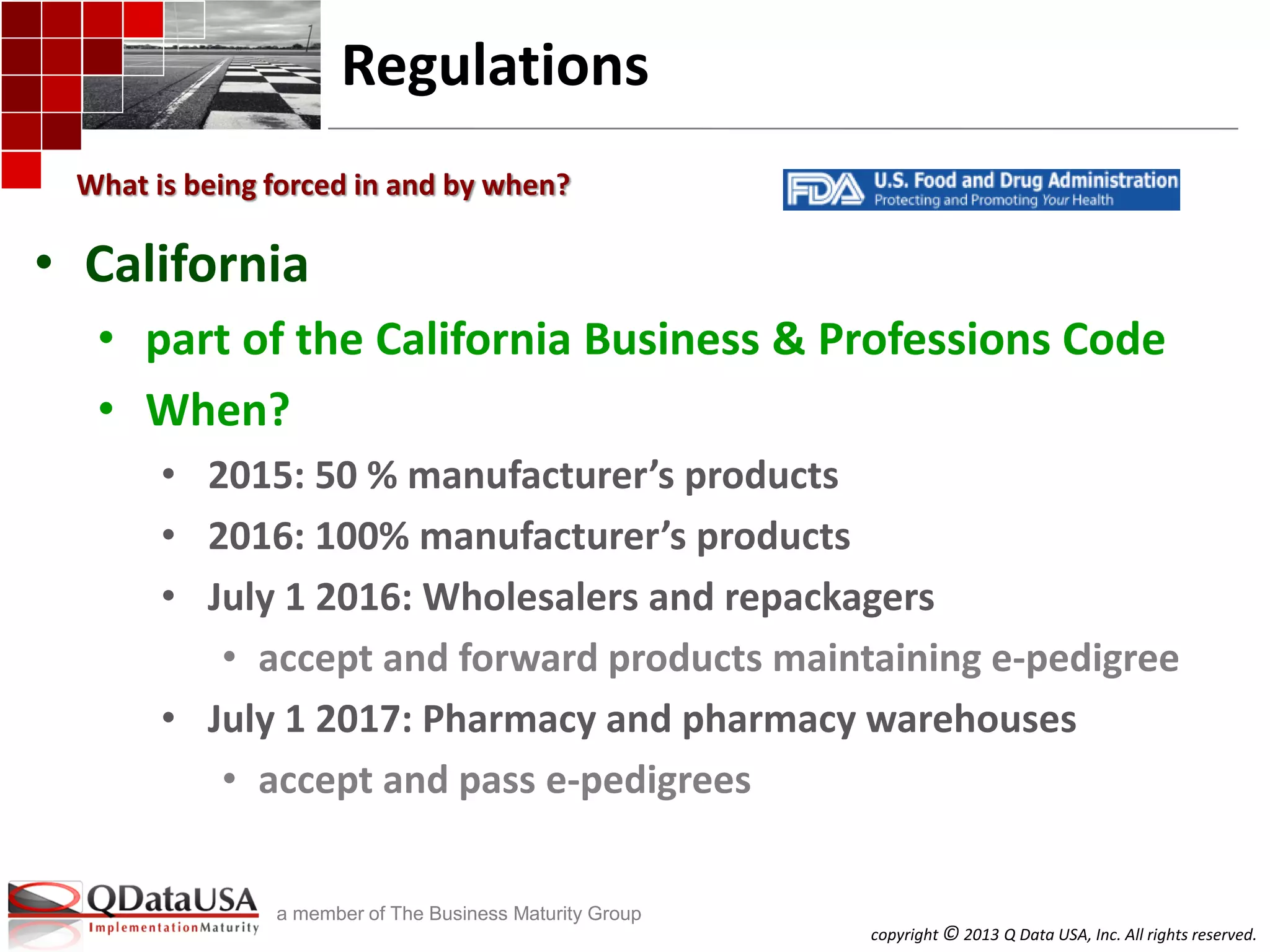
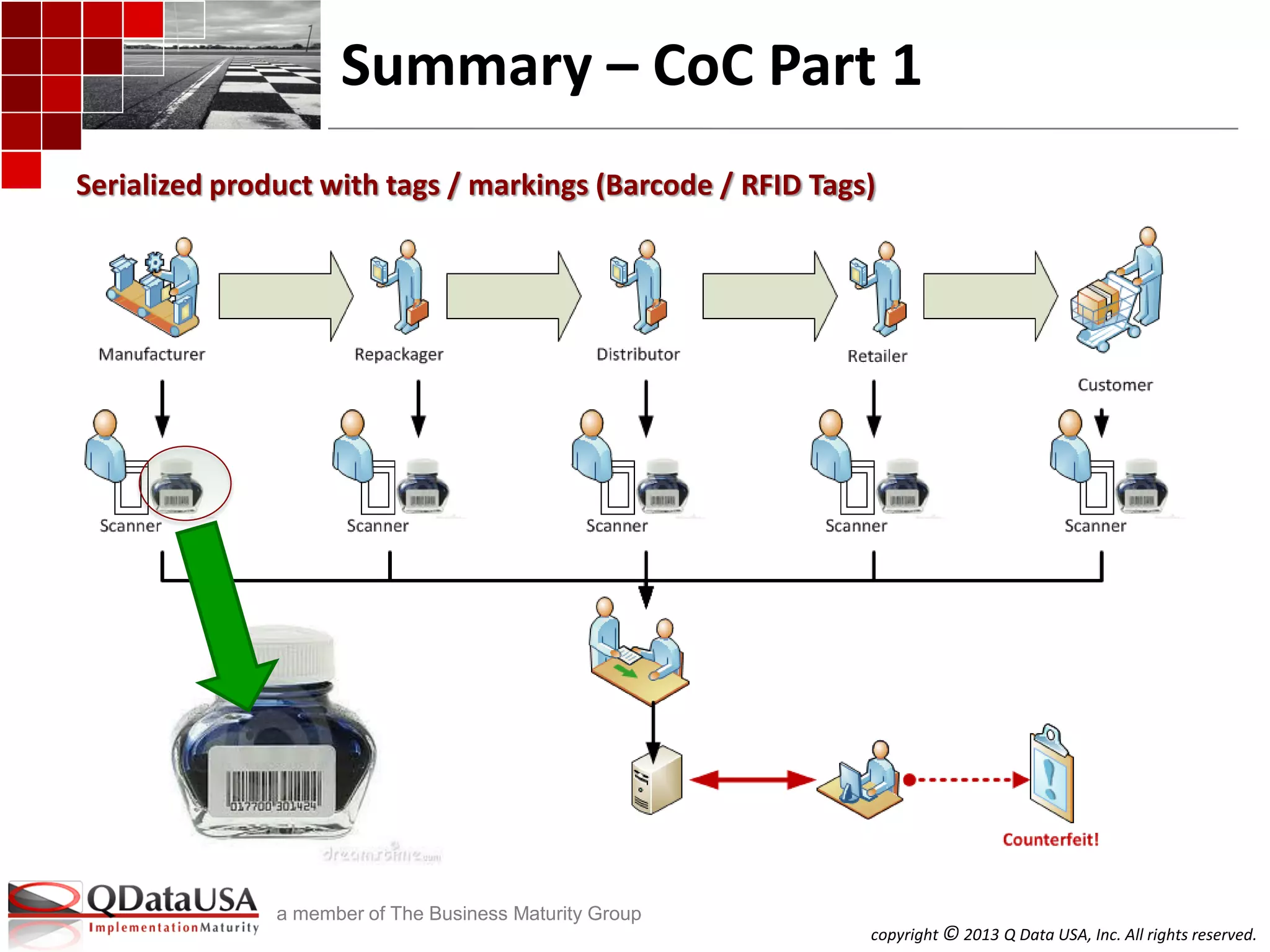
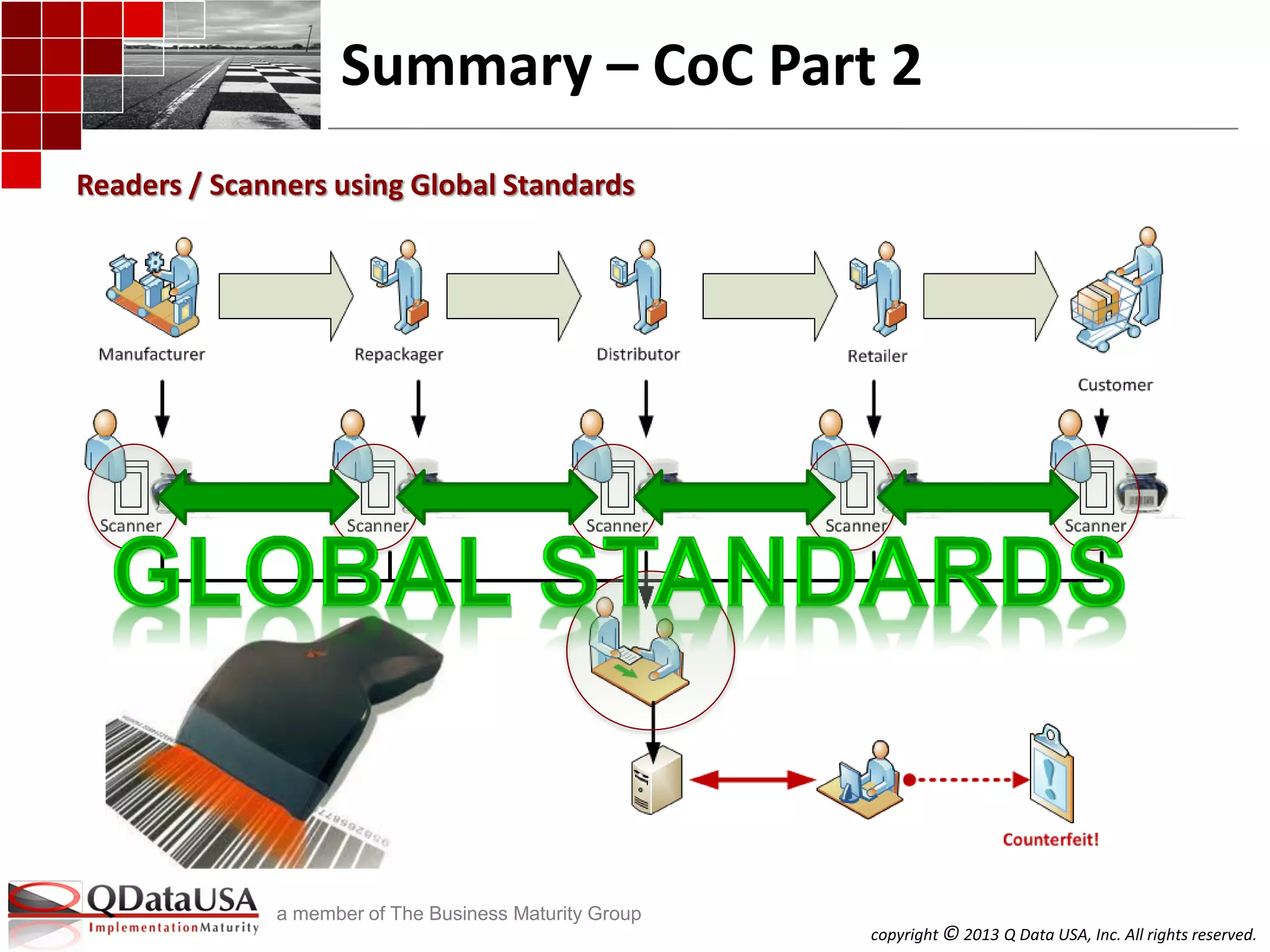
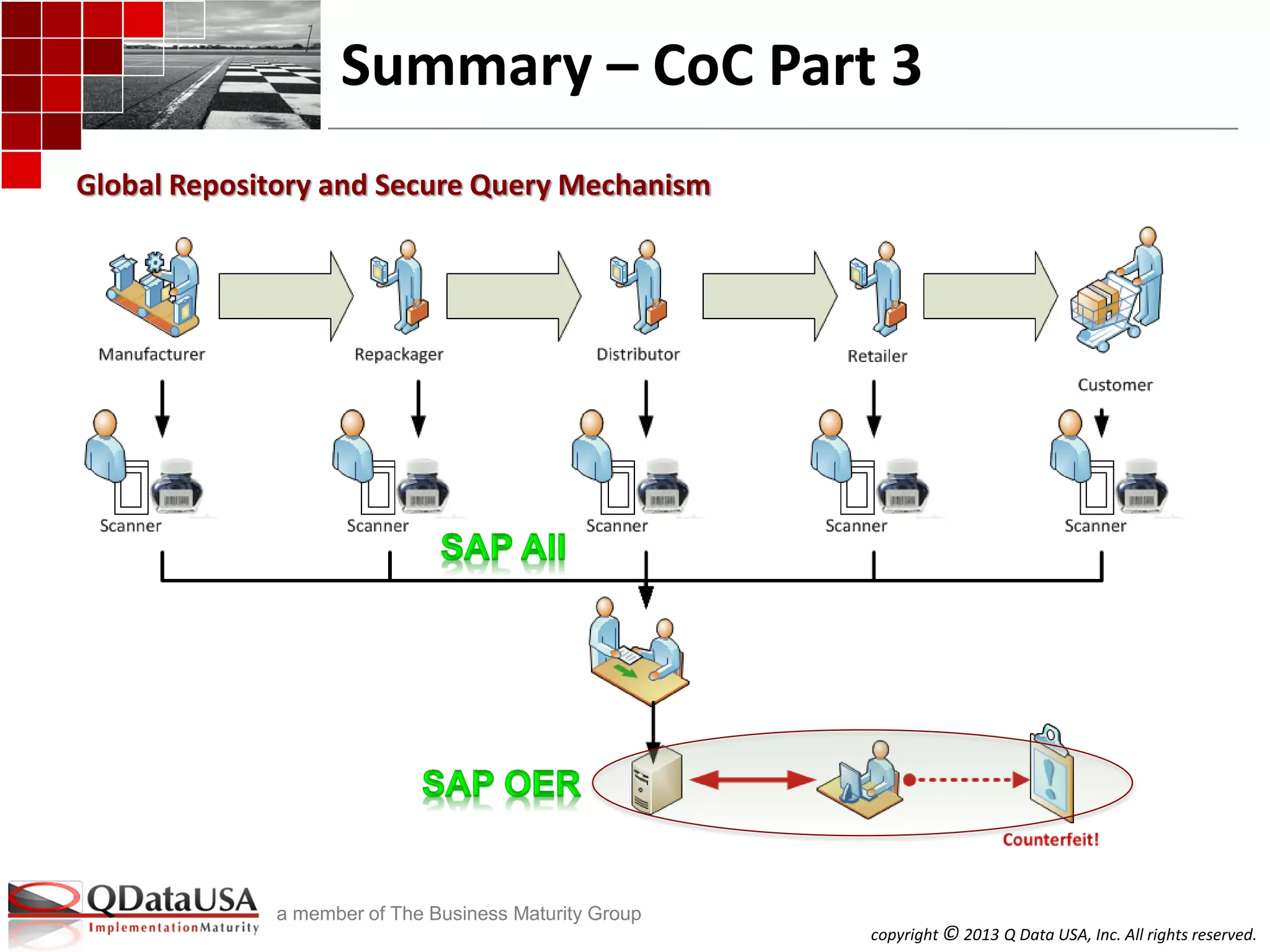
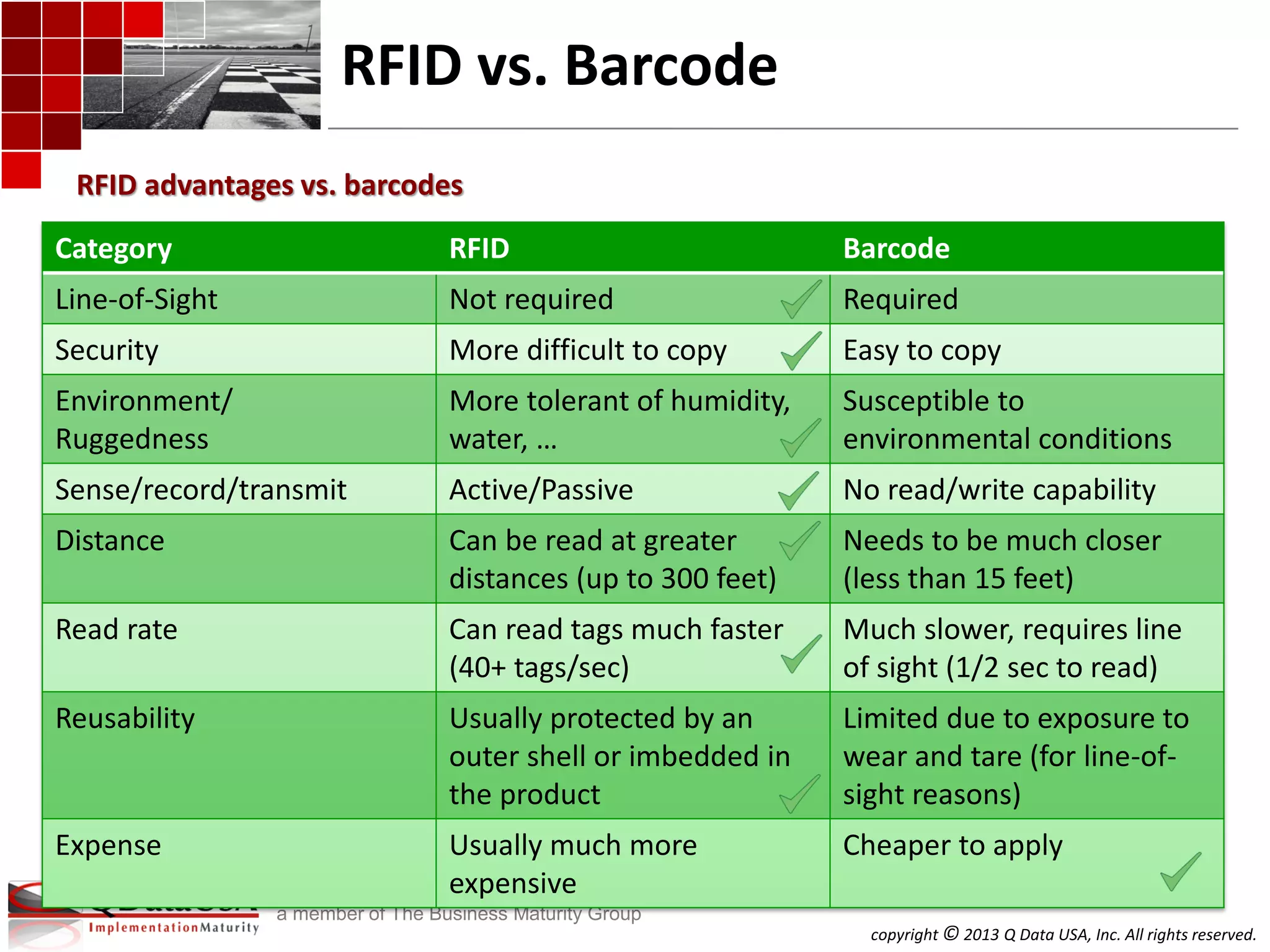
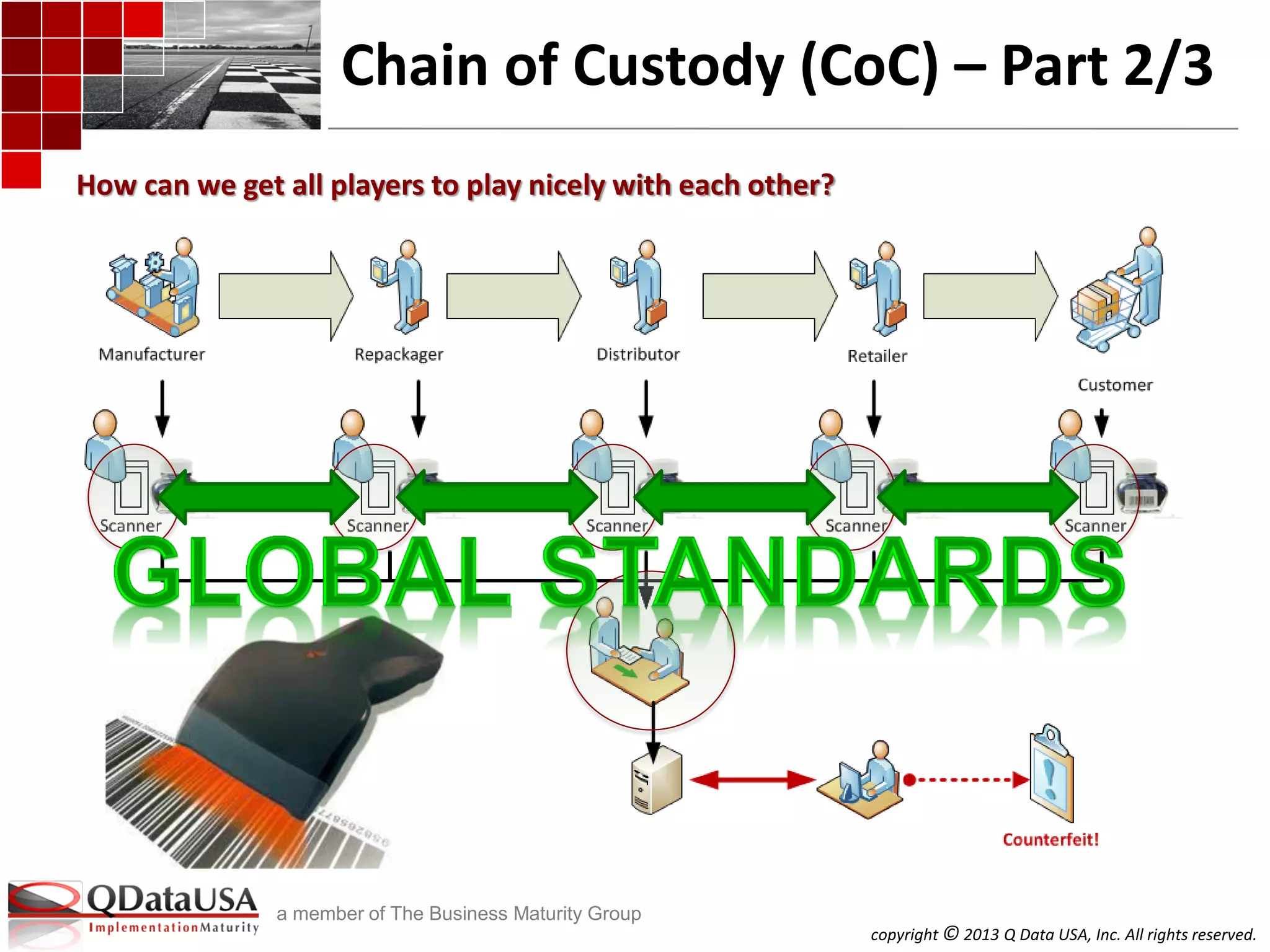
The document discusses serialized track and trace and solutions to business problems. It provides an agenda for a webinar on serialized track and trace, including vocabulary, standards, components, and regulations. It then covers various topics in depth, such as chain of custody, unique identification standards like GTIN and RFID, and SAP solutions for implementing auto-ID and item serialization.
























![copyright © 2013 Q Data USA, Inc. All rights reserved.
a member of The Business Maturity Group
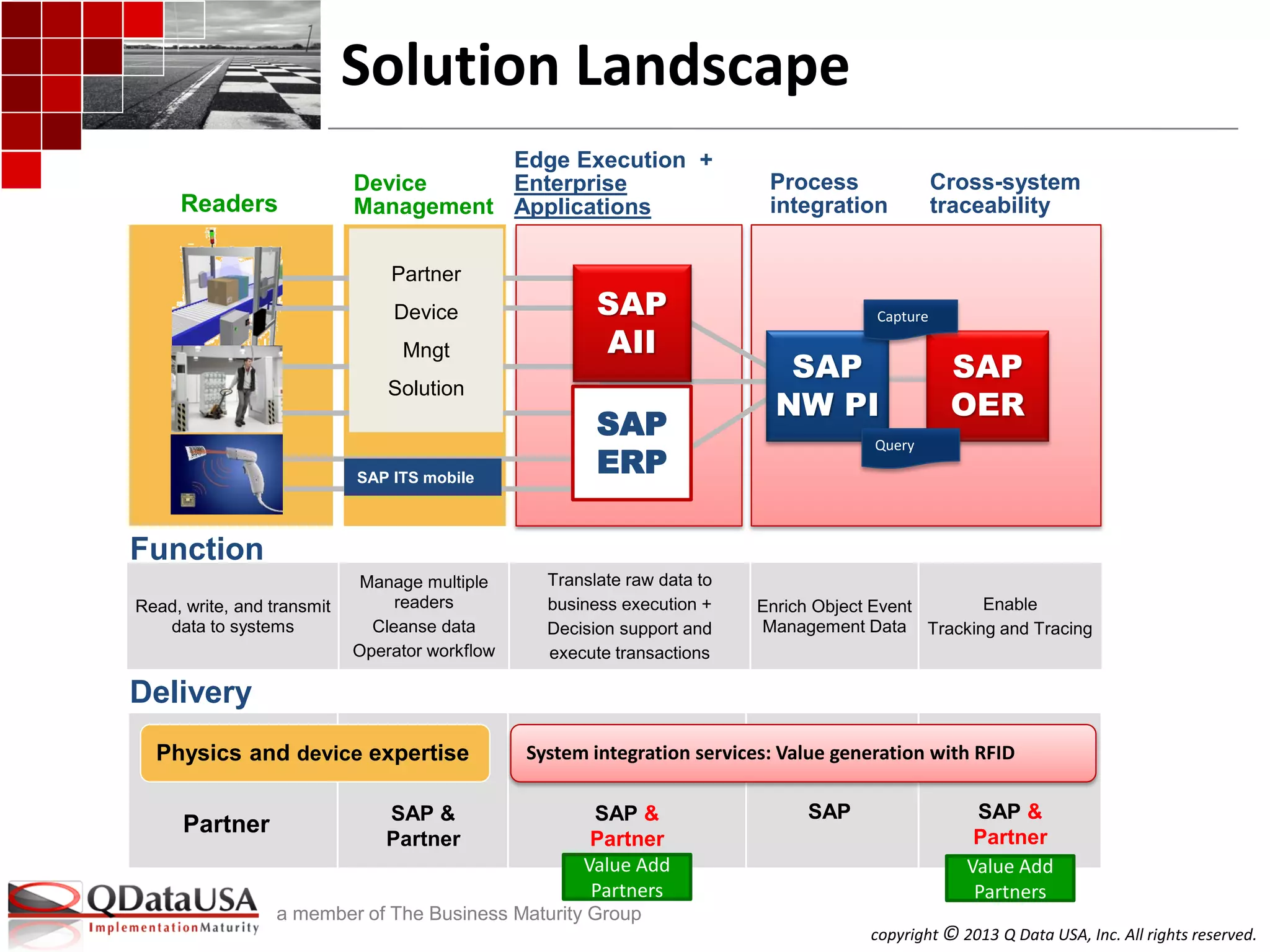
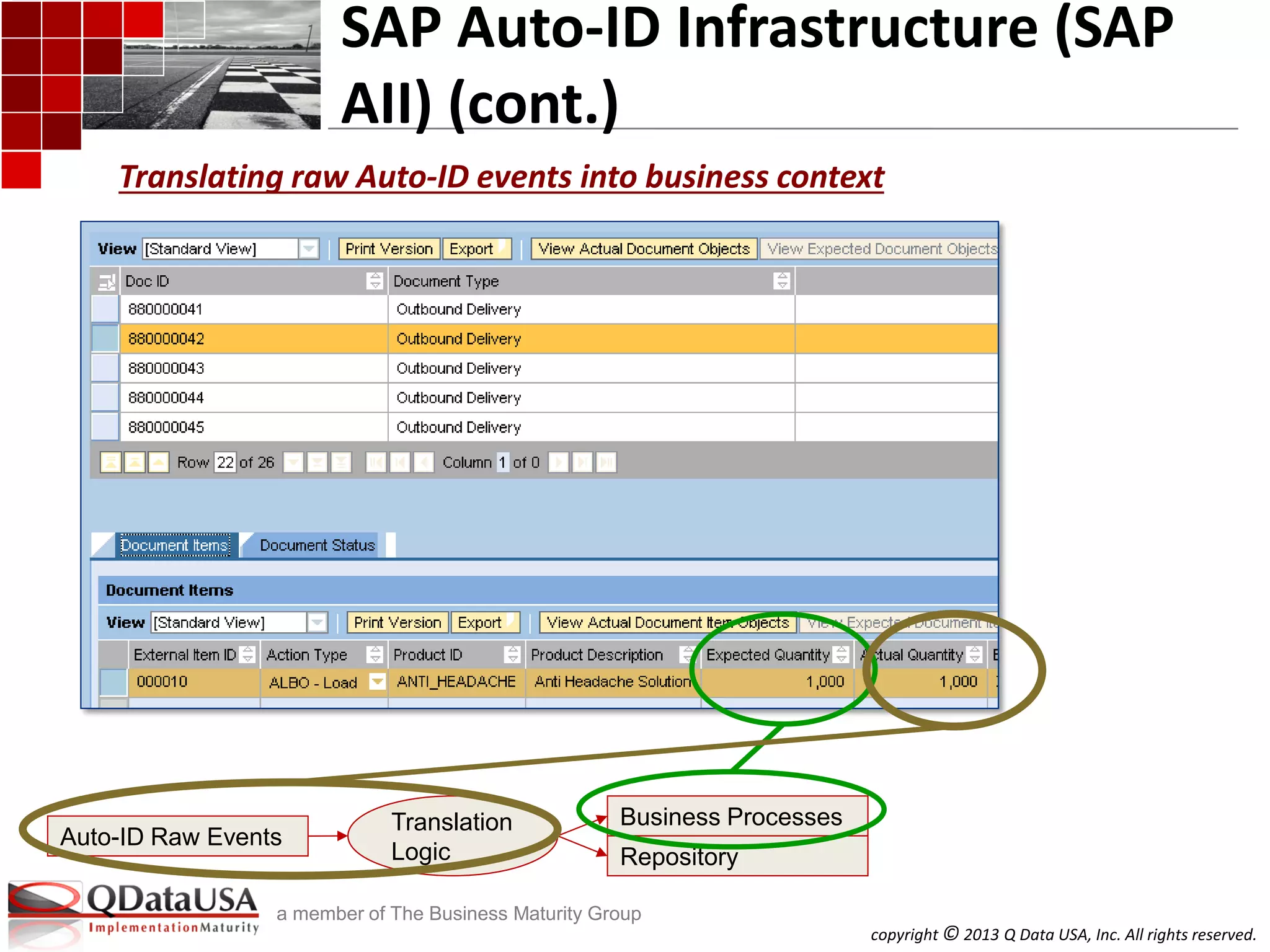
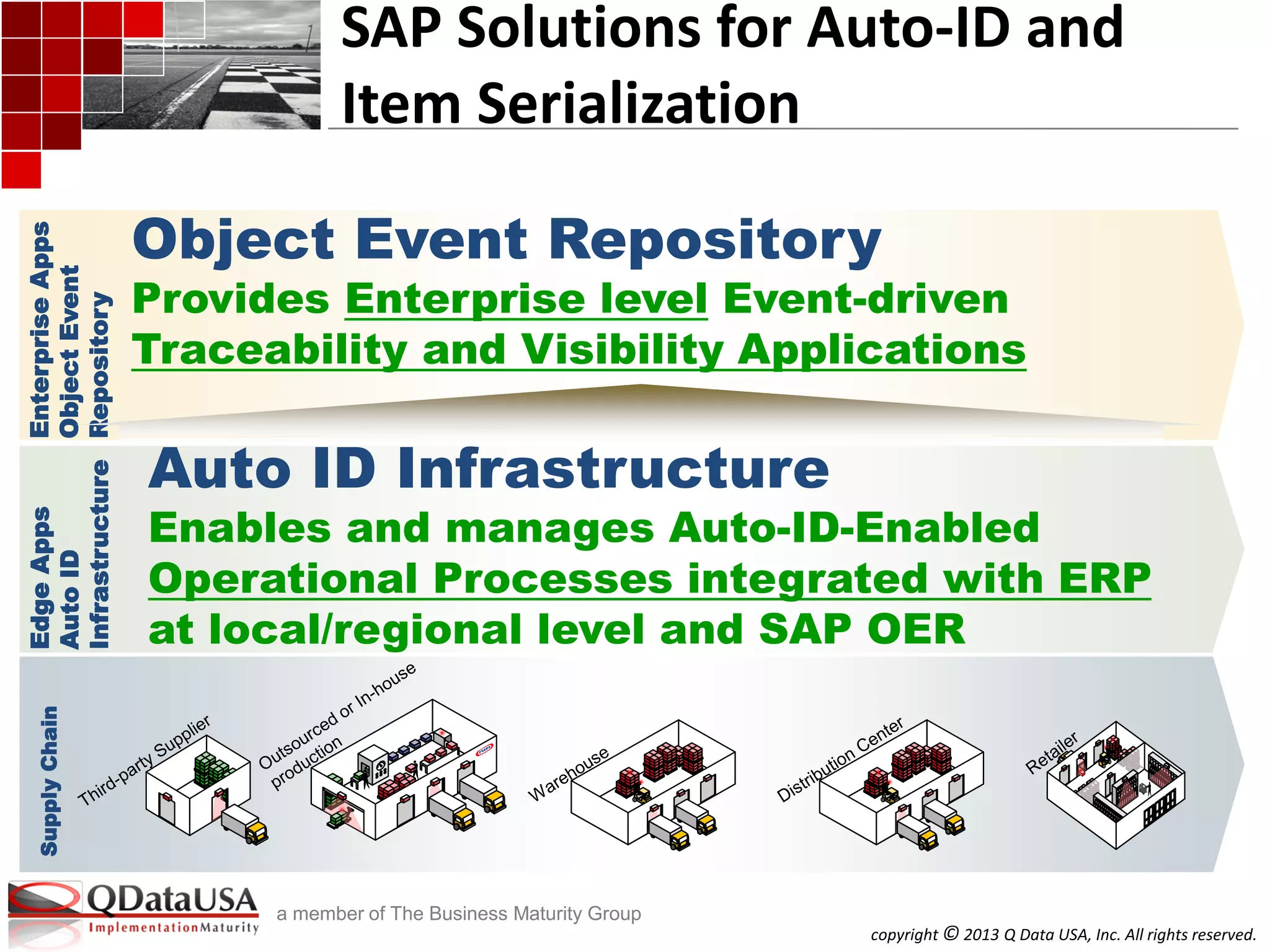
SAP Solutions for Auto-ID and Item
Serialization (cont.)
Auto-ID-Enabled Business Process
Automation
Item serialized Product, Part, Asset, and
Hierarchy Life Cycle Traceability
Auto-ID-Enabled Outbound Processing
Auto-ID Enabled Inbound Processing
RFID-Kanban
Returnable Transport Items
Extensible to other ERP document
integration
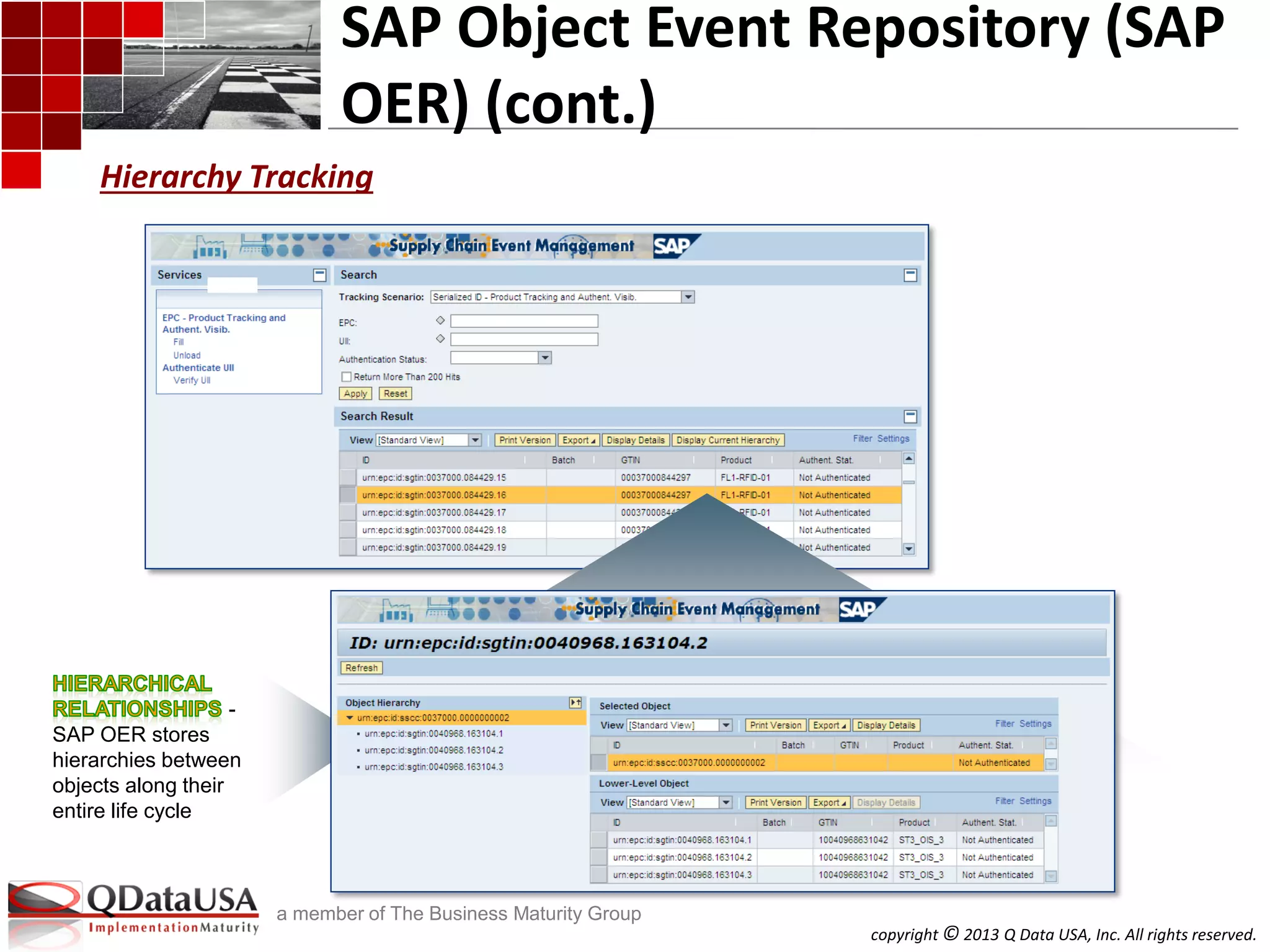
SAP AII SAP OER/EM
Product Tracking and Authentication
Returnable Transport Items
Linear barcode
2-D barcode
Passive/Active RFID
Sensor events: Conditions, temperature,
locations
GS1/ EPCglobal EPCIS – certified conformance to
EPCIS Standard as an EPCIS Repository
[Beyond this OER can be easily extended to
custom attributes, vocabularies, etc.]
GS1/EPCglobal Tag Data (TD) Standard
GS1/EPCglobal TD Translation Std.
US DOD Guide to Uniquely Identifying Items
Beyond this AII can be easily configured to
other unique ID schemes
Domain
Core Solution
Standard
Processes
and Beyond
AIDC Support
Standards
Support
Hardware
Integration
in to App Store the Data](https://image.slidesharecdn.com/a3serializedtrackandtrace-isitarequirementyouneedtoconformtoorneed-130612002857-phpapp02/75/Serialized-track-and-trace-Is-it-a-requirement-you-need-to-conform-to-or-need-32-2048.jpg)