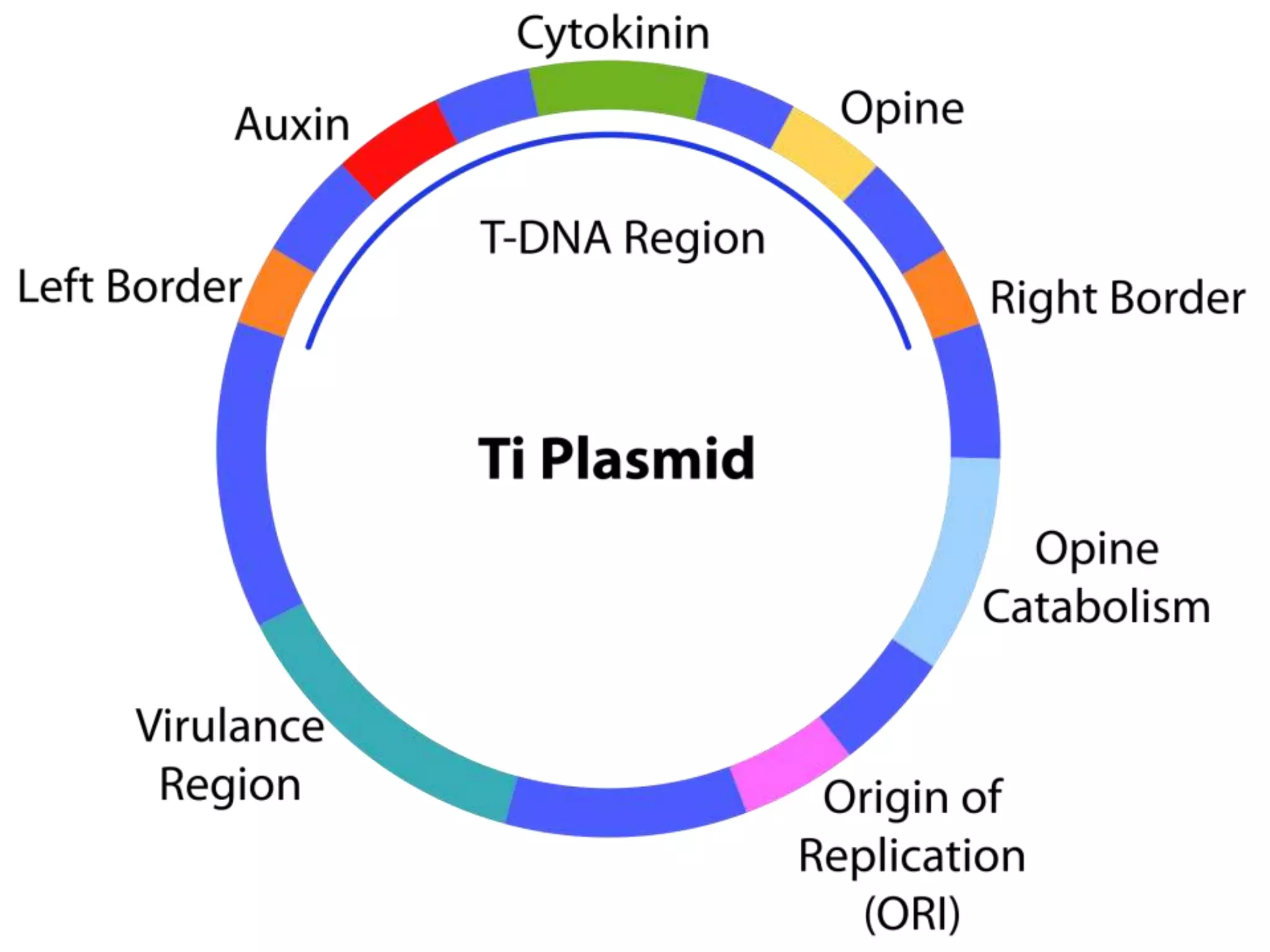
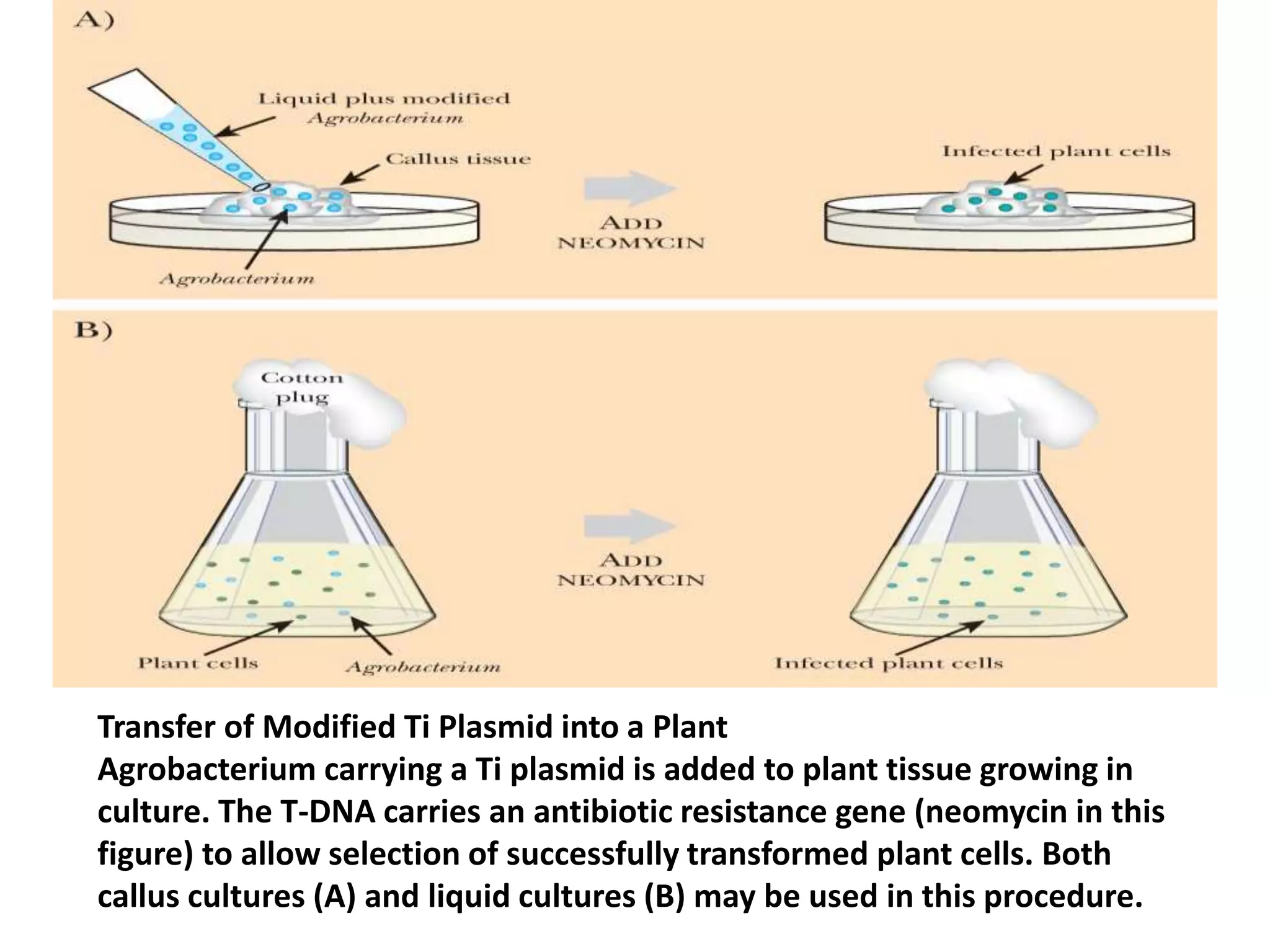
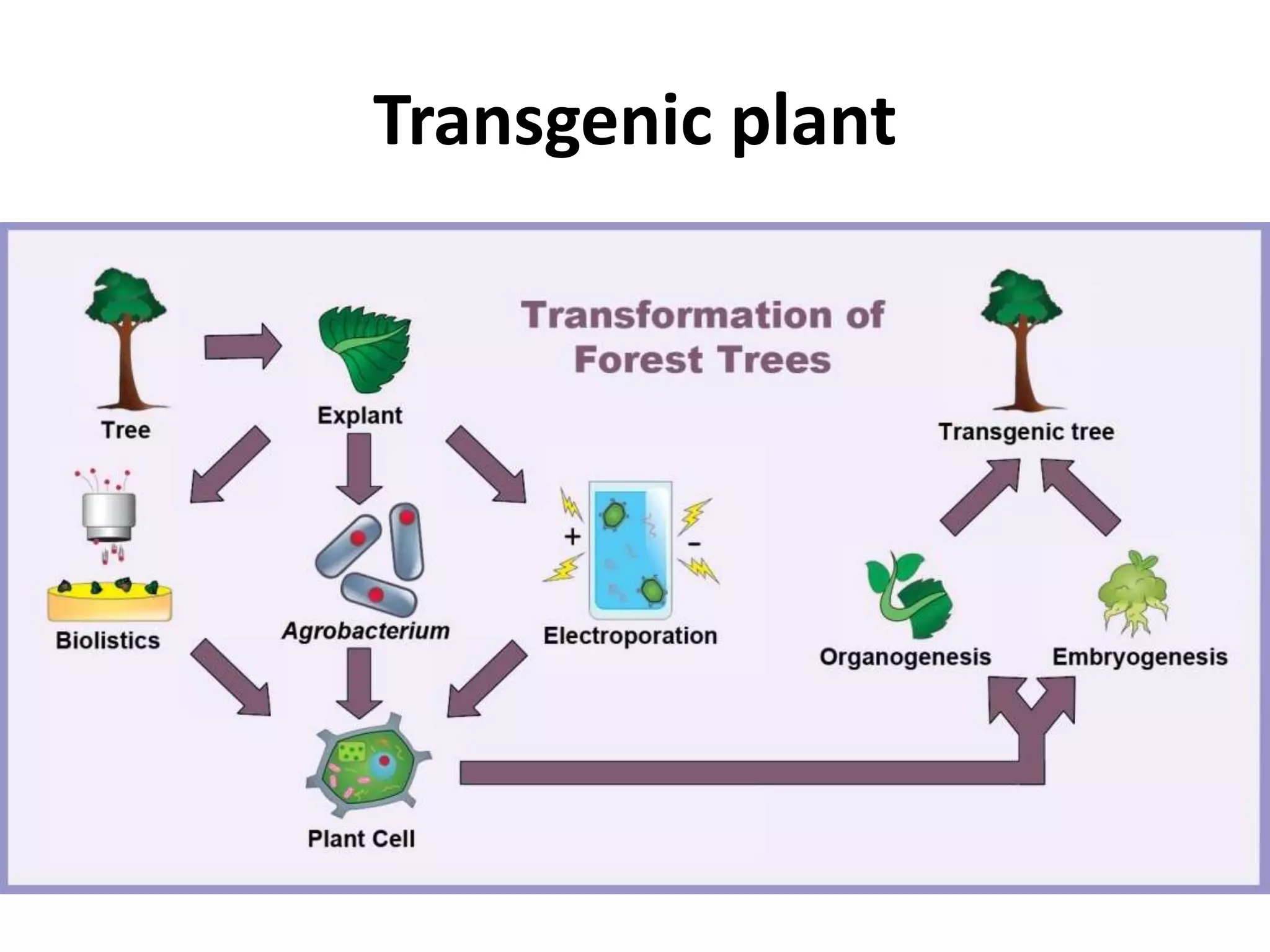
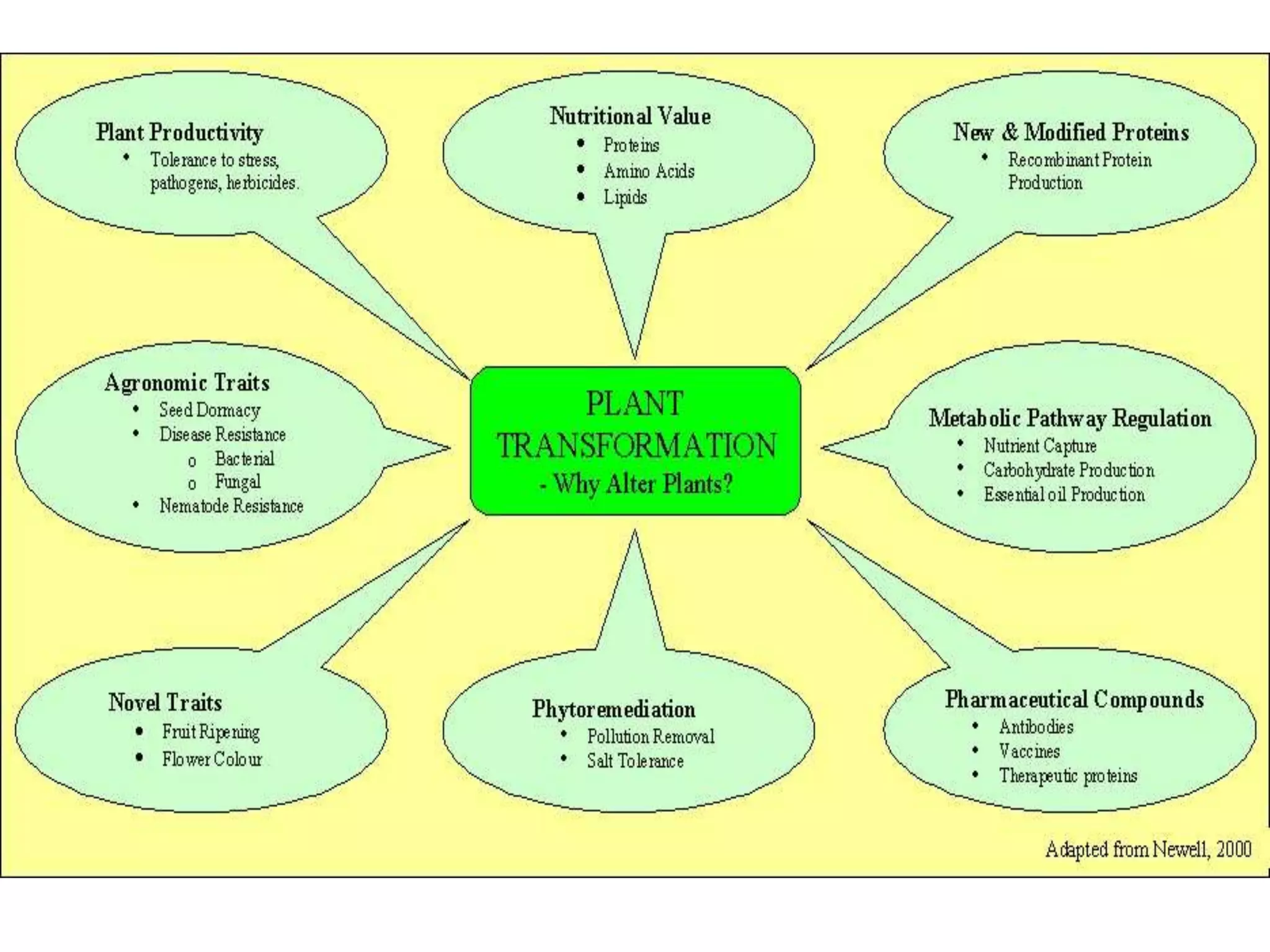
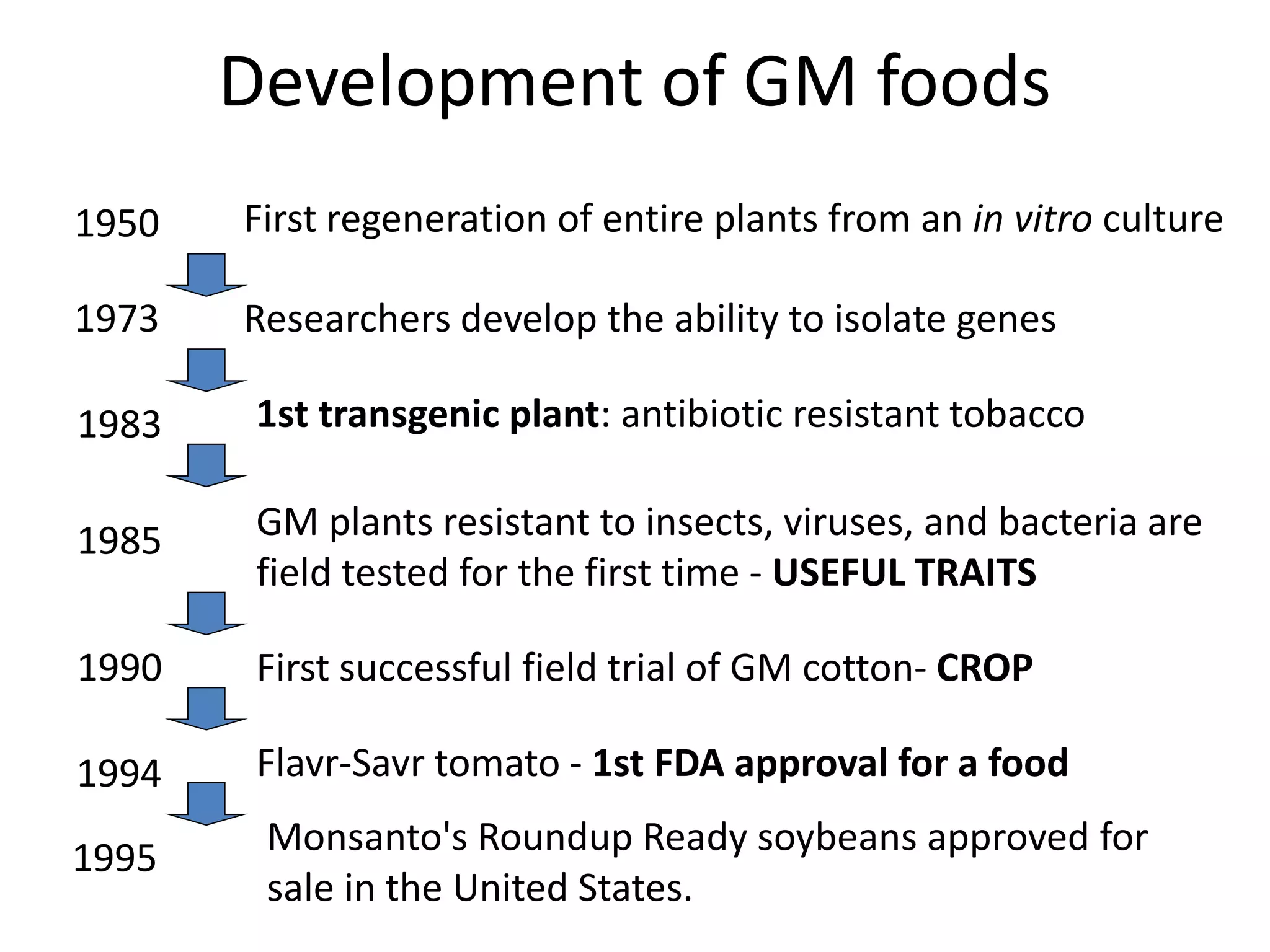
This document summarizes micropropagation techniques used to rapidly multiply plants in vitro. It discusses explant selection, initiation and establishment of aseptic cultures, multiplication of plantlets, and acclimatization. Micropropagation allows for mass production of disease-free plants through small propagules in controlled environments. The document also covers plant genetic transformation methods using Agrobacterium tumefaciens to introduce exogenous DNA and produce transgenic plants with desired traits.