Metalsnon metalsandmetalloids
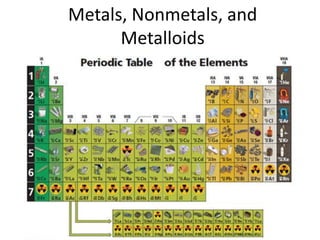
- 3. Color and label your periodic table like the one below:
- 4. • Put the colored periodic table to the side, we will glue it in later • Find this page, we will begin taking notes:
- 5. Metals • Most elements are metals. • The 88 elements to the left of the stair step line are metals or metal-like elements except Hydrogen. Examples of metals:
- 6. Physical Properties • A physical property is a characteristic you can observe or measure • In our lesson yesterday, we focused on physical properties. • For example, a physical property of some metals are that they are shiny.
- 7. Physical Properties of Metals • Good conductors of heat and electricity. • Luster: shiny • Ductile (can be stretched into thin wires) • Malleable (can be pounded into thin sheets)
- 8. Properties of Metal, Cont. • Usually solids at room temperature • High melting points and boiling points (except Hg - Mercury) • High densities and reflect light
- 9. Chemical Properties of Metals • A chemical property of metals is that it Corrodes easily – Gradual wearing away – Ex: Silver tarnishing or iron rusting
- 10. What are Chemical Properties? • A chemical property may only be observed by chemical reaction. • This property measures the potential for undergoing a chemical change. • A chemical reaction is the process by which one or more substances change to produce one or more different substances.
- 11. Say what? • Ok, let’s look at it another way: • Why is Mater the color of orange dirt? Rust (that orange coloring) is caused by iron coming into contact with oxygen molecules. That ability to react with oxygen is a chemical property of iron!
- 12. Metalloids • The periodic table contains a stair-step line, these elements along that line are called metalloids. • The “-oid” ending means – like or similar to • Elements in this group have properties of both metals and non-metals.
- 13. Physical Properties of Metalloids • Solids that can be shiny or dull. • They can conduct heat and have electrical charges,but not as well as metals. • They are ductile and malleable. Silicon
- 14. Non-Metals • Nonmetals are found on the right side of the periodic table. • Their characteristics are opposite those of metals.
- 15. Physical Properties of Non-Metals • poor conductors of heat and electricity. • Non-metals are not ductile or malleable. • Solid non-metals are brittle and break easily. • Dull, no shiny luster, don’t reflect light. • Many non-metals are gases. Sulfur
- 16. Important Words to Know! • So now that you’ve got a good understanding of metals, nonmetals and metalloids… • Let’s make sure you know the properties that describe them!
- 17. Malleable • Matter that can be pounded or rolled into thin sheets without shattering.
- 18. Brittle • Matter that shatters and breaks into pieces when pounded
- 19. Conductor • Allows heat or electrical charge can pass through material • Does metal have good conductivity? Give me an example!
- 20. Insulator • Does not allow electricity to pass through easily – Known as a poor conductor, what in this picture would you consider to be the insulator?
- 21. Luster • The way matter reflects light from its surface • Matter that reflects a large amount of light is called SHINY (write: high light=shiny) • Matter that reflects little light from its surface is called DULL. (write: low light=dull)
- 22. Ductile – • ability to be stretched into a wire
- 24. Next ….. • Find this page • Color these properties to match the periodic table of elements Metals – blue Non-metals – green Metalloids – purple • Now glue these in UNDER your periodic table
- 25. This is what your page in your science notebook will look like when we are done. METALS •Good conductors of heat and electricity •Luster is shiny (reflect light) •Ductile •Malleable •Corrosive in water •Solid at room temperature •High density •High melting point and boiling points NON-METALS •Poor conductors of heat and electricity •Luster is dull (don’t reflect light) •Non Ductile •Not Malleable •Brittle •Exist in 3 states of matter (but many are gases) METALLOIDS •Have properties of both metals and non-metals •Luster is shiny or dull •Ductile •Malleable •Do conduct heat and electricity, but not as well as metals 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Groups 1 2 3 4 5 6 7 Periods Warm-Up for Thursday, 9/15/11 Color and label your periodic table as the one below:
- 26. Learning Check
