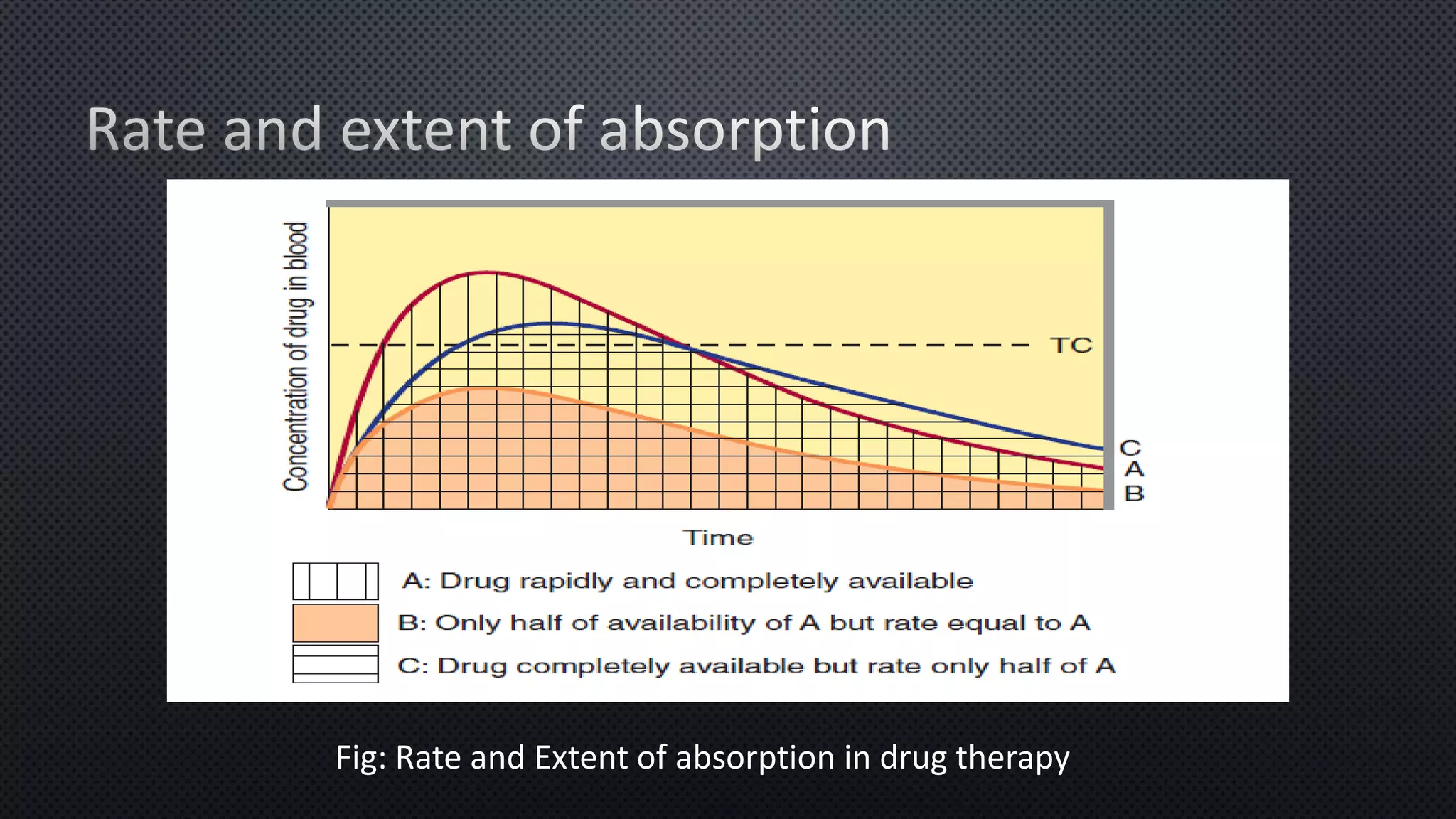
The document discusses the absorption and bioavailability of drugs, emphasizing factors influencing their extent of absorption through various routes. It explains how drug form, particle size, and physiological conditions can affect drug dissolution and absorption rates, as well as the concept of first-pass metabolism. Additionally, it highlights pharmacogenetic influences on bioavailability and presents questions regarding drug properties and administration routes.




















![Peak plasma concentration (Cmax) :
• Peak plasma concentration that gives an indication whether the drug is sufficiently
absorbed systemically to provide a therapeutic response
• The extent of bioavailability can be determined by following equation:
F =
[AUC]oral Div
[AUC]iv Doral
Where D stands for dose administered](https://image.slidesharecdn.com/9-200815115109/75/Absorption-Extent-and-Rate-22-2048.jpg)




















