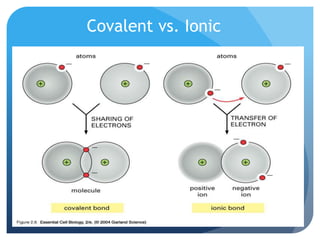
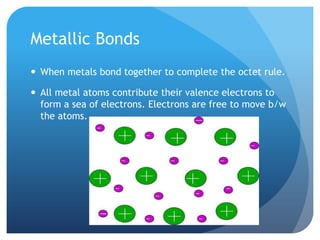
This document discusses the three main types of chemical bonds: ionic, covalent, and metallic. Ionic bonds form between ions when electrons are transferred from metals to nonmetals. Covalent bonds form when atoms share electrons in either single, double or triple bonds. Metallic bonds occur when metal atoms contribute electrons to form a "sea of electrons" that are free to move between atoms. Each bond type has distinct properties related to bonding strength, lattice structure, conductivity and melting/boiling points.