Combined sci c5 syllabus
•Download as PPT, PDF•
1 like•304 views
Combined Sci
Report
Share
Report
Share
Recommended
More Related Content
What's hot (20)
ENGINEERING CHEMISTRY- Solved Model question paper,2017-18

ENGINEERING CHEMISTRY- Solved Model question paper,2017-18
Viewers also liked
Viewers also liked (14)
Similar to Combined sci c5 syllabus
Similar to Combined sci c5 syllabus (20)
Updated ppt notes Unit-2_compressed_ec31d11e401e3dd93bf67ff9cda06486 (1).pdf

Updated ppt notes Unit-2_compressed_ec31d11e401e3dd93bf67ff9cda06486 (1).pdf
engineering material atomic structure of atom Lecture2.

engineering material atomic structure of atom Lecture2.
More from cartlidge
More from cartlidge (20)
Recently uploaded
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
TataKelola dan KamSiber Kecerdasan Buatan v022.pdf

TataKelola dan KamSiber Kecerdasan Buatan v022.pdf
18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf

18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Hybridoma Technology ( Production , Purification , and Application ) 

Hybridoma Technology ( Production , Purification , and Application )
Presiding Officer Training module 2024 lok sabha elections

Presiding Officer Training module 2024 lok sabha elections
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Combined sci c5 syllabus
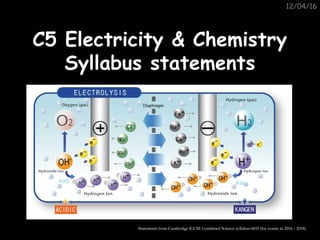
- 1. 12/04/16 C5 Electricity & ChemistryC5 Electricity & Chemistry Syllabus statementsSyllabus statements Statements from Cambridge IGCSE Combined Science syllabus 0653 (for exams in 2016 – 2018)
- 2. C5 Electricity & ChemistryC5 Electricity & Chemistry 1. State that electrolysis is the chemical effect of electricity on ionic compounds, causing them to break up into simpler substances, usually elements. 2. Use the terms electrode, electrolyte, anode and cathode. 3. Describe electrolysis in terms of the ions present and the reactions at the electrodes.
- 3. C5 Electricity & ChemistryC5 Electricity & Chemistry 4. Describe the electrode products, using inert electrodes, in the electrolysis of: •molten lead(II) bromide; •aqueous copper chloride. 5. Predict the products of the electrolysis of a specified binary compound in the molten state.