Electrons & Photons: Understanding the Photoelectric Effect
- 3. Photoelectric effect (Hertz & Lenard’s Observations) • Discovered in 1887 by Heinrich Hertz. • The phenomenon of emission of electrons by certain substance (metal), when it is exposed to radiations of suitable frequencies is called as photoelectric effect and emitted electrons are called photoelectrons.
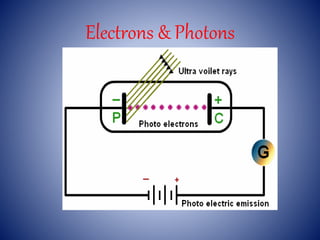
- 4. Experimental study of photoelectric effect
- 5. Effect of frequency on photoelectric current 0 0.5 1 1.5 2 2.5 3 3.5 4 4.5 5 0 0.5 1 1.5 2 2.5 3 PHOTOELECTRICCURRENT FREQUENCY
- 6. The minimum frequency of incident radiation for which photoelectrons are just emitted from photosensitive material is called threshold frequency. It is denoted by V0 Wavelength corresponding to threshold frequency V0 is known as threshold wavelength.
- 7. Effect of intensity on photoelectric current The photoelectric current is directly proportional to the number of photoelectrons emitted per second. Hence , Photoelectrons emitted per second α Intensity
- 8. Effect of potential difference on photoelectric current
- 9. Characteristics of photoelectric effect • Below cutoff frequency of incident radiation also known as threshold frequency V0 no emission of photoelectrons takes place. V0 is different for different metals. • Photoelectric current is directly proportional to the intensity of incident light . • Above the threshold frequency V0 the maximum K.E of the emitted photoelectrons increases linearly with the frequency of the incident radiation, but is independent of intensity of incident light. • The emission of photoelectrons is an instantaneous process. • No time lag between the irradiation of the metal surface and emission of photoelectrons.
- 10. Einstein’s photoelectric equation: • Einstein extended Planck’s idea in 1905 by deriving an equation . He made two assumptions: A radiation of frequency v consist of discrete quanta or photons. E = hv Where h is Planck’s constant. Photons move with speed of light. When radiation of frequency v is incident on photosensitive surface there are collisions between photons & electrons. Entire energy of photon is transferred to electron without time lag.
- 11. • The absorbed energy hv by electron is used in two ways
- 12. • ev = hv-Φ • We can now explain all characteristics of photoelectric effect with the help of Einsteins photoelectric equation • If frequency of incident radiation is decreased ,the kinetic energy of photoelectrons also decreases and finally becomes zero for particular frequency say vo • v= vo then K.E = ½ mv2 =0 • 0 = • ½ mv2 =h (v- vo) • If v > vo ,photoelectrons are emitted with some velocity • If v<vo ,no photoelectrons are emitted. • If v= vo , photoelectrons are emitted with K.E= 0 hv-Φ
- 14. APPLICATIONS OF PHOTOELECTRIC CELL • EXPOSURE METER • BURGLAR ALARM • SOUND REPRODUCTION FROM MOTION PICTURES
- 15. THANK YOU