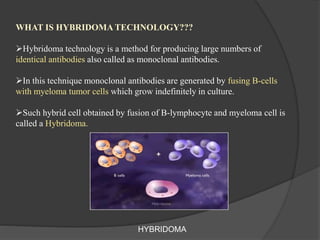
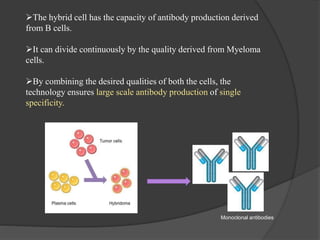
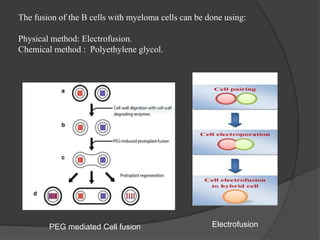
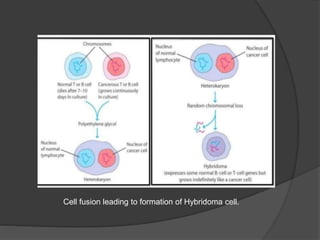
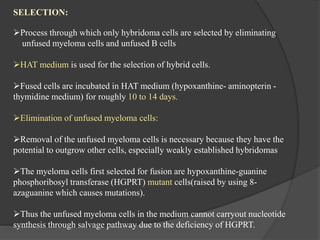
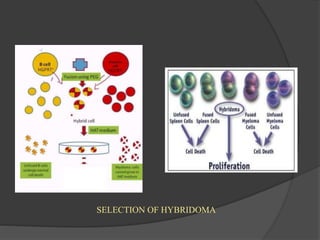
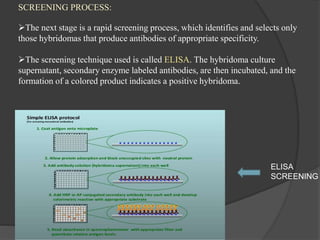
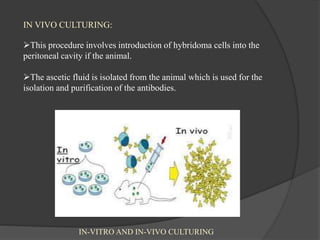
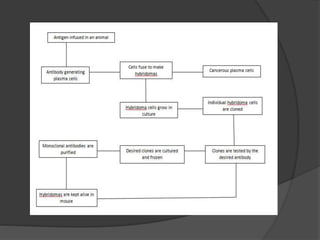
Hybridoma technology is a method for generating monoclonal antibodies by fusing B cells with myeloma cells. Georges Köhler and César Milstein developed this technique in 1975 and were awarded the Nobel Prize for it. The hybridoma cells produced from this fusion have the antibody production ability of B cells and indefinite growth ability of myeloma cells, allowing large-scale monoclonal antibody production. Hybridomas are selected using HAT medium, which eliminates unfused B and myeloma cells. The resulting hybridomas are then screened and cultured to produce monoclonal antibodies, which have wide applications in areas like cancer diagnosis, treatment, and research.