04 reactive flows - eddy disipation model
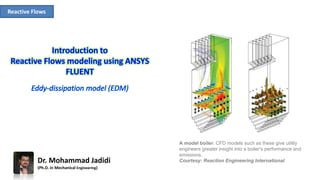
- 1. Reactive Flows A model boiler. CFD models such as these give utility engineers greater insight into a boiler's performance and emissions. Courtesy: Reaction Engineering InternationalDr. Mohammad Jadidi (Ph.D. in Mechanical Engineering)
- 2. Presented by: Mohammad Jadidi 2 Equations governing reacting flowsReactive Flows FLUENT can model the mixing and transport of chemical species by solving conservation equations describing convection, diffusion, and reaction sources for each component species. Conservation equations – Continuity equation (conservation of mass) – Transport of momentum – Transport of Energy – Transport of molecular species Equation of State Turbulence Transport – Transport of turbulent kinetic energy – Transport of turbulent dissipation rate – Transport of turbulent Reynolds stresses – Transport of moments such as 𝑢′ 𝑖 𝑌′ 𝑖
- 3. Presented by: Mohammad Jadidi 3 Species Transport EquationsReactive Flows To solve conservation equations for chemical species, ANSYS Fluent predicts the local mass fraction of each species, 𝑌𝑖 , through the solution of a convection- diffusion equation for the 𝑖 𝑡ℎ species 𝑅𝑖 is the net rate of production of species by chemical reaction 𝑅𝑖 modeling using eddy dissipation model (EDM) is discussed in details in this presentation NOTE: Reaction may occur as a volumetric reaction or be a surface reaction.
- 4. Presented by: Mohammad Jadidi 4 Species Transport Equations-reaction rates modelingReactive Flows 1- finite-rate kinetics: The effect of turbulent fluctuations on kinetics rates are neglected, and reaction rates are determined by general finite-rate chemistry directly. 2- Eddy-dissipation model (EDM): Reaction rates are assumed to be controlled by the turbulence, ignoring the effect of chemistry timescales 3- Eddy-dissipation-concept (EDC) model: Detailed chemical kinetics can be incorporated in turbulent flames, considering timescales of both turbulence and kinetics. See Previous lecture (part 2) Current lecture (part 3) See next lecture (part 4)
- 5. 5Presented by: Mohammad Jadidi 5 Species Transport Equations-Eddy-dissipation model (EDM)Reactive Flows 2- Eddy-dissipation model (EDM): Reaction rates are assumed to be controlled by the turbulence, ignoring the effect of chemistry timescales Based on the work of Magnussen and Hjertager (1976) Bjørn H. Hjertager Bjørn F. Magnussen
- 6. Presented by: Mohammad Jadidi 6 Reactive Flows Eddy-dissipation model (EDM) Under some combustion conditions, fuels burn quickly and the overall rate of reaction is controlled by turbulent mixing. For mixed-is-burned approximation, ANSYS Fluent provides a turbulence-chemistry interaction model called the Eddy-Dissipation Model
- 7. Presented by: Mohammad Jadidi 7 Reactive Flows Fast / Slow Chemistry Reactions limited by turbulent mixing Selection of turbulence closure model is important Combustion in Furnaces Boilers Gas Turbines Gasifiers, Incinerators Flares, etc. Fast Chemistry Da >> 1 Slow Chemistry Da ~ 1 Reactions limited by chemistry and turbulence interactions Turbulence/chemistry interactions are important Selection of reaction mechanism is important Reactions associated with Pollutants formation Ignition and Extinction Chemical Vapor Deposition (CVD) Non-Equilibrium Phenomenon Air dissociation at hypersonic speed
- 8. Presented by: Mohammad Jadidi 8 Reactive Flows Eddy-dissipation model (EDM) Basic idea of EDM: Remove the influence of chemistry A good assumption for fast reacting fuels (Da >> 1) Most of the useful fuels are fast burning Note #1: Brian Spalding (1971) suggested eddy break-up (EBU) model Introduced eddy lifetime ( = 𝑘 / 𝜀) Reaction rate is proportional to the inverse of eddy lifetime Note #2: F Magnussen and B. H. Hjertager (1976) adapted EBU and generalized it for non-premixed and partially premixed combustion Eddy dissipation model (EDM)`
- 9. Da >> 1 chemical reaction is fast relative to the transport processes in the flow. When reactants mix at the molecular level, they instantaneously form products. The model assumes that the reaction rate may be related directly to the time required to mix reactants at the molecular level. In turbulent flows, this mixing time is dominated by the eddy properties and, therefore, the rate is proportional to a mixing time defined by the turbulent kinetic energy, 𝑘, and dissipation, 𝜀. There is no kinetic control of the reaction process Presented by: Mohammad Jadidi 9 Reactive Flows Note: The eddy dissipation model is based on This concept of reaction control is applicable in many industrial combustion problems where reaction rates are fast compared to reactant mixing rates. Eddy-dissipation model (EDM)
- 10. Presented by: Mohammad Jadidi 10 Reactive Flows In EDM the net rate of production of species 𝑖 due to reaction 𝑟, 𝑅𝑖,𝑟 , is given by the smaller (that is, limiting value) of the two expressions below Eddy-dissipation model (EDM) In these equations the chemical reaction rate is governed by the large eddy mixing time scale, k/𝜺, as in the eddy-breakup model of Spalding. Combustion proceeds whenever turbulence is present (k/𝜺 > 𝟎), and an ignition source is not required to initiate combustion.
- 11. Presented by: Mohammad Jadidi 11 Reactive Flows The Eddy-Dissipation Model for LES When the LES turbulence model is used, the turbulent mixing rate , 𝜺 𝒌 , is replaced by the subgrid-scale mixing rate. 𝝉 𝑺𝑮𝑺 −𝟏 = 𝟐 𝑺𝒊𝒋 𝑺𝒊𝒋
- 12. Presented by: Mohammad Jadidi 12 Reactive Flows EDM is computationally cheap, but, for realistic results, only one or two step heat-release mechanisms should be used. WHY? The reason is that multi-step chemical mechanisms are typically based on Arrhenius rates, which differ for each reaction. In the eddy-dissipation model, every reaction has the same, turbulent rate, and therefore the model should be used only for one-step (reactant → product), or two-step (reactant → intermediate, intermediate → product) global reactions. The model cannot predict kinetically controlled species such as radicals. Eddy-dissipation model (EDM)
- 13. Presented by: Mohammad Jadidi 13 Reactive Flows Simple and physically based Applicable to every flow configuration Eddy-dissipation model (EDM) Rates are independent of temperature React towards complete products Cannot capture detailed chemistry effects Does not predict intermediate species and dissociation effects Temperature over-predicted Model constants sometimes require calibration DisadvantagesAdvantages
- 14. Presented by: Mohammad Jadidi 14 Reactive Flows Eddy-dissipation model (EDM)
- 15. Presented by: Mohammad Jadidi 15 Reactive Flows Species Transport tutorial #1
- 16. 16 Thanks Eddy-Dissipation-Concept (EDC) Model Next part: Reactive Flows https://ir.linkedin.com/in/moammad-jadidi-03ab8399 Jadidi.cfd@gmail.com Dr. Mohammad Jadidi (Ph.D. in Mechanical Engineering) https://www.researchgate.net/profile/Mohammad_Jadidi https://www.slideshare.net/MohammadJadidi
