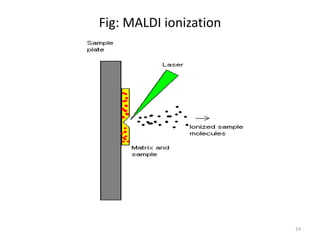
The document discusses various ionization techniques used in mass spectrometry. It describes how samples must be ionized before being introduced into the mass spectrometer's vacuum system. The techniques discussed include electron ionization (EI) and chemical ionization (CI) for gas-phase samples, and fast atom bombardment (FAB), electrospray ionization (ESI), and matrix-assisted laser desorption ionization (MALDI) for condensed-phase samples. Each technique has advantages and limitations depending on the type of sample being analyzed and information required.















![2-nitrophenyl octyl ether (NPOE)
. C14H21NO3, m/z 251.1521
. only FAB matrix with no reactive hydrogen
. good choice for reactive compounds such as
organometallics
• Note: this example contains trace PEG 600
contamination
Triethanolamine
. C6H15NO3, m/z 149.1052
. good matrix for negative-ion FAB
. enhances [M-H]- formation.
16](https://image.slidesharecdn.com/presentation1-151221174636/85/Mass-Spectrometry-Ionization-Techniques-16-320.jpg)