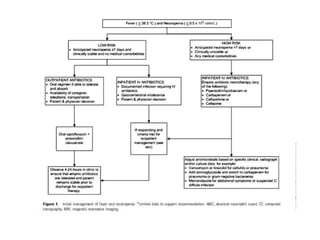
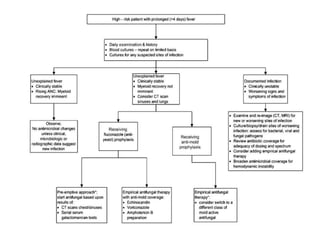
1. The IDSA guidelines provide recommendations for managing neutropenic patients with cancer who develop fever, focusing on antimicrobial treatment.
2. It distinguishes between high-risk and low-risk patients based on factors like anticipated duration of neutropenia, severity of neutropenia, and comorbidities. High-risk patients require initial IV antibiotics in the hospital, while low-risk patients may be candidates for oral or outpatient treatment.
3. The guidelines make recommendations on appropriate empiric antibiotic therapy, modifying treatment, treatment duration, and use of prophylaxis for both high-risk and low-risk neutropenic fever patients. It also provides guidance on use of empirical and preempt