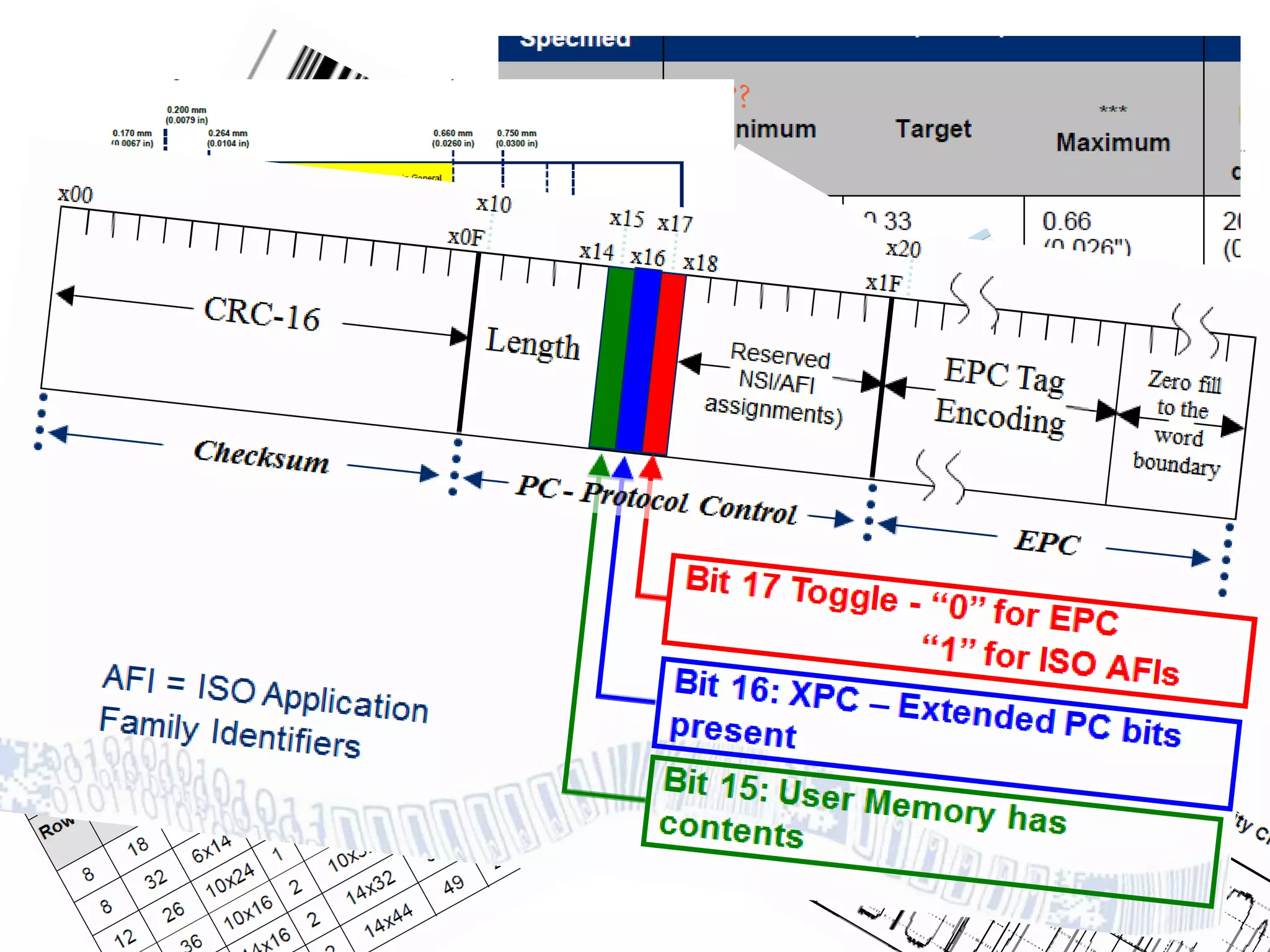
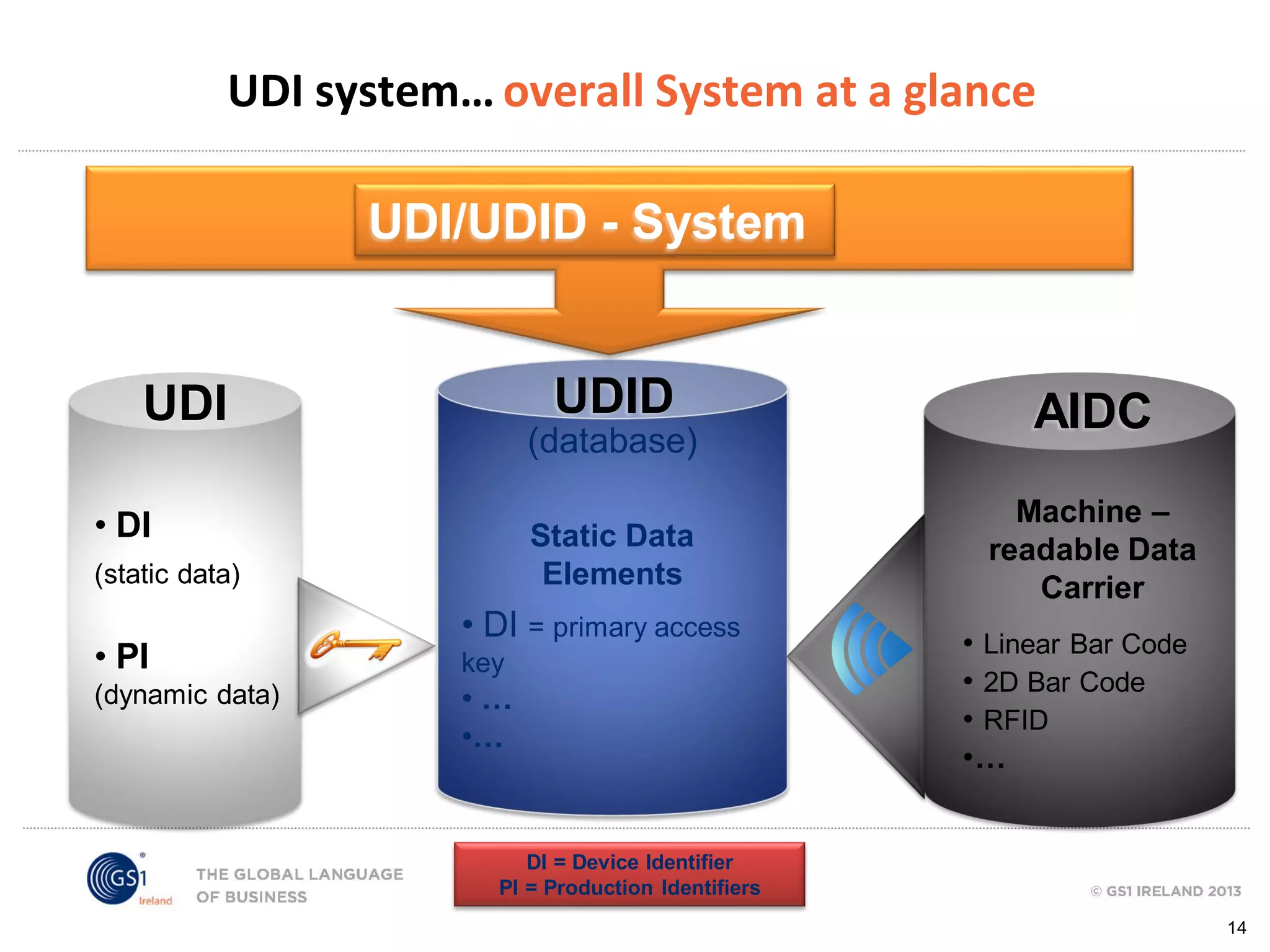
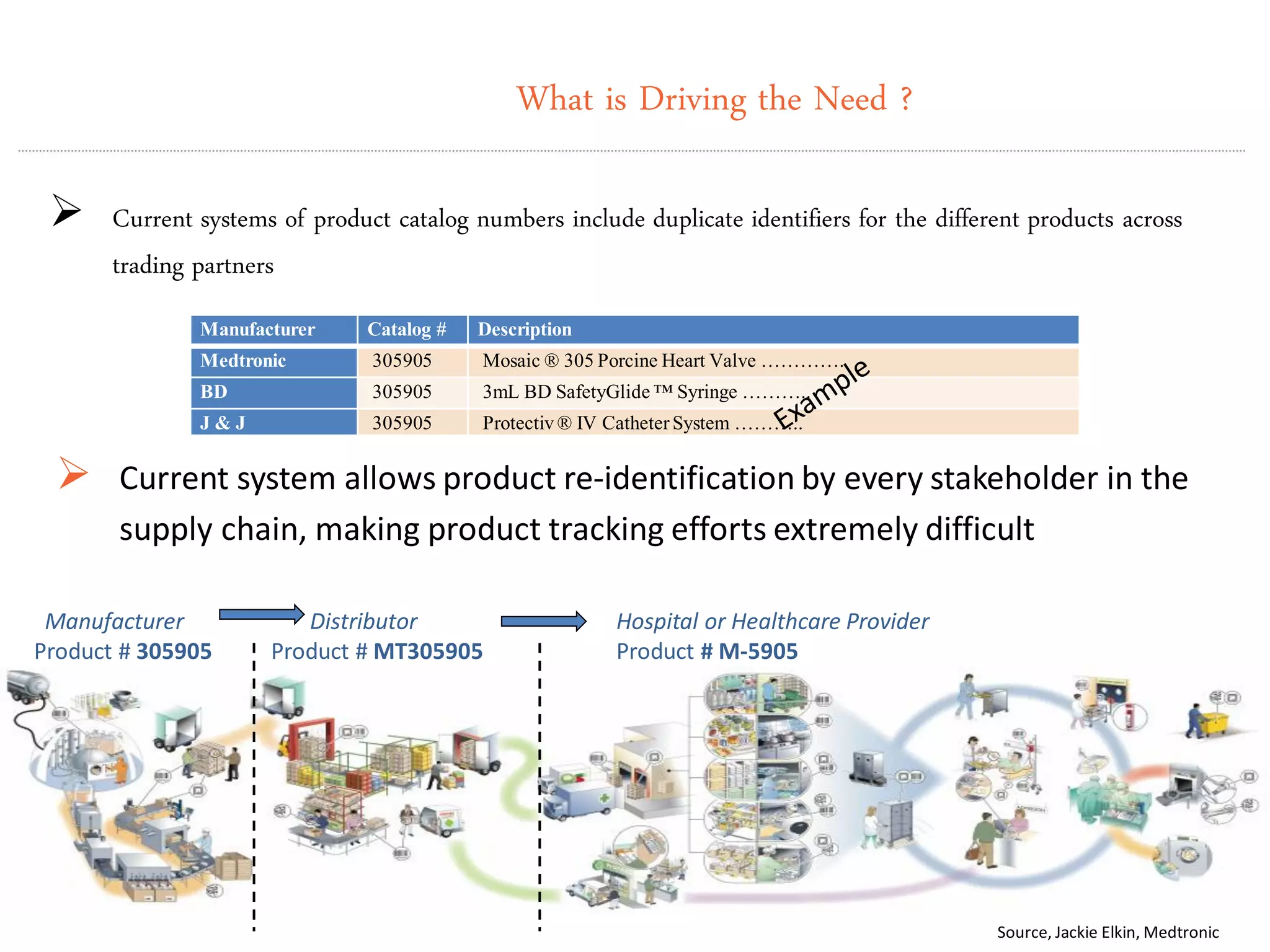
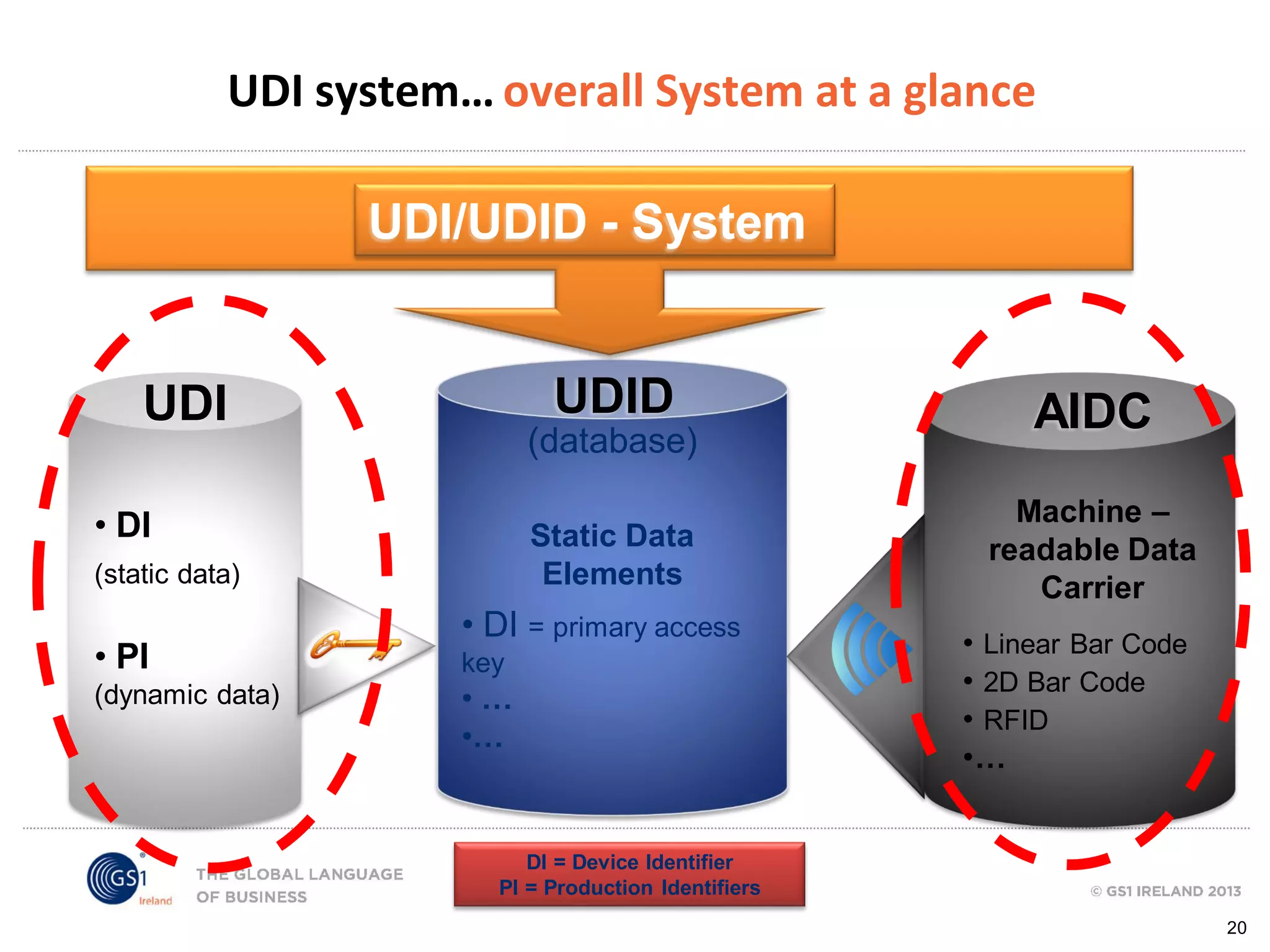
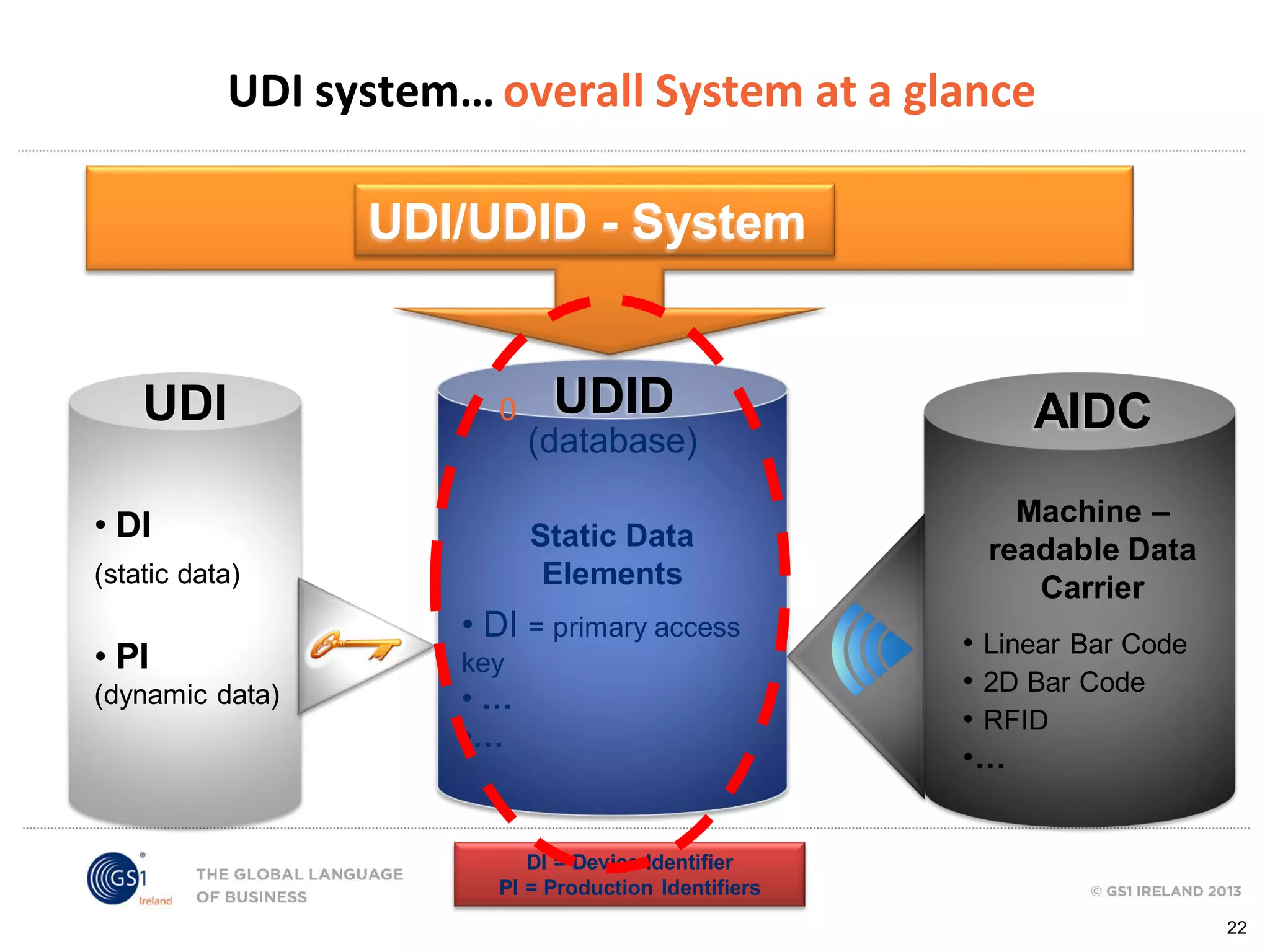
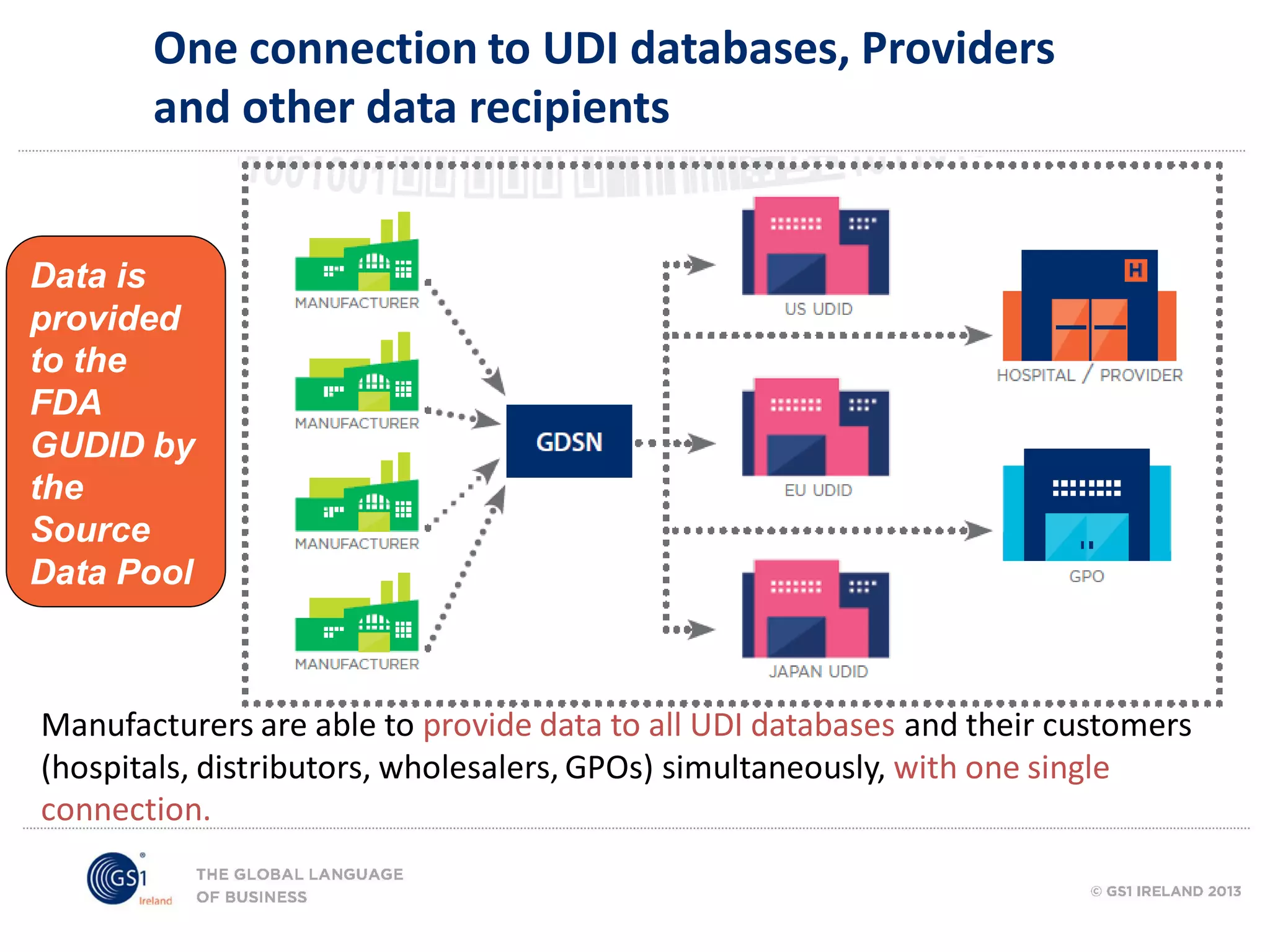
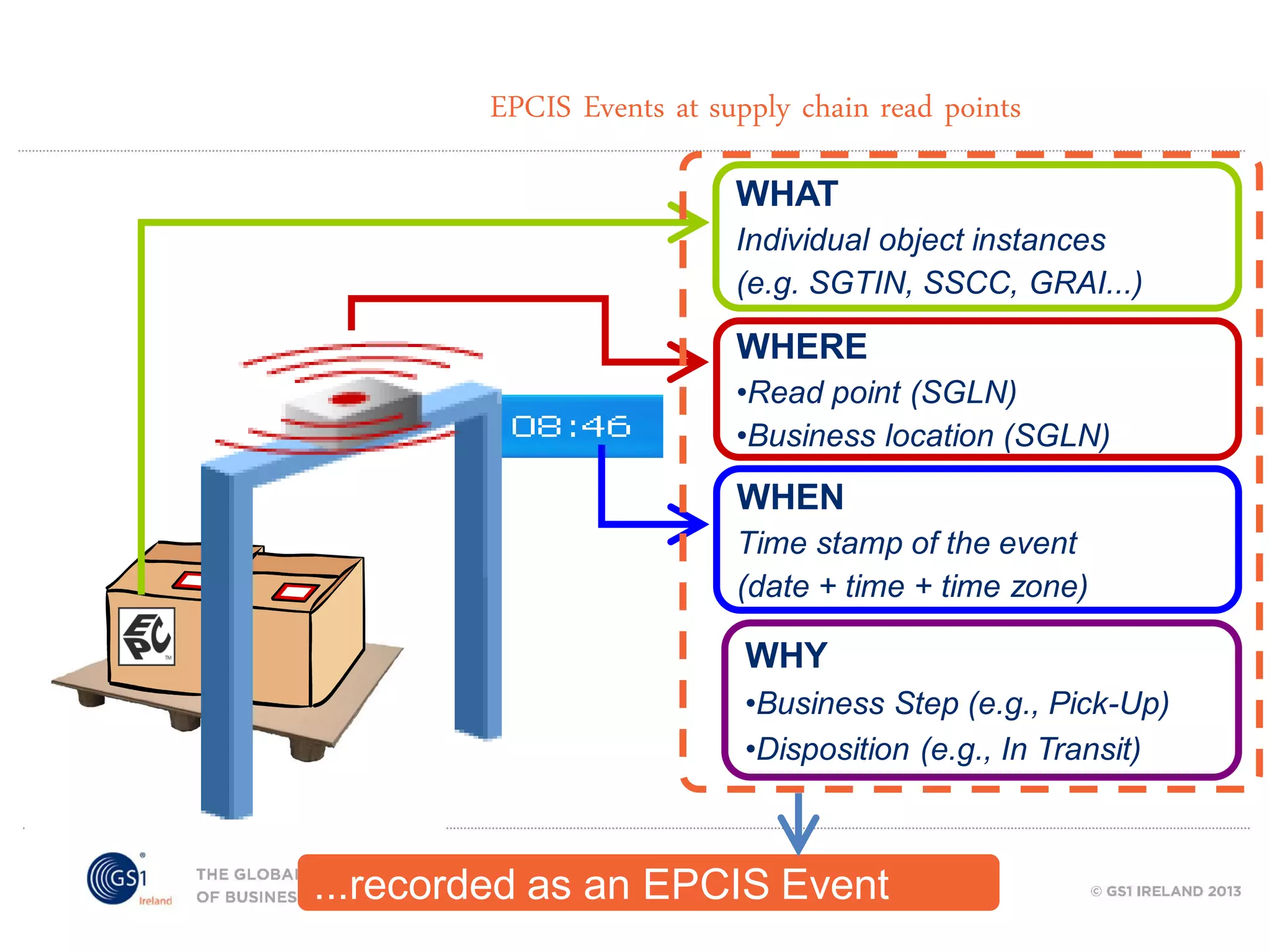
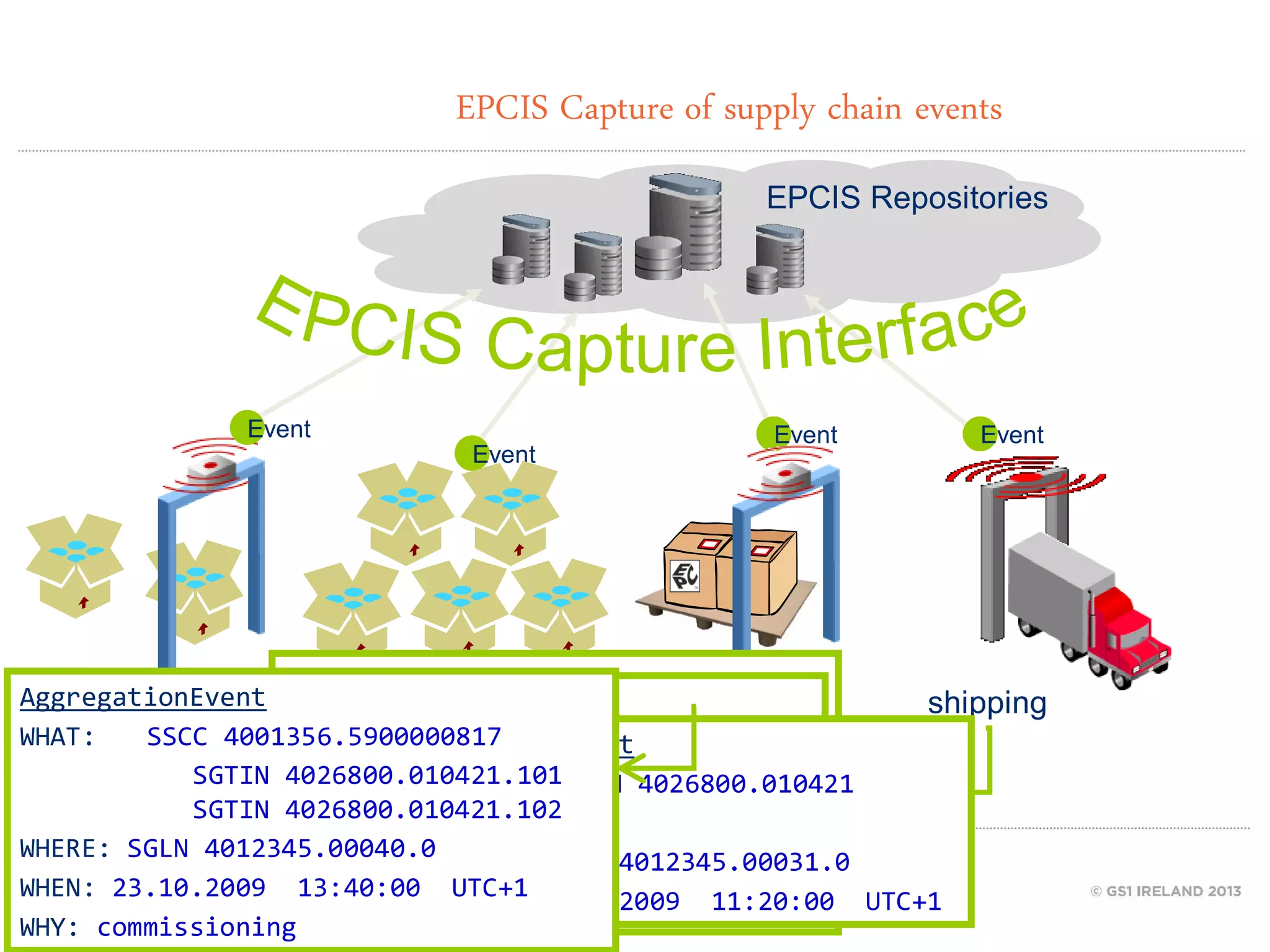
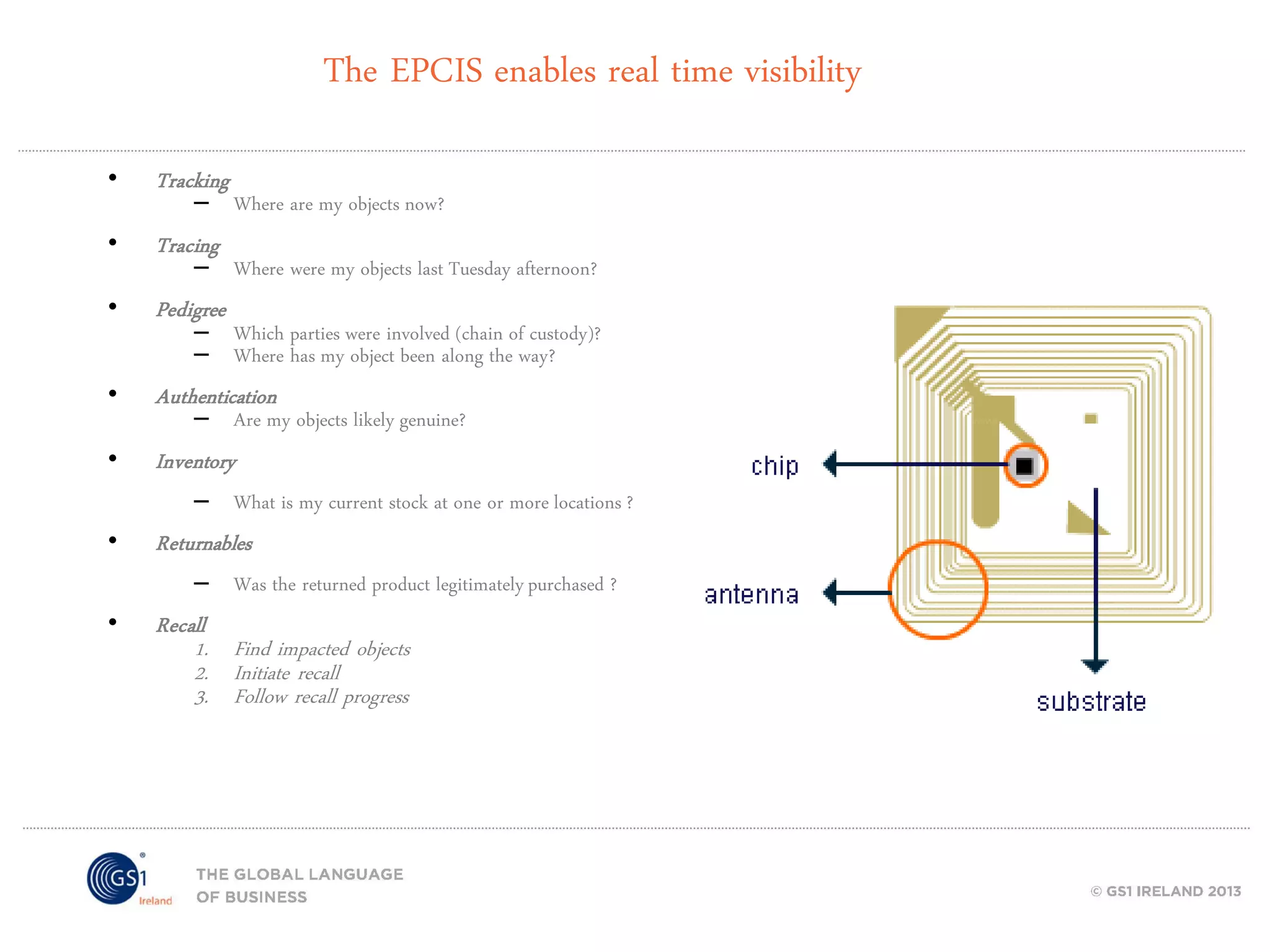
GS1 standards provide a framework for identifying, capturing, and sharing supply chain information through barcodes and electronic data interchange. The presentation discusses how GS1 standards can be applied to ensure traceability, enable unique device identification (UDI) as required by the FDA, and provide real-time visibility through standards like EPCIS. It also outlines specific GS1 solutions for sectors like transportation and logistics, healthcare, and customs that leverage global standards to improve supply chain collaboration and efficiency.