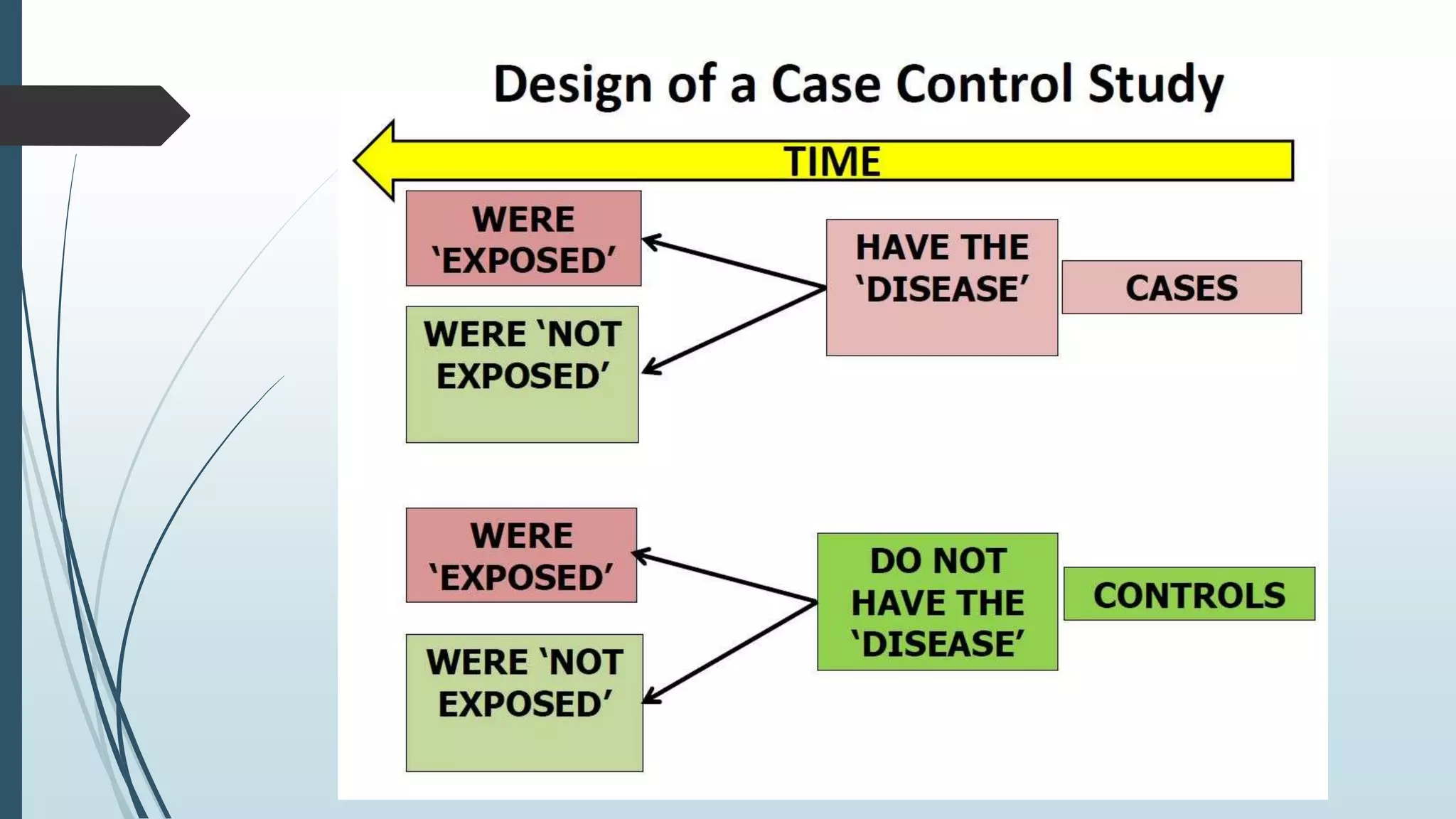
This document provides an overview of case-control surveillance studies. It discusses that case-control studies are retrospective and aim to determine if an exposure is associated with an outcome. The document outlines how cases and controls are selected and defined, and how odds ratios are used to interpret the association between an exposure and disease. It also discusses potential sources of bias and confounding, and notes some key advantages and disadvantages of the case-control study design.