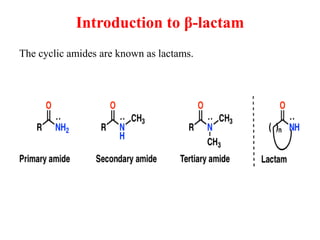
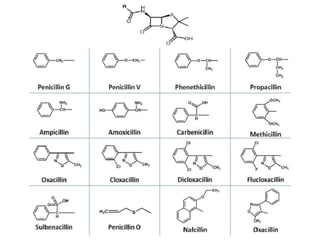
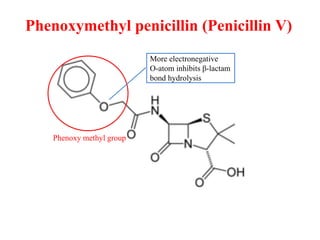
The document discusses various β-lactam antibiotics including penicillins and cephalosporins. It describes the chemical structure of penicillins including the β-lactam ring. It discusses the classes of penicillins, their history, mechanisms of action, degradation pathways, structure-activity relationships, resistance issues and classification. Specific penicillins discussed include benzylpenicillin, phenoxymethyl penicillin, methicillin, ampicillin, amoxicillin, oxacillin, cloxacillin and dicloxacillin.