Quantum and nuclear physics clil photoelectric
•Download as PPT, PDF•
1 like•974 views
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Diploma sem 2 applied science physics-unit 5-chap-2 photoelectric effect

Diploma sem 2 applied science physics-unit 5-chap-2 photoelectric effect
Dual nature of Radiation and Matter - MR. RAJA DURAI APS

Dual nature of Radiation and Matter - MR. RAJA DURAI APS
Similar to Quantum and nuclear physics clil photoelectric
Similar to Quantum and nuclear physics clil photoelectric (20)
Modern_Physics,_Photoelectric_Effect_&_X_rays_BOUNCE_BACK_24_DEC (1).pdf

Modern_Physics,_Photoelectric_Effect_&_X_rays_BOUNCE_BACK_24_DEC (1).pdf
09 UNIT-9(Electronics and down of Modern Physics) (1).pptx

09 UNIT-9(Electronics and down of Modern Physics) (1).pptx
202004091956360894nkpandey_Photoelectric_Effet.pdf

202004091956360894nkpandey_Photoelectric_Effet.pdf
Shubham Kashyap _20240226_222839_0000.pptx_20240226_223507_0000.pptx

Shubham Kashyap _20240226_222839_0000.pptx_20240226_223507_0000.pptx
Subhadeep_BSPH101.pptx dsuyfgwf7ywidvweiufwetdfiuqefuhygedfiewgfi

Subhadeep_BSPH101.pptx dsuyfgwf7ywidvweiufwetdfiuqefuhygedfiewgfi
Class 12th Physics Photoelectric effect dual nature of matter radiations

Class 12th Physics Photoelectric effect dual nature of matter radiations
.trashed-1675533462-photoelectric_effect_dual_nature_of_matter_radiations.ppt

.trashed-1675533462-photoelectric_effect_dual_nature_of_matter_radiations.ppt
Recently uploaded
Recently uploaded (20)
Dev Dives: Streamline document processing with UiPath Studio Web

Dev Dives: Streamline document processing with UiPath Studio Web
Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...

Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...
Powerpoint exploring the locations used in television show Time Clash

Powerpoint exploring the locations used in television show Time Clash
Scanning the Internet for External Cloud Exposures via SSL Certs

Scanning the Internet for External Cloud Exposures via SSL Certs
Gen AI in Business - Global Trends Report 2024.pdf

Gen AI in Business - Global Trends Report 2024.pdf
Human Factors of XR: Using Human Factors to Design XR Systems

Human Factors of XR: Using Human Factors to Design XR Systems
"Federated learning: out of reach no matter how close",Oleksandr Lapshyn

"Federated learning: out of reach no matter how close",Oleksandr Lapshyn
Developer Data Modeling Mistakes: From Postgres to NoSQL

Developer Data Modeling Mistakes: From Postgres to NoSQL
My INSURER PTE LTD - Insurtech Innovation Award 2024

My INSURER PTE LTD - Insurtech Innovation Award 2024
SQL Database Design For Developers at php[tek] 2024![SQL Database Design For Developers at php[tek] 2024](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![SQL Database Design For Developers at php[tek] 2024](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
SQL Database Design For Developers at php[tek] 2024
Unraveling Multimodality with Large Language Models.pdf

Unraveling Multimodality with Large Language Models.pdf
Unleash Your Potential - Namagunga Girls Coding Club

Unleash Your Potential - Namagunga Girls Coding Club
Quantum and nuclear physics clil photoelectric
- 2. Unespected results • The emission of electrons depends on frequency • energy of electrons generated by dim illumination is the same as that generated by bright illumination so, NO DEPENDANCE ON LIGHT INTENSITY (electrons have the same speed) • It is a contradiction if compared to basic wave theory • (classically energy is proportional to wave amplitude squared)
- 3. Proof • Stopping potential applied to the anode: (reverse potential to prevent electrophotons to reach the anode) • KE = ½ mv2 = eVs • It was shown that Vs is directly proportional to the maximum kinetic energy of the photoelectrons and does not depend on Intensity (same V when intensity changes)
- 4. Previous results used by Einstein • Planck had showed that radiation is emitted in pulses of energy hf • E = hf
- 5. Einstein explanations - Some energy is needed to release the electron (the work function φ)… …and some energy is given to the electron as kinetic energy. Photon Energy = work function + kinetic energy of electron
- 6. Photoelectric Effect: Vstop vs. Frequency stopeV hf φ= − min0stopV hf φ= ⇒ = Slope = h = Planck’s constant hfmin −φ
- 7. Photoelectric Effect: Vstop vs. Frequency stopeV hf φ= − min0stopV hf φ= ⇒ = Slope = h = Planck’s constant hfmin −φ
Editor's Notes
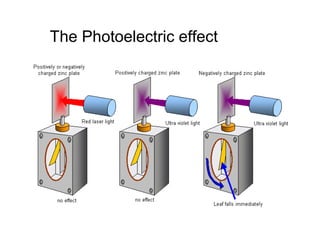
- Hertz 1887 illuminated a zinc strip with an ultraviolet spark (can be the sun) -> conduction Only when negatively charged 1897 -> electrons Effect due to frequency not to intensity!