Embed presentation
Download as PDF, PPTX

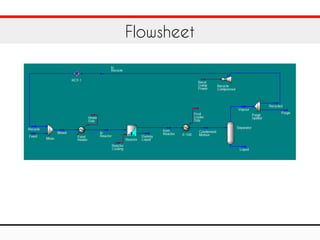


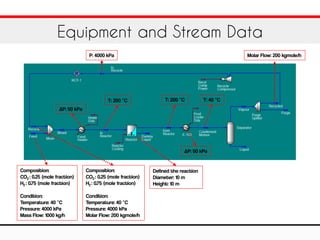

The document outlines a training exercise related to the methanol process using Aspen HYSYS, detailing components like CO2, hydrogen, methanol, and water. It provides specific parameters for the reaction, including kinetic parameters and operational conditions such as temperature and pressure. Additionally, it gives technical specifications for the reaction equipment, including dimensions and flow data.





