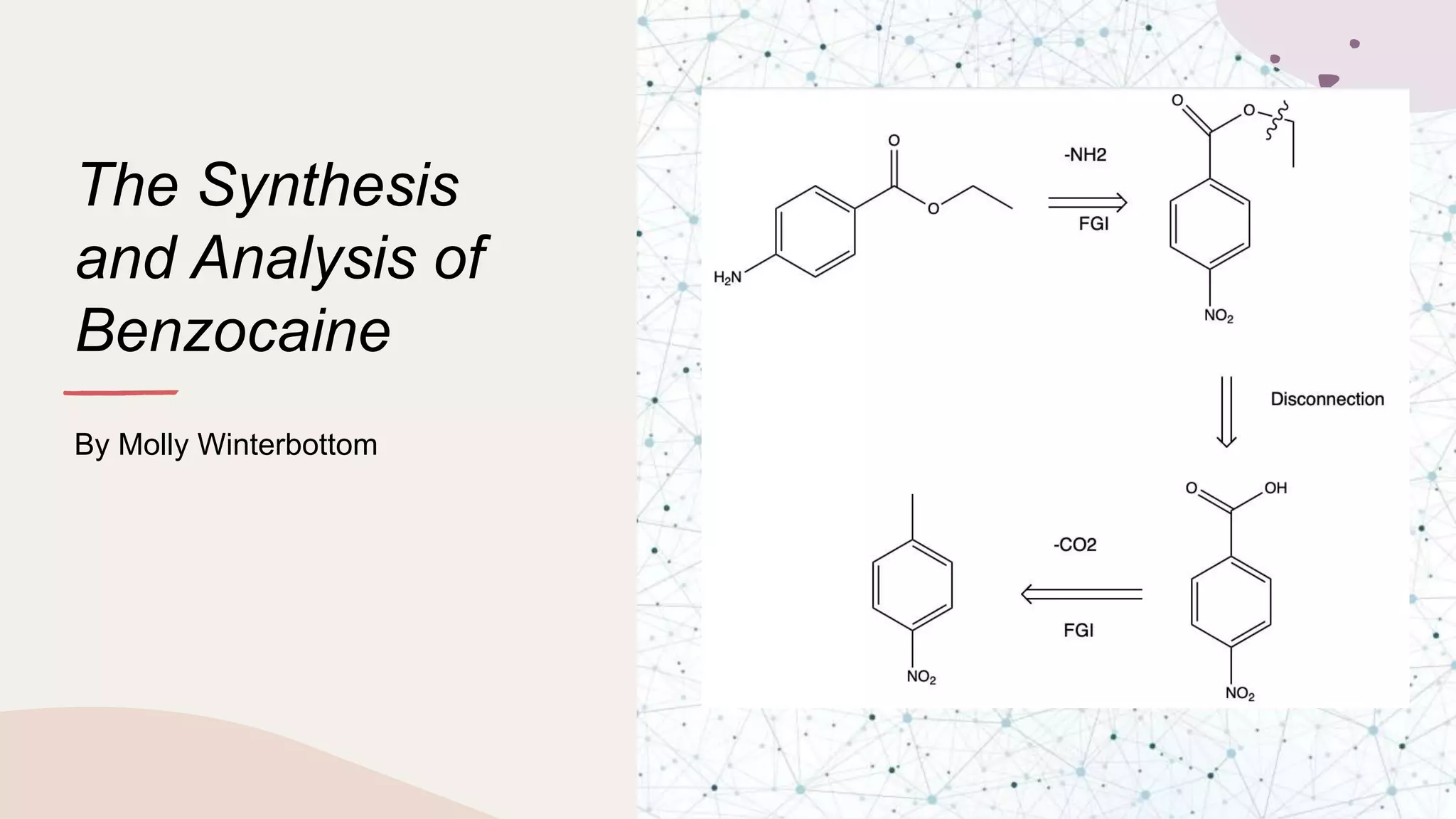
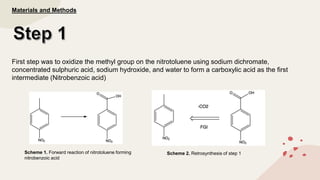
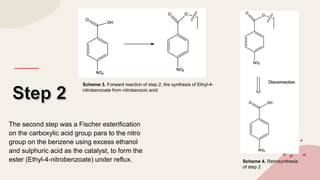
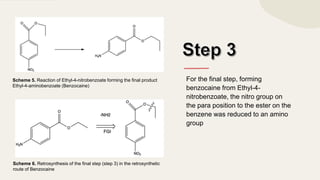
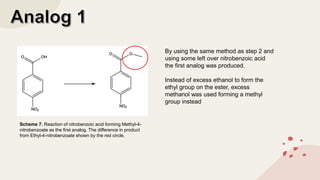
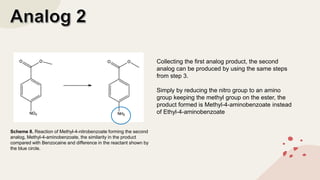
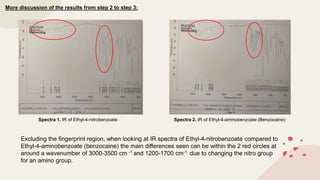
The document summarizes Molly Winterbottom's experiment to synthesize benzocaine through a three-step process from nitrotoluene. The first step was oxidizing nitrotoluene to form nitrobenzoic acid. The second step was a Fischer esterification of nitrobenzoic acid to form ethyl 4-nitrobenzoate. The third step reduced the nitro group to an amino group to form the final product, ethyl 4-aminobenzoate (benzocaine). TLC plates and IR spectroscopy confirmed the products of each step. Two analogs were also synthesized by modifying the second and third steps.