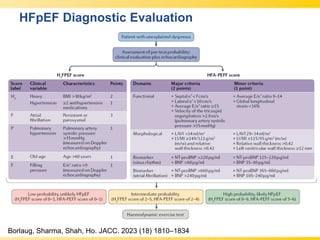
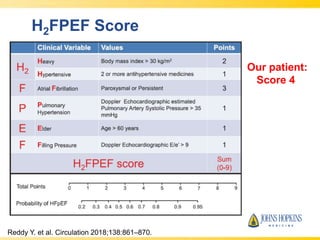
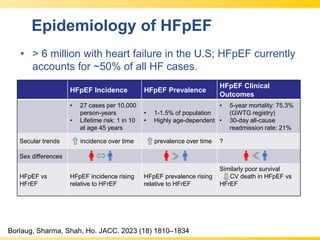
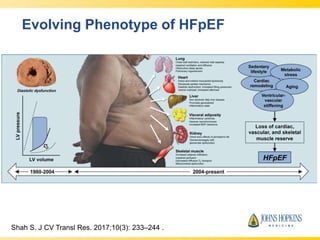
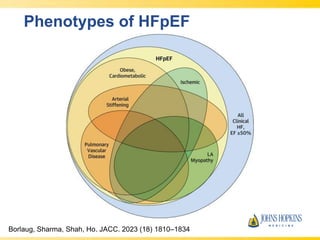
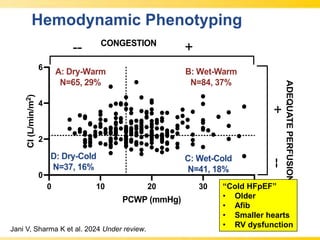
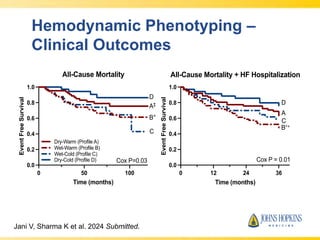
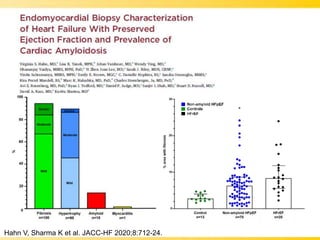
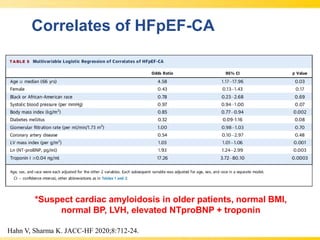
The document discusses heart failure with preserved ejection fraction (HFpEF), focusing on diagnostic updates and algorithms for evaluation. It presents a case study of a patient demonstrating typical symptoms and test results relevant to HFpEF, alongside current guidelines for diagnosis and treatment. The increasing incidence and prevalence of HFpEF, challenges in diagnosis, and the significance of ongoing research for targeted therapies are highlighted.