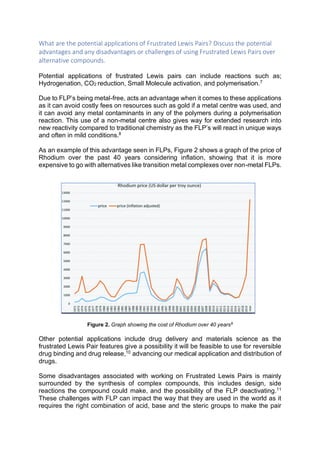
This document discusses frustrated Lewis pairs (FLPs), which are compounds containing a Lewis acid and base that cannot form the expected adduct due to steric hindrance. FLPs have potential applications in hydrogenation, CO2 reduction, and polymerization due to their non-metal nature. They avoid costly metal catalysts and enable new reactivity. However, FLP synthesis presents challenges like complex design, characterization, and reactivity with air/water. Despite difficulties, FLPs show promise over metal complexes for catalysis applications.