Report
Share
More Related Content
What's hot
What's hot (20)
Types of reaction by Muhammad Fahad Ansari 12IEEM14

Types of reaction by Muhammad Fahad Ansari 12IEEM14
4th Lecture on Elements of groups 16, 17 & 18 | Chemistry Part I | 12th Std

4th Lecture on Elements of groups 16, 17 & 18 | Chemistry Part I | 12th Std
Viewers also liked
Viewers also liked (6)
Similar to Redox intro
Similar to Redox intro (20)
Ncert class 10 - science - chapter 1 - chemical reactions and equations

Ncert class 10 - science - chapter 1 - chemical reactions and equations
CBSE CLASS 10 CHEMISTRY CHAPTER 1 CHEMICAL REACTIONS AND EQUATIONS PART 2 TYP...

CBSE CLASS 10 CHEMISTRY CHAPTER 1 CHEMICAL REACTIONS AND EQUATIONS PART 2 TYP...
Oxidation – reduction reactions by Muhammad Fahad Ansari 12IEEM14

Oxidation – reduction reactions by Muhammad Fahad Ansari 12IEEM14
Redox Reactions. Chemical Reactions occurring in the body, Oxidation & Reduction

Redox Reactions. Chemical Reactions occurring in the body, Oxidation & Reduction
Recently uploaded
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...Postal Advocate Inc.
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Procuring digital preservation CAN be quick and painless with our new dynamic...

Procuring digital preservation CAN be quick and painless with our new dynamic...
Visit to a blind student's school🧑🦯🧑🦯(community medicine)

Visit to a blind student's school🧑🦯🧑🦯(community medicine)
Difference Between Search & Browse Methods in Odoo 17

Difference Between Search & Browse Methods in Odoo 17
AMERICAN LANGUAGE HUB_Level2_Student'sBook_Answerkey.pdf

AMERICAN LANGUAGE HUB_Level2_Student'sBook_Answerkey.pdf
Like-prefer-love -hate+verb+ing & silent letters & citizenship text.pdf

Like-prefer-love -hate+verb+ing & silent letters & citizenship text.pdf
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...
Influencing policy (training slides from Fast Track Impact)

Influencing policy (training slides from Fast Track Impact)
Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Tilak Nagar Delhi reach out to us at 🔝9953056974🔝
Redox intro
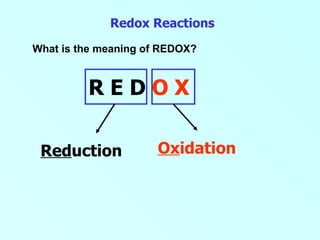
- 1. Redox Reactions What is the meaning of REDOX? R E D O X Ox idation Red uction
- 3. Defn 1 (In terms of oxygen) Oxidation = addition/gain of oxygen A substance that gains oxygen is said to be oxidised. Reduction = removal/loss of oxygen. A substance that loses oxygen is said to be reduced.
- 5. Example based on Defn 1 (In terms of oxygen) 2. Fe 2 O 3 + 3CO -> 2Fe + 3CO 2 Carbon monoxide (CO) is oxidised. Iron (III) oxide (Fe 2 O 3 ) is reduced.
- 6. Defn 2 (In terms of hydrogen) Oxidation = removal/loss of hydrogen A substance that loses hydrogen is said to be oxidised. Reduction = addition/gain of hydrogen. A substance that gains hydrogen is said to be reduced.