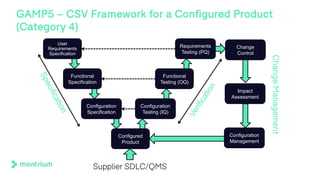
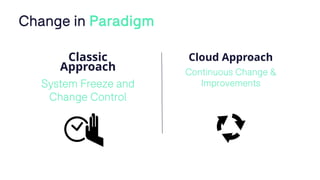
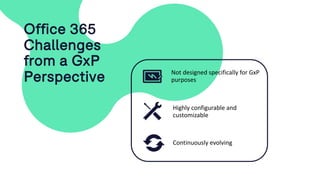
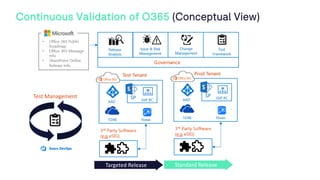
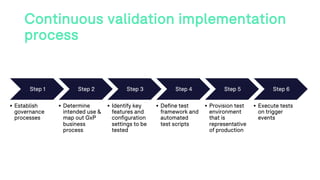
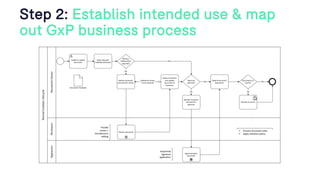
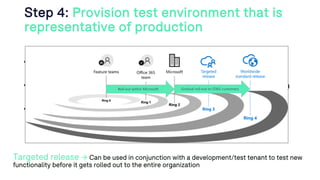
The document outlines a webinar discussing continuous validation of Office 365 compliance, emphasizing traditional validation models, key implementation steps, and tools available for continuous validation in a cloud context. It highlights the necessity of adapting validation practices to the evolving nature of cloud systems, especially in life sciences, and provides a structured implementation process for maintaining compliance. The session also includes polls on the audience's current practices and interest in further compliance resources.