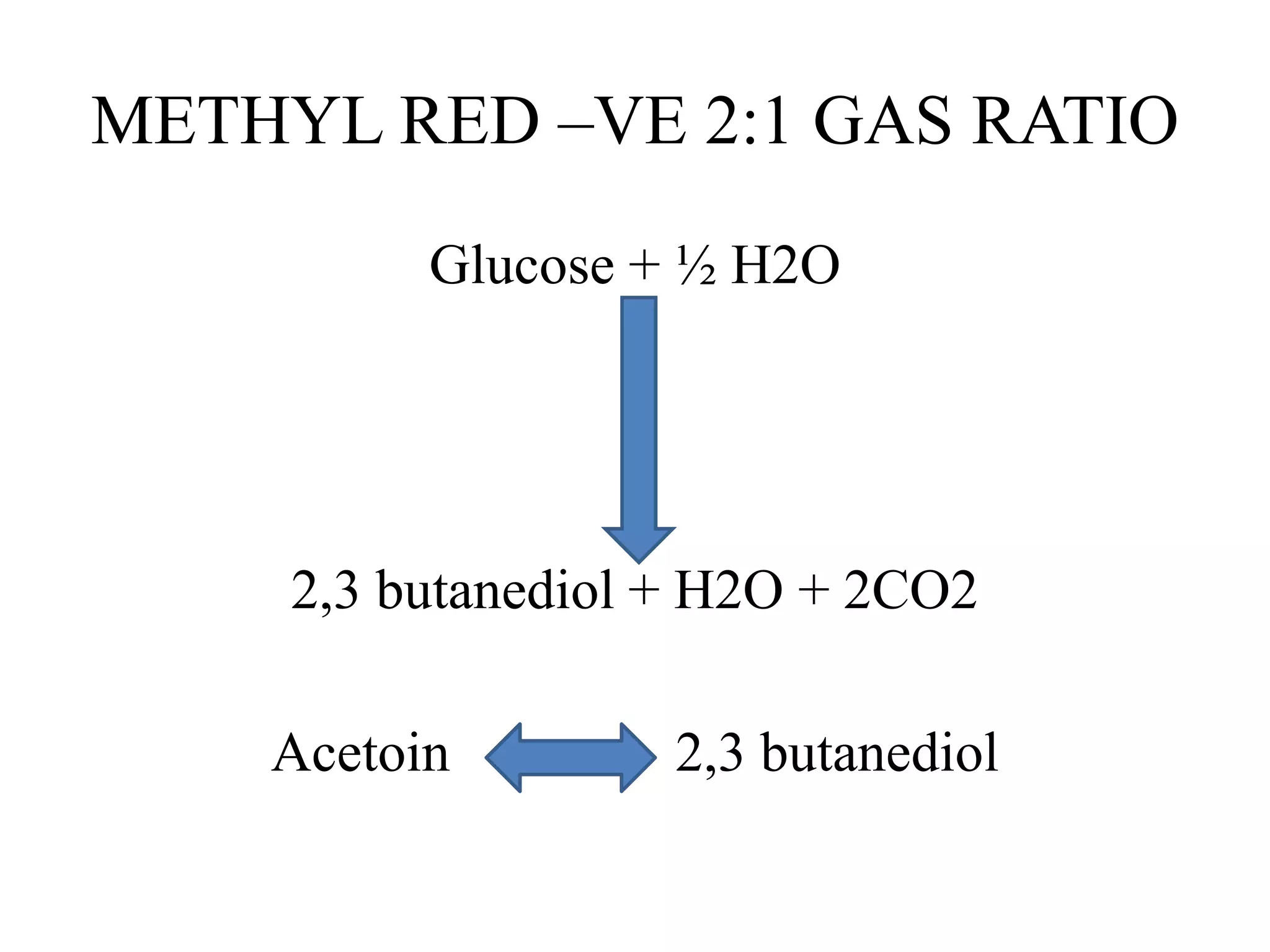
The document outlines various microbiological tests, including the indole, methyl red, Voges-Proskauer, and citrate tests. It details the principles, procedures, and interpretation criteria for each test, as well as lists of positive and negative organisms. Additionally, it provides information on media preparations, reagents, and quality control measures for accurate testing.