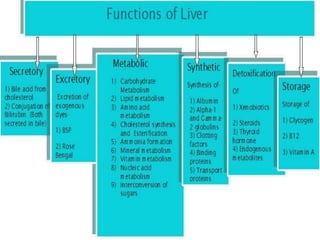
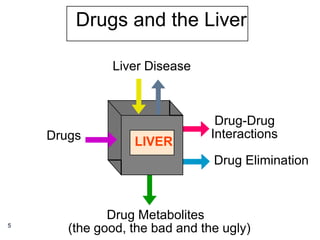
1) The liver plays a key role in metabolism and detoxification, and is susceptible to damage from toxins like alcohol, chemicals, and certain drugs.
2) Drugs are a common cause of liver injury (DILI), with anti-tuberculosis drugs, anti-convulsants, NSAIDs, anti-microbials, and anesthetics carrying risks. DILI can range from asymptomatic enzyme elevations to acute liver failure.
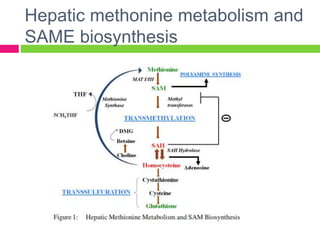
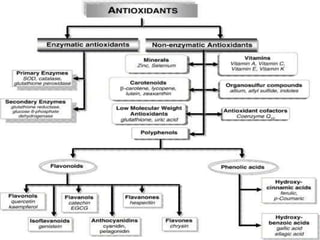
3) Many agents have hepatoprotective properties, including N-acetylcysteine, penicillamine, antioxidants, S-adenosylmethionine (SAMe), and herbal medicines like Silybum marianum (