Anthracene.pdf
•
0 likes•58 views
This document discusses anthracene and phenanthrene, providing their IUPAC and common names, resonance structures showing four structures for anthracene and the structures for phenanthrene. It also describes that both undergo oxidation at the 9 and 10 positions on the central ring, with anthracene sacrificing 12 kcal/mol of energy and phenanthrene sacrificing 20 kcal/mol. Bromination of both occurs at the C9 position, with anthracene more prone to addition forming 9,10-dibromo-9,10-dihydro derivatives.
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
What's hot
What's hot (20)
Organic chemistry: Hydrocarbons, Alkyl Halides and alcohols

Organic chemistry: Hydrocarbons, Alkyl Halides and alcohols
synthetic Reagents and its application Diazomethane

synthetic Reagents and its application Diazomethane
Bayers theory & conformational analysis of cylohexane

Bayers theory & conformational analysis of cylohexane
Similar to Anthracene.pdf
Similar to Anthracene.pdf (20)
Phenanthrene, Haworth Synthesis, Chemical Properties .pptx

Phenanthrene, Haworth Synthesis, Chemical Properties .pptx
naphthalene for organic chemistry to understand industrial chemistry

naphthalene for organic chemistry to understand industrial chemistry
Polynuclear Hydrocarbons Preparations and Reactions

Polynuclear Hydrocarbons Preparations and Reactions
POLYNUCLEAR AROMATIC HYDROCARBON- PHARMACEUTICAL ORGANIC CHEMISTRY

POLYNUCLEAR AROMATIC HYDROCARBON- PHARMACEUTICAL ORGANIC CHEMISTRY
Sandrogreco Stereoselective Mannich Reaction Of Camphor Titanium Enolate

Sandrogreco Stereoselective Mannich Reaction Of Camphor Titanium Enolate
Heterocyclic compounds containing mono hetero atom pptx

Heterocyclic compounds containing mono hetero atom pptx
More from ShotosroyRoyTirtho
More from ShotosroyRoyTirtho (20)
202004156776434777433688633485334684357744578321651466499567c2a.pdf

202004156776434777433688633485334684357744578321651466499567c2a.pdf
ACFrOgC7khHosc_lpmK-qF_ncop6xRFmvof4eRovzDeBdhgNnZ-Cvyl3EiRkcq3OWucJ6R045G3UT...

ACFrOgC7khHosc_lpmK-qF_ncop6xRFmvof4eRovzDeBdhgNnZ-Cvyl3EiRkcq3OWucJ6R045G3UT...
Chapter 6 - Stereochemistry of Fused and Bridged Ring System.pdf

Chapter 6 - Stereochemistry of Fused and Bridged Ring System.pdf
Recently uploaded
Recently uploaded (20)
Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II

Food Chain and Food Web (Ecosystem) EVS, B. Pharmacy 1st Year, Sem-II
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
Asian American Pacific Islander Month DDSD 2024.pptx

Asian American Pacific Islander Month DDSD 2024.pptx
Role Of Transgenic Animal In Target Validation-1.pptx

Role Of Transgenic Animal In Target Validation-1.pptx
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi

Russian Escort Service in Delhi 11k Hotel Foreigner Russian Call Girls in Delhi
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes

Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Ecological Succession. ( ECOSYSTEM, B. Pharmacy, 1st Year, Sem-II, Environmen...

Ecological Succession. ( ECOSYSTEM, B. Pharmacy, 1st Year, Sem-II, Environmen...
Anthracene.pdf
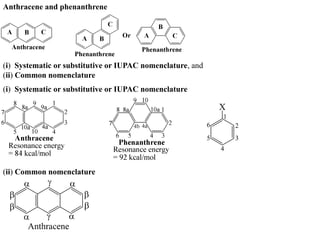
- 1. Anthracene and phenanthrene (i) Systematic or substitutive or IUPAC nomenclature, and (ii) Common nomenclature (i) Systematic or substitutive or IUPAC nomenclature (ii) Common nomenclature
- 2. Resonance structures of anthracene (Resonance energy = 84 kcal/mol) There are four resonance structures of anthracene which are shown below:
- 3. The resonance structures of phenanthrene are shown below (resonance energy 92kcal/mol).
- 4. Reactions of anthracene and phenanthrene (a) Oxidation reaction Both anthracene and phenanthrene are oxidized and reduced in the 9, 10 positions. Attack at the 9 and 10 positions of the middle ring of anthracene and phenanthrene leaves two benzene rings intact. Anthracene sacrifice 12 kcal/mol energy (84-2×36 =12; Phenanthrene sacrifice 20 kcal/mol energy (92-2×36=20).
- 5. Reduction of anthracene and phenanthrene
- 6. Electrophilic aromatic substitution reaction Bromination of anthracene and phenanthrene take place at the position-9 (at C9 ). In both cases, especially for anthracene, there is a tendency for addition to take place with the formation of 9,10-dibromo-9,10-dihydro derivatives. They also undergo nitration, sulfonation, Friedel-Crafts reactions.
- 8. Question: How can you chemically distinguish between anthracene and phenanthrene? Synthesis of anthracene (Haworth Synthesis)
- 9. Synthesis of phenanthracene (Haworth Synthesis)