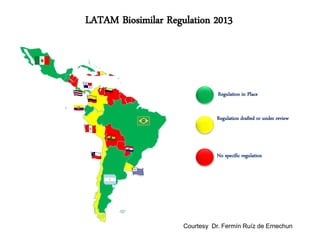
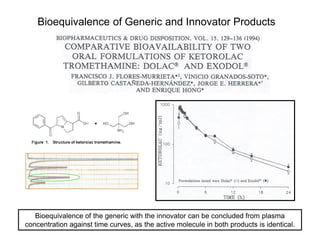
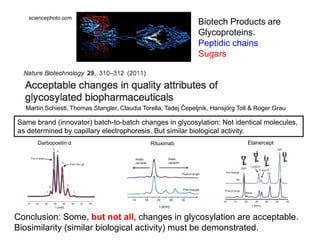
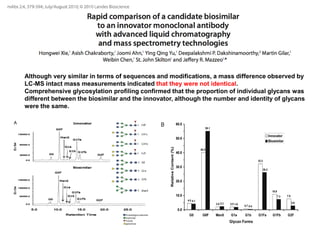
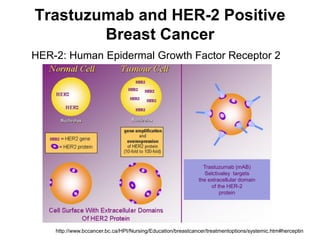
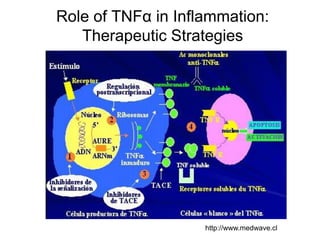
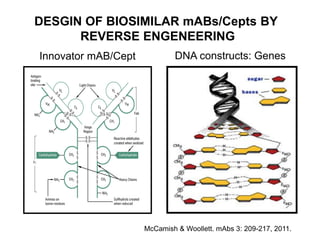
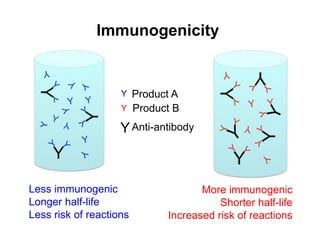
This document summarizes a keynote address about biosimilars and biotechnological medicines. It begins with statements that the speaker has received funding from various pharmaceutical companies but does not work for them. It then discusses that biotechnological medicines like proteins are much larger than conventional drugs. Biosimilars are similar but not generic versions of innovative biologics. The document outlines differences between biosimilars and intended copies, and concludes that biosimilars require their own clinical data to demonstrate similar efficacy and safety, while intended copies were approved without such regulations.