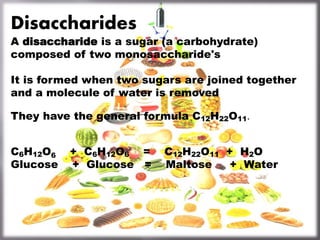
Important Disaccharides: Sucrose, Lactose, Maltose
- 1. Disaccharides A disaccharide is a sugar (a carbohydrate) composed of two monosaccharide's It is formed when two sugars are joined together and a molecule of water is removed They have the general formula C12H22O11. C6H12O6 + C6H12O6 = C12H22O11 + H2O Glucose + Glucose = Maltose + Water
- 2. Important Disaccharides are:- • sucrose (glucose and fructose) • lactose (glucose and galactose) • maltose (2 molecules of glucose)
- 4. Maltose Maltose does not have a specific function in the body Maltose or malt sugar, is a disaccharide formed from two units of glucose. It is formed by O-glycosidic linkage between one molecule of α-D- glucose with another molecule of α-D- glucose Maltose is the disaccharide produced when amylase breaks down starch. Maltose can be broken down into two glucose molecules by hydrolysis.
- 5. In living organisms, the enzyme maltase can achieve this very rapidly. Formed in vivo by the action of salivary amylase on starch and in vitro by the partial hydrolysis of starch by acids. Produced commercially by hydrolysis of starch. Malt sugar,present in germinating seeds. Used as fementive reagent.
- 6. Lactose Commercially known as milk sugar. Lactose is a disaccharide derived from the condensation of galactose and glucose. Contains O-glycosidic linkage between one molecule of galactose with another molecule of glucose. Lactose is milk sugar and found in appreciable quantities in milk to the extent of about 5%. Specific enzyme which hydolyses is lactase present in intestinal juice.
- 7. Lactase in mucosal cells hydrolyzes lactose to its constituent monosaccharides. Milk sugar,cows milk contains 5% lactose,human milk contains 7%. Bacteria cause fermentation of lactose forming lactic acid. When these reaction occur ,it changes the taste to a sour The nutritional source of energy for infants during nursing Lactose makes milk tastes sweet and is an ingredient in many processed foods that contain dairy
- 8. Manufacturers add whey, a byproduct of dairy production that contains lactose, to certain food products, such as breads, cookies, cakes, doughnuts, breakfast bars and ice cream Many people are lactose intolerant and do not produce sufficient amounts of lactase to digest lactose, causing such symptoms as nausea, diarrhea, gas You can take a lactase supplement to help you digest lactose and ameliorate the symptoms.
- 9. Sucrose Sucrose is the disaccharide present most in the human diet Sucrose is commonly known as table sugar Is a sugar used at home The molecule is a disaccharide derived from glucose and fructose with the molecular formula C12H22O11. On hydrolysis yields one molecule each of D- glucose and D- fructose.
- 10. It is very soluble and very sweet. The specific enzyme which hydrolyses sucrose is sucrase present in intestinal juice. Sucrose is dextrorotatory,because glucose and fructose(furanose form) are dextrorotatory. This 50:50 mixture of glucose and fructose is called invert sugar because it reverses the rotation of polarized light Invert sugar is more sweeter than sucrose due to the presence of free fructose. Honey is rich in invert sugar Jams and jellies also contain invert sugar
- 11. Source of sucrose is sugar beets and sugar cane. Caramel is solid residue produced by heating sucrose. Used in pharmaceutically to make syrups.
- 12. Reducing properties In maltose The aldehyde groups are at carbon 1 in each of the original glucose molecules. Since the linkage is 1.4, one free aldehyde group remains. Therefore, maltose acts as a reducing sugar. In sucrose The glucose part had the aldehyde at carbon 1, and the fructose part had the ketone group at carbon 2. since the linkage is 1,2, neither group is free. Therefore, sucrose is not a reducing sugar. In lactose Which has a 1,4-linkage, acts as a reducing sugar because both of the original aldehyde group wre on carbon 1, and one of them is free to react.
- 13. Fermentation Sucrose and maltose will ferment when yeast is added because yeast contains the enzyme sucrase and maltase. Lactose will not ferment because yeast does not contain lactase.
- 14. Testing for disaccharides The chemical reactions of these sugars can be used to distinguish them in the laboratory. If you have 2 test tubes containing a disaccharide,C12H22O11.To determine if it is sucrose lactose or maltose. We can use the alkaline Cu complex reaction of glucose and the principle of fermentation.
- 15. In terms of Solubility Sucrose – very soluble in water Maltose – fairly soluble Lactose – only slightly soluble The disaccharides, just like monosaccharide are white crystalline, sweet solids. The disaccharides are also optically active; they rotate the plane of polarized light. However, even though they are soluble in water, they are too large to pass through cell membranes.
- 16. Polysaccharides • Are polymers of monosaccharides. • Hydrolysis produces many molecules of monosaccharide. • Can be formed from pentoses or the 5 carbon sugars or the hexoses or six carbon sugars. • Those from pentoses are called pentosans • Those from hexoses are hexosans or sometimes called the glucosans. • The hexosans are most common in terms of physiology. • The hexosans have the general formula ( C6H10O5 )x Where x is a large number. Some of the common hexosans are starch, cellulose and dextrin.
- 17. Homopoysaccharides Homopoysaccharides are polymers composed of a single type of sugar monomers STARCH DEXTRINS GLYCOGEN CELLULOSE CHITIN INULIN
- 18. STARCH Starch is a polymer consisting of D-glucose units. Starches (and other glucose polymers) are usually insoluble in water because of the high molecular weight, but they can form thick colloidal suspensions with water. Main sources of starch are rice, corn, wheat, potatoes. A storage polysaccharide in plants. It contains two polysaccharide units,amylose and amylopectin.
- 19. STARCH –AMYLOSE Amylose consists of long unbranched chains of Alpha D- glucose(50-5000 glucose units) connected by ᾳ(1→4) glycosidic linkages. 10%-20% of the starch in plants is in this form. Water soluble. Amylose forms helices (coils) which can trap molecules of iodine, forming a characteristic deep blue-purple color(Iodine is often used as a test for the presence of starch).
- 20. STARCH — AMYLOPECTIN Amylopectin consists of long chains of glucose up to 1 million glucose units connected by ᾳ(1→4) glycosidic linkages, with ᾳ(1→6) branches every 24 to 30 glucose units along the chain. 80-90% of starch is in this form in plants.
- 21. ACTION OF ENZYMES ON STARCH The enzymes hydrolyzing starch are called amylases. Saliva contains salivary amylase and pancreatic juice contains pancreatic amylase. These amylases are α amylases and hydrolyze starch into maltose,isomaltose and α dextrins.
- 22. DEXTRINS These are immediate products of hydrolysis of starch by acids or by the enzyme amylase. Consists of complex mixture of molecules of different sizes and structures. Present in the leaves of all starch producing plants. Sweet in taste. α- dextrins have eight α -D-glucose residues showing mostly glycosidic linkages in addition to an α(1 →4) linkage at α(1 →6) each of branching site. It is occasionally used to replace lost blood in emergency situations, when replacement blood is not available. It is used in some eye drops as a lubricant. Also used as molecular sieves to separate proteins and other large molecules (gel filtration chromatography)
- 23. GLYCOGEN Also known as animal starch. Stored in muscle and liver (mostly). Present in cells as granules (high MW). Contains both a(1,4) links and a(1,6) branches at every 12- 14glucose unit (more frequent than in starch). Forms a colloidal solution in water and gives a red color with iodine. Glycogen is formed in the body cells from molecules of glucose. In a process called glycogenesis. When glycogen is hydrolyzed to glucose, the process is called glycogenolysis.
- 24. CELLULOSE A polymer consisting of long,unbranched chains of D-glucose connected by glycosidic linkages,may contain from 15000 glucose units in one molecule.
- 26. INULIN b-(1,2) linked fructofuranoses linear only; no branching Lower molecular weight than starch Hydrolysis yields fructose Sources include onions, garlic etc. Used as diagnostic agent for the evaluation of glomerular filtration rate (renal function test)
- 27. CHITIN chitin is the second most abundant carbohydrate polymer Like cellulose, chitin is a structural polymer present in the cell wall of fungi and in the exoskeletons of crustaceans, insects and spiders Consists of N-acetyl-D-glucosamine units joined by
- 28. Heteropolysaccharides In general, heteropolysaccharides (heteroglycans) contain two or more different monosaccharide units Such as: Hyaluronic acid Heparin
- 29. Hyaluronic acid Hyaluronan is a polymer of disaccharides, themselves composed of D-glucuronic acid and D-N-acetylglucosamine, linked via alternating β-1,4 and β-1,3 glycosidic bonds. Hyaluronan can be 25,000 disaccharide repeats in length Hyaluronan is also a major component of skin, where it is involved in tissue repair Hyaluronan is a common ingredient in skin-care products. Distribution in connective tissue, skin etc.