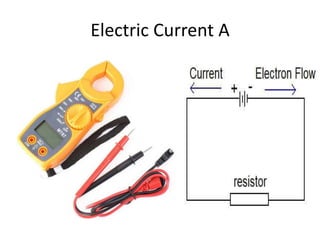
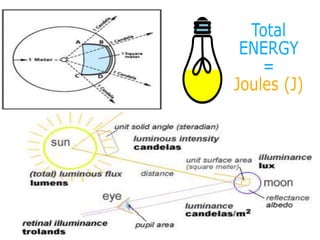
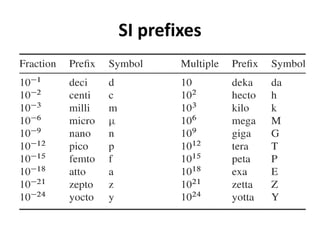
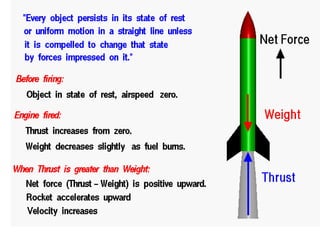
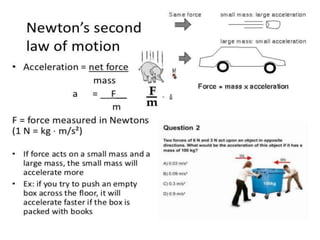
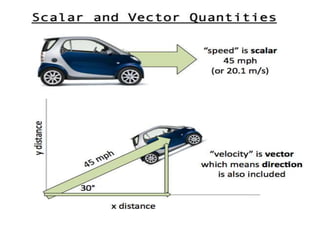
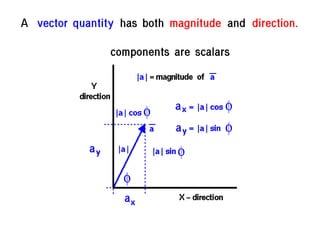
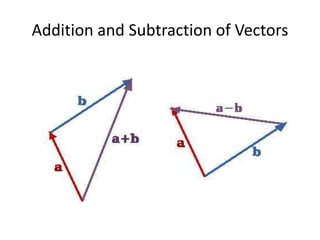
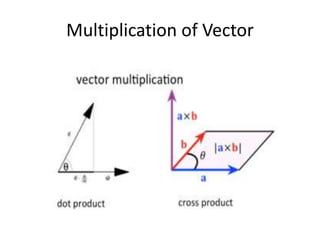
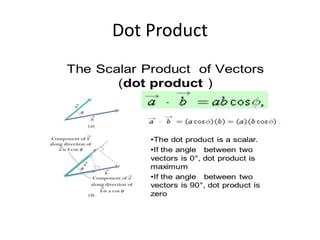
This document discusses units of measurement in the International System of Units (SI) and various physics concepts. It begins by introducing the seven base SI units - meter, kilogram, second, ampere, kelvin, mole, and candela. It then defines each unit, how it relates to physical quantities, and how it is measured. The document also covers derived units, SI prefixes, physical quantities, Newton's laws of motion, and vector properties including addition/subtraction, multiplication, dot products and cross products.