EMRadiation.ppt
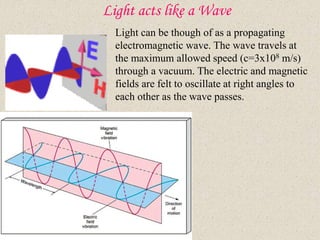
- 1. Light acts like a Wave Light can be though of as a propagating electromagnetic wave. The wave travels at the maximum allowed speed (c=3x108 m/s) through a vacuum. The electric and magnetic fields are felt to oscillate at right angles to each other as the wave passes.
- 2. Properties of a Wave The distance between crests or troughs is the wavelength l. If the wave travels at speed c, the crests will pass with a frequency f. The relation between these is l x f = c. People wondered what “medium” the light waves are travelling in. Today we just say it is the vacuum (which isn’t totally empty in modern physics. We also now know that the speed in this medium is the same no matter how fast the emitter is travelling (very odd: a result of Einstein’s relativity).
- 3. Light also acts like a particle : the “Photon” When detectors (or atoms) “see” light, it arrives in discreet packages, which we call “photons”. You can think of each photon as having a wavelength. The energy of a photon depends on its wavelength or frequency: E ~ f ~ c / l . As a whole, the photons have the statistical behavior of waves of that wavelength. They experience “interference” like waves.
- 4. Light is Electromagnetic Radiation All wavelengths (or energies) of light are the same basic stuff. Together they constitute the “electromagnetic spectrum”. Visible light is a tiny portion of this. Although our eyes cannot detect the rest of the spectrum, we now have detectors that can. We give different names to the different “colors”. We use different units for the different wavelengths – whatever is convenient.
- 5. The Spectrum of E-M radiation Astronomical objects are capable of producing different parts of the spectrum depending on how energetic the processes that are going on, or how hot the object is.
- 7. Thermal or “Blackbody” Radiation Any opaque body produces E-M radiation characteristic of its temperature. It follows the “Planck curve” shape, which has a peak. The wavelength of the peak follows “Wein’s law”: lmax(nm)=3x106 / T(K) (so hotter sources are bluer). The total energy emitted (or total area under the curve) by hotter sources of the same size goes up like T4 , and they are brighter at all wavelengths. This is called “blackbody” radiation because it’s what you get from a little hole in a dark cavity, or a black-looking absorber (which is also an excellent emitter). A better name is “thermal” radiation, because it is related to T.
- 8. Astro Quiz • Two stars are the same distance and size, but one looks brighter. It must also be hotter. • Two stars are the same distance and temperature, but one looks brighter. It must also be larger. • Two stars are the same size and temperature, but one looks brighter. It must also be bluer. Which statement below is FALSE? Reminder: The wavelength of the peak follows “Wein’s law”: lmax(nm)=3x106 / T(K) (so hotter sources are bluer). The total energy emitted (or total area under the curve) by hotter sources of the same size goes up like T4 , and they are brighter at all wavelengths.
- 9. Thermal Radiation from Objects There are also a variety of “non-thermal” processes (often involving magnetic fields) which produce radiation at all wavelengths (and can produce VERY high energy radiation) all the way up through gamma rays. They are often associated with violent phenomena (explosions, black holes, etc.).
- 10. Energy Levels in Atoms We can think of an atom as consisting of a positive nucleus (protons and neutrons) surrounded by negative electrons. The electrons can be thought of as “orbiting” the nucleus, but are only allowed in certain orbits (or energy levels). A photon with exactly the right energy can excite the electron from one level to another. The electron will drop back to the “ground state”, and emit photons with specific energies as it does so. “Chemistry” is caused by the fact that no 2 identical electrons can be in the same orbital at the same time.
- 11. Energy transitions and photons The energy of photon that can interact with a level jump just depends on the energy difference between the levels. Levels can be skipped.
- 12. Unique Atomic Signatures Each atom has a specific set of energy levels, and thus a unique set of photon wavelengths with which it can interact.
- 13. The Doppler Shift – how it works c v rest rest new l l l l When a source is moving, an observer gets the waves either stretched out or crunched together, depending on their relative motion with the source. In the case of light, longer wavelengths look redder and shorter wavelengths look bluer. This is given by the Doppler formula: v is negative for an approaching source: if the distance is shrinking, the wavelength is too To get an appreciable change, you have to be moving with an appreciable fraction of the speed of the wave
- 14. The Doppler Shift – how we use it Atomic energy transitions leave features in the spectrum whose rest wavelengths are known from laboratory work. We can measure observed shifts in these wavelengths from astronomical objects, and see how fast they are moving (you only get the line-of-sight motion: towards or away from you). More subtle analysis can also yield other motions, like rotation or turbulent motions. These are all direct uses of the Doppler shift. It doesn’t matter how far away the source is, either.