Transfection methods
- 1. Aakifah Amreen H.E MSc (BT)
- 3. The cell membrane consists of a phospholipid bilayer with embedded proteins and carries a net negative charge. Thus, It presents an impenetrable barrier to large molecules that, like the phosphate backbones of DNA and RNA, are also negatively charged. To sneak nucleic acids through the cell membrane, researchers have developed a number of techniques each using a different approach—from using chemicals and carrier molecules that coat the nucleic acids to neutralize them to physical methods that create transient pores in the membrane to introduce the DNA directly into the cell. Introduction
- 4. Transfection Is the process of artificially introducing nucleic acids (DNA or RNA) into cells, utilizing means other than viral infection. Such introductions of foreign nucleic acid using various chemical, biological, or physical methods can result in a change of the properties of the cell.
- 5. Transfection technologies available today can be broadly classified into three groups: chemical, biological, and physical. Chemical methods that use carrier molecules to neutralize or impart a positive charge to the negatively charged nucleic acids Biological methods that rely on genetically engineered viruses to transfer non-viral genes into cells Physical methods directly deliver nucleic acids into the cytoplasm or the nucleus of the cell
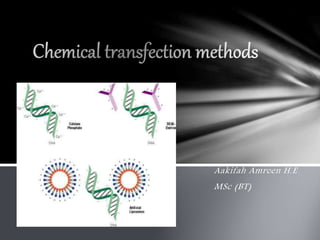
- 6. Chemical transfection is a popular technique due to the ease, cost, and wide variety of transfection reagents available. There are different methods included : DNA transfer by calcium chloride Transfer of DNA by polyethelene glycol Use of DEAE- Dextran for DNA transfer Liposome mediated transfer Chemical methods
- 7. Lipofection is the transfection using liposomes, small molecules which can fuse with cell membranes and are able to release their contents. Liposomes can be lipid-based (more common), or non-lipid based. Cationic polymers, carrying a positive charge, bind well to negatively charged nucleic acids, and are another method of transfection. Lipofection
- 8. Principle Lipofection is to associate nucleic acids with cationic lipid formulation. The resulting molecular complexes, known as lipoplexes, are then taken up by the cells. The main advantages of lipofection are its high efficiency, its ability to transfect all types of nucleic acids in a wide range of cell types, its ease of use, reproducibility and low toxicity. In addition this method is suitable for all transfection applications Working The lipid-based reagents used for lipofection are generally composed of synthetic cationic lipids that are often mixed with helper lipids such as DOPE or cholesterol. These lipids mixture assembles in liposomes or micelles with an overall positive charge at physiological pH and are able to form complexes (lipoplexes) with negatively charged nucleic acids The association of the lipid-based transfection reagent with nucleic acids results in a tight compaction and protection of the nucleic acids and these cationic complexes are mainly internalized by endocytosis.
- 9. Once inside the cells two mechanisms leading to the nucleic acids release into the cytoplasm: 1. The endosomes buffering capacity of the polycationic residues. 2. the ability of cellular negatively charged lipids to neutralize the cationic residues of the transfection reagent leading to destabilization of endosomal membranes. Finally, the cellular and molecular events leading to the nuclear uptake of DNA following by gene expression remain highly speculative. However, the significance of cell division on transfection efficiency favours the assumption that nuclear membrane disruption during the mitosis process promote DNA nuclear uptake. Continued…..
- 11. Competent cells are bacterial cells that can accept extra-chromosomal DNA or plasmids (naked DNA) from the environment. The generation of competent cells may occur by two methods: natural competence and artificial competence. oNatural competence is the genetic ability of a bacterium to receive environmental DNA under natural or in vitro conditions. oArtificial competence Bacteria can also be made competent artificially by chemical treatment and heat shock to make them transiently permeable to DNA. Calcium chloride mediated gene transfer
- 12. Calcium chloride transformation technique is the most efficient technique among the competent cell preparation protocols. It increases the bacterial cell’s ability to incorporate plasmid DNA, facilitating genetic transformation. Addition of calcium chloride to the cell suspension allows the binding of plasmid DNA to LPS. Thus, both the negatively charged DNA backbone and LPS come together and when heat shock is provided, plasmid DNA passes into the bacterial cell. Principle
- 14. Prepare a overnight culture of the bacteria in LB broth. Grow at 37°C without shaking. About 2 h before to begin the main procedure, use 1.0 mL of the overnight culture to inoculate 100 mL of fresh LB broth. This culture is grown with rapid shaking at 37°C until it reaches roughly 5 x 107 cells/ml. Thus corresponds to an OD650 for culture Take a 5 mL aliquot of each transformation reaction and transfer to sterile plastic centrifuge tubes. Cool on ice for 10 mm. Pellet the cells by spinning for 5 min at 5000g. It is necessary for the centrifugation to be performed at 4°C.. Pour off the supernatant and resuspend cells in 25 mL of cold 0.1M CaCl2. Leave on ice for at least 20 min. Centrifuge as in Step 3. Procedure
- 15. Resuspend the cells in 0.2 mL of cold 0.1M CaCl2. Transfer the suspensions to sterile, thin-walled glass bottles or tubes. The use of glass makes the subsequent heat shocks more effective. To each tube add up to 0.1 mg of DNA, made up in a standard DNA storage buffer such as TE to a volume of 100 mL. Leave on ice for 30 min. Transfer to a 42°C water bath for 2 min and return briefly to ice. Transfer the contents of each tube to 2 mL of LB broth in a small flask. Incubate with shaking at 37°C for 60-90 min. Plate 0.1 mL aliquots of undiluted, 10-1 and 10-2 dilutions onto LB plates to which the antibiotics to be used for selection have been added. Incubate overnight at 37°C.
Editor's Notes
- The terminology used for various gene delivery systems has evolved to keep pace with technological advances in the field and further refined to distinguish various methods and cell types.
- DOPE (1,2-dioleoyl-phosphatidyl-ethanolamine)
- LPS -lipopolysaccharide(LPS) receptor molecules on the competent cell surface.