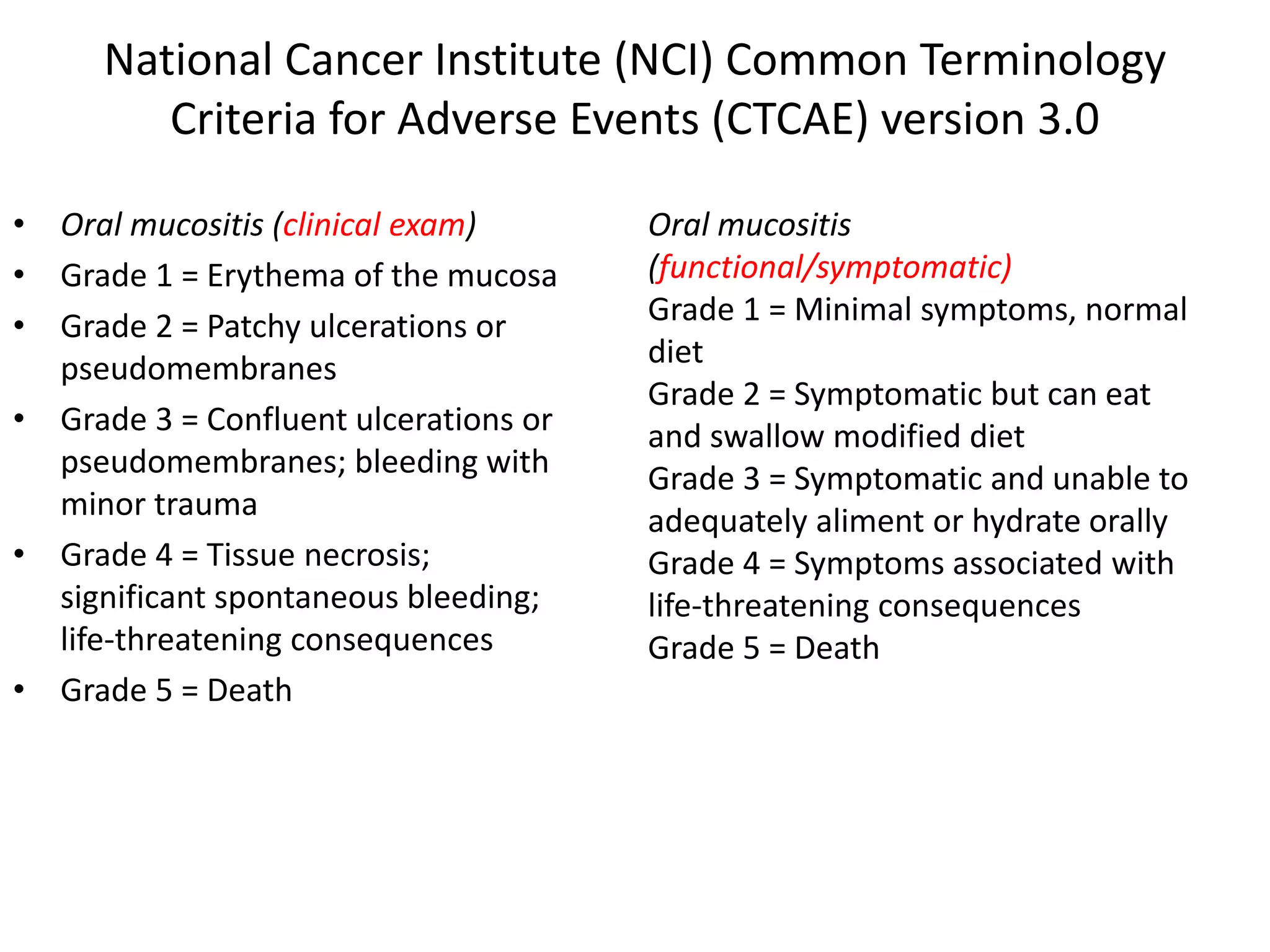
- Oral mucositis is a common side effect of cancer therapy like chemotherapy and radiation therapy. It involves inflammation and ulceration of the mucous membranes in the mouth.
- Chemotherapy-induced mucositis is caused by drugs damaging the basal epithelial cells. This triggers an inflammatory response and ulceration. Risk is higher with certain chemo agents and combinations with radiation.
- Radiation-induced mucositis occurs as radiation damages the mucosal stem cells, inhibiting cell regeneration and causing the epithelium to break down over the treatment period. The severity depends on factors like daily dose and total cumulative dose.