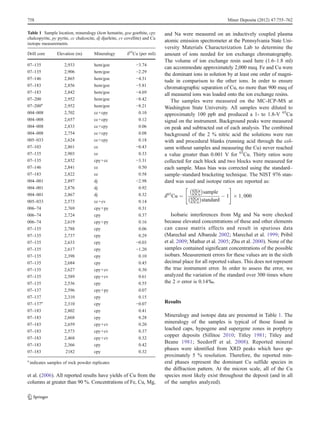
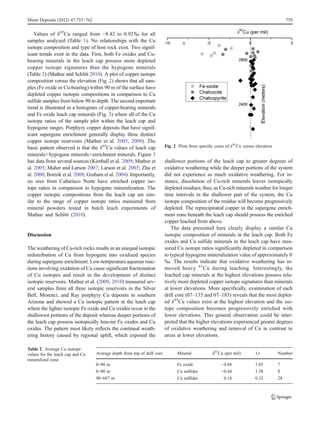
The copper isotope ratios were measured in 42 mineral separates from the Cañariaco Norte porphyry copper deposit in northern Peru. Isotopic ratios ranged from -8.42‰ to 0.61‰, with near-surface iron oxides having the most depleted values and deeper chalcocite, covellite, and chalcopyrite having more typical hypogene ratios. The data indicate that most of the chalcocite and covellite formed from high-temperature hypogene processes, with only a minor portion resulting from supergene enrichment. The lack of an enriched isotopic signature suggests little potential for an extensive supergene enrichment blanket at depth due to the weathering history.