Embed presentation
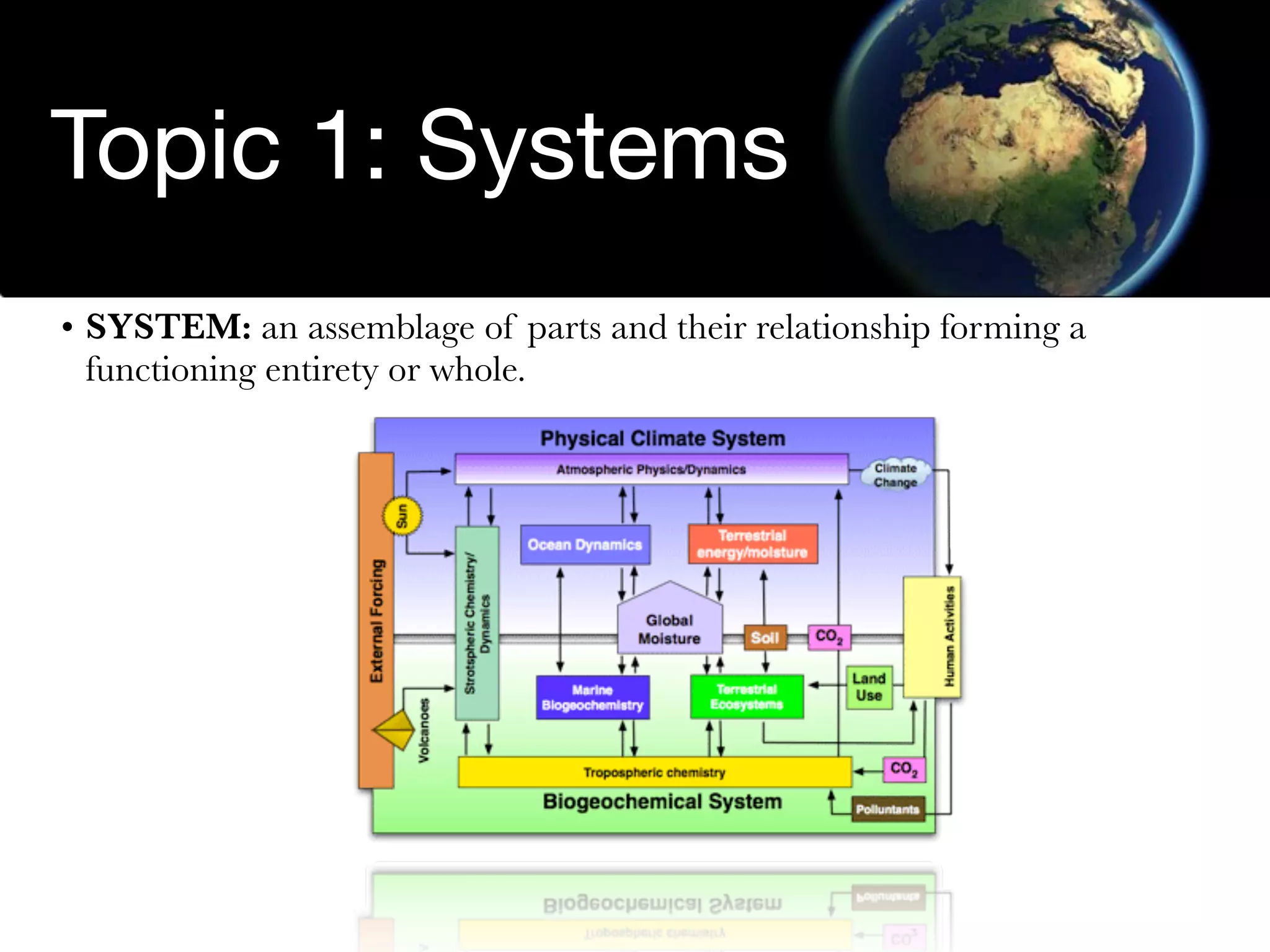

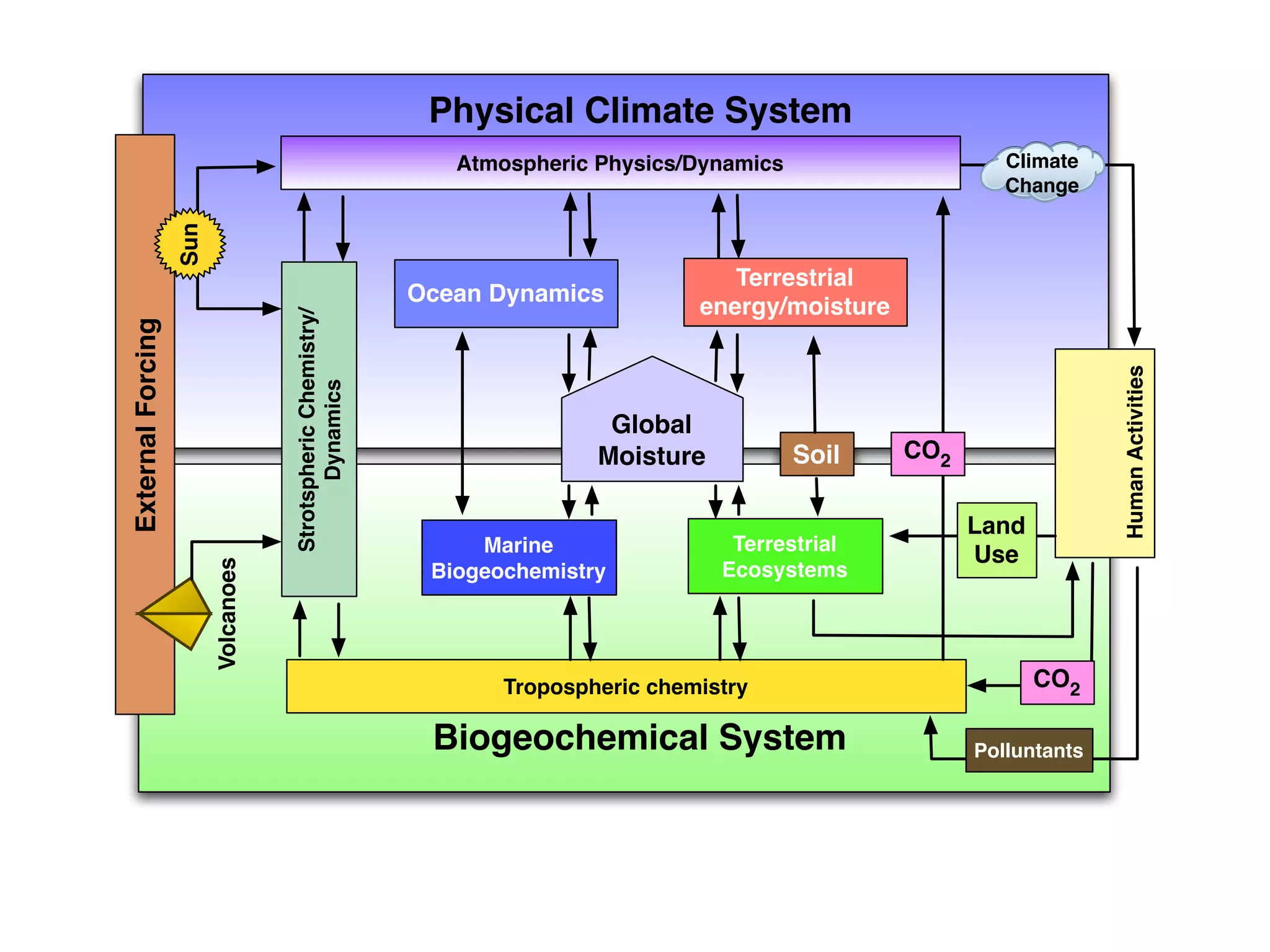
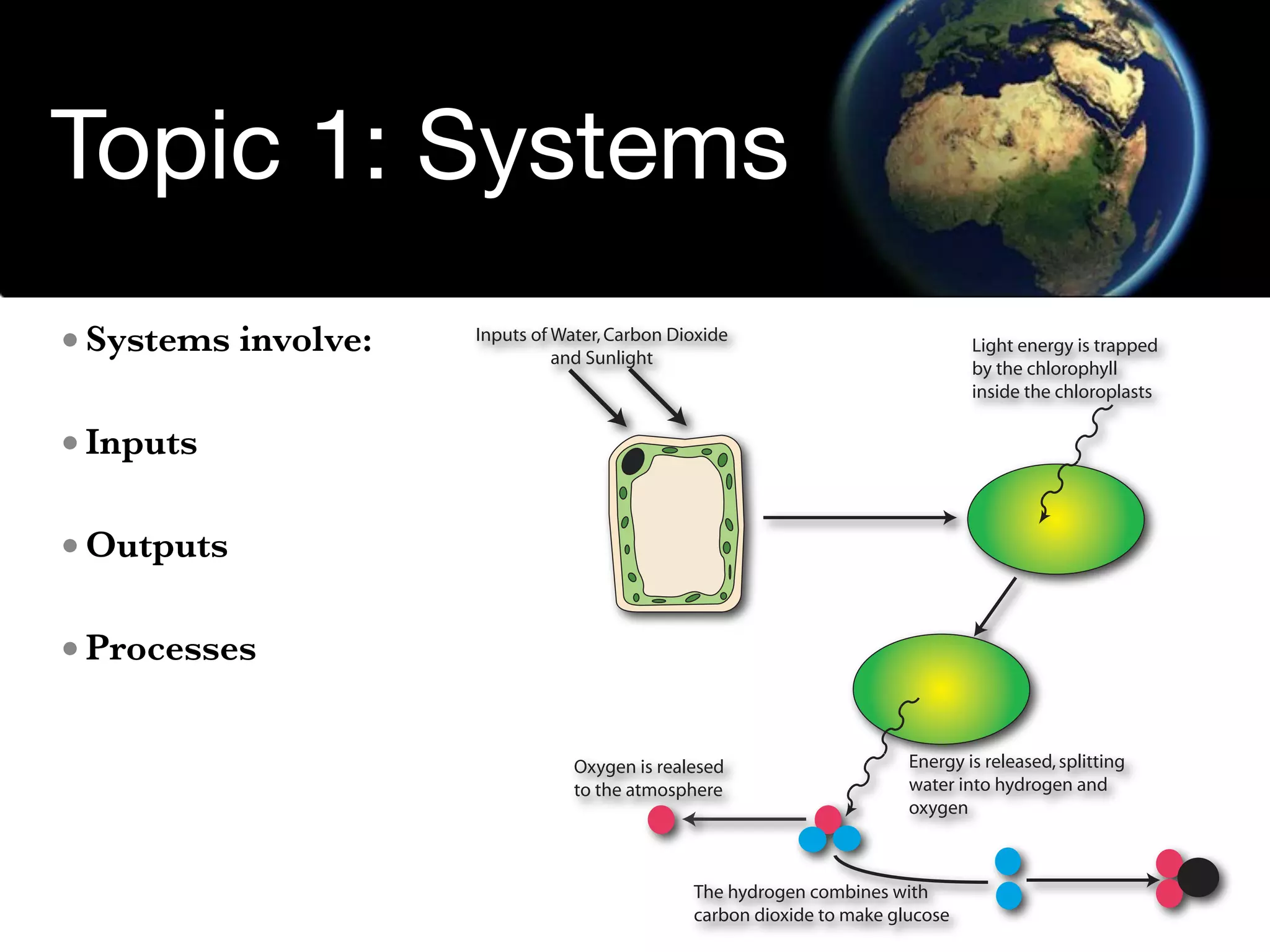

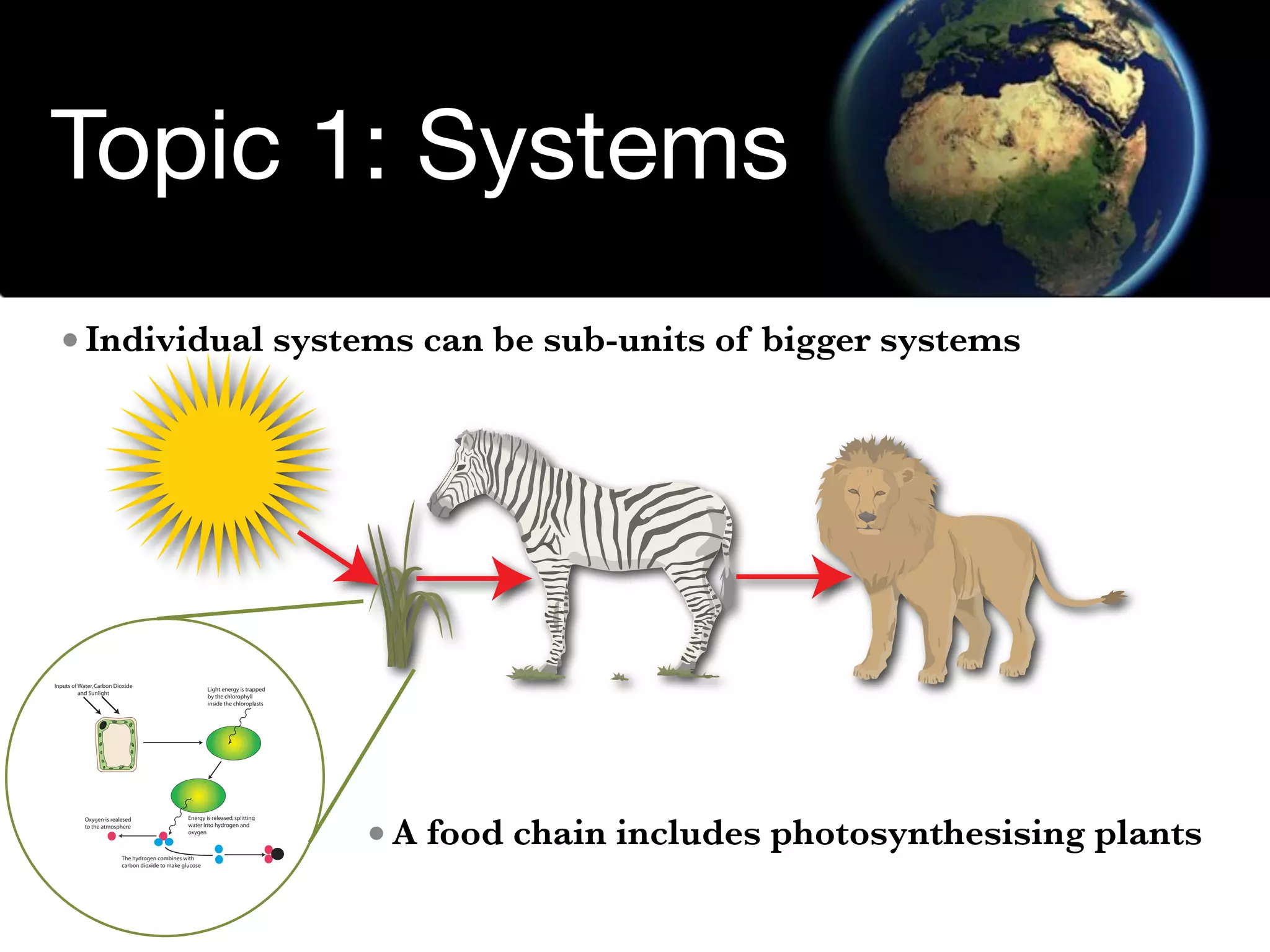







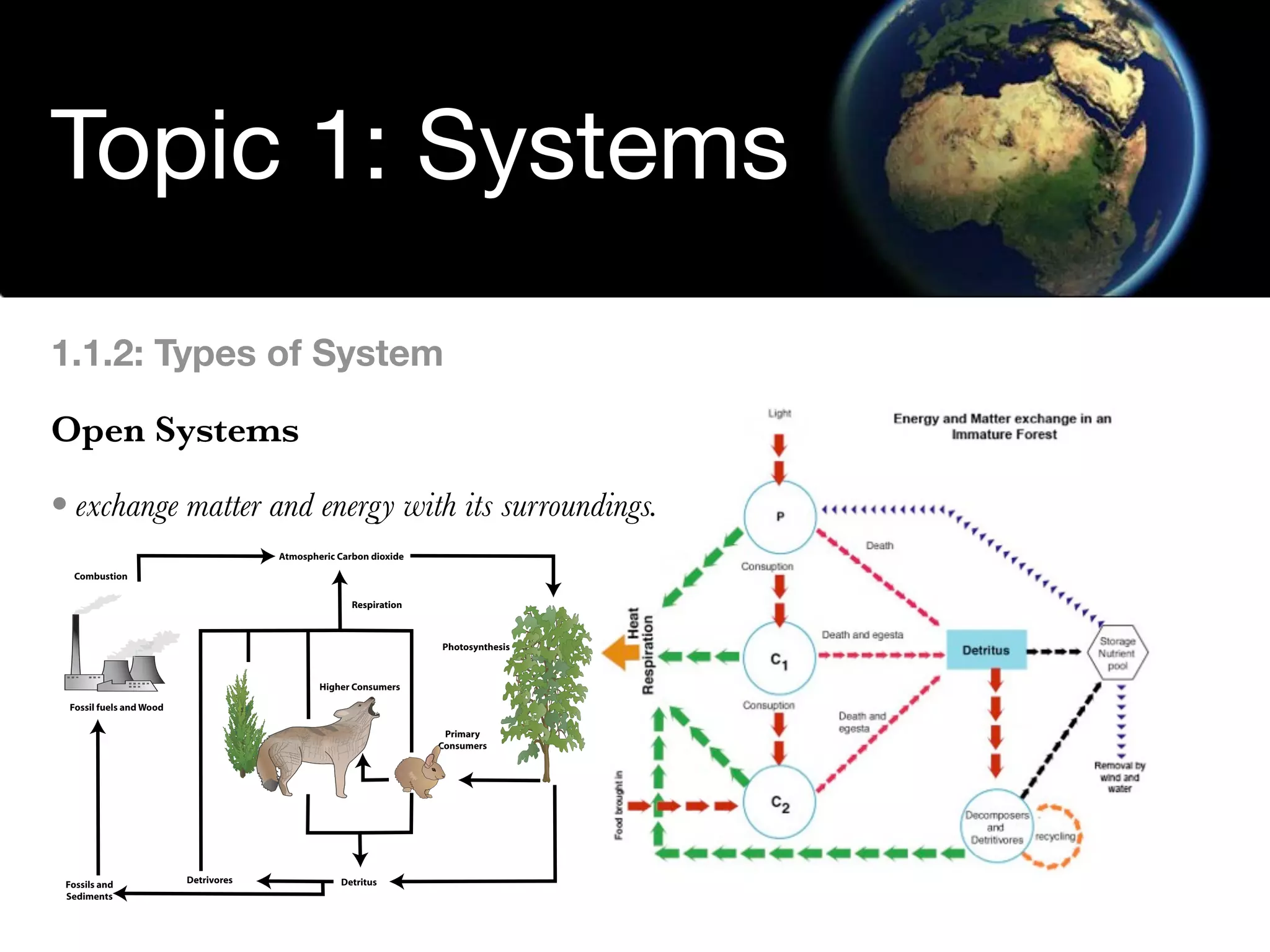




















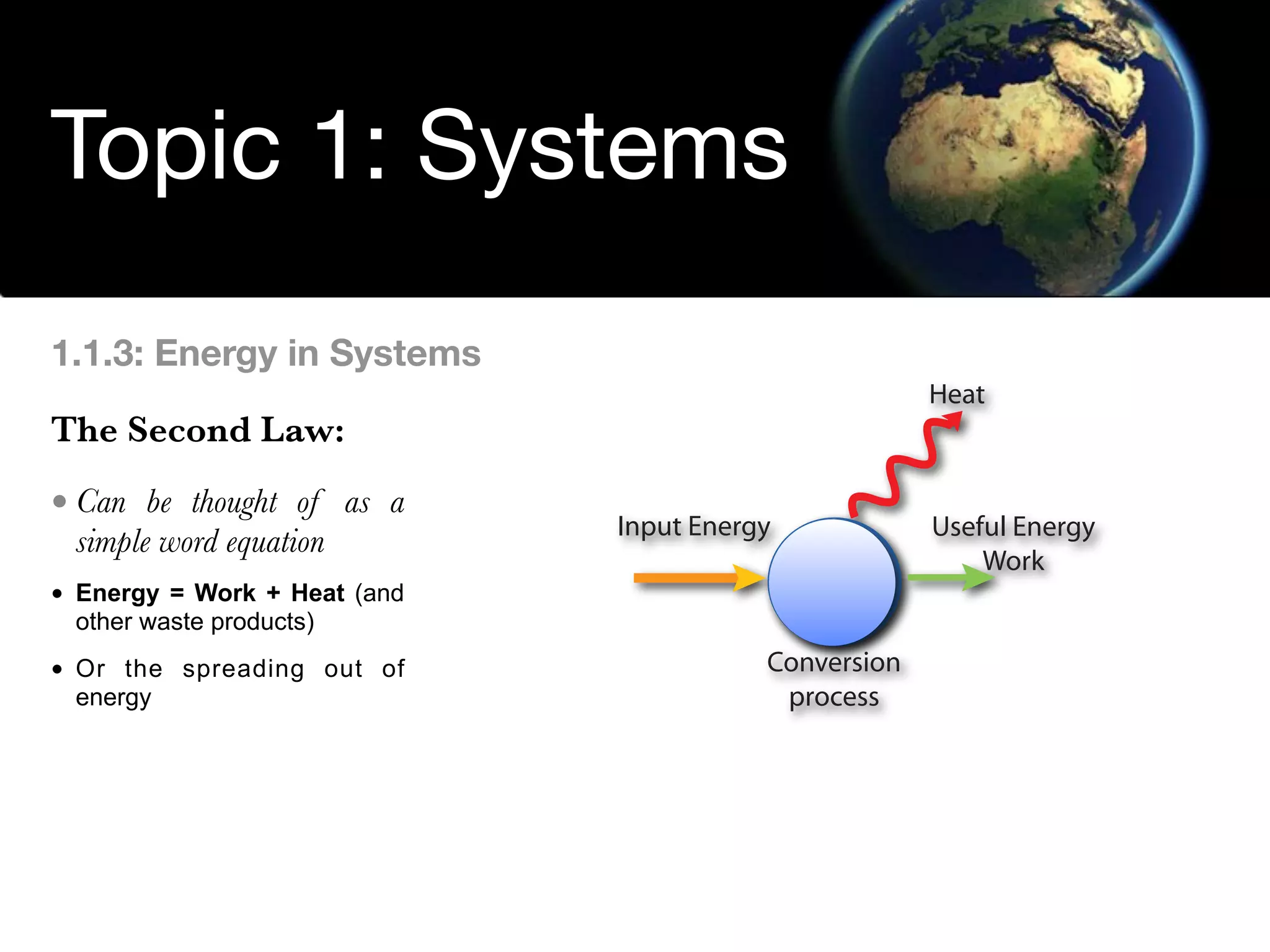



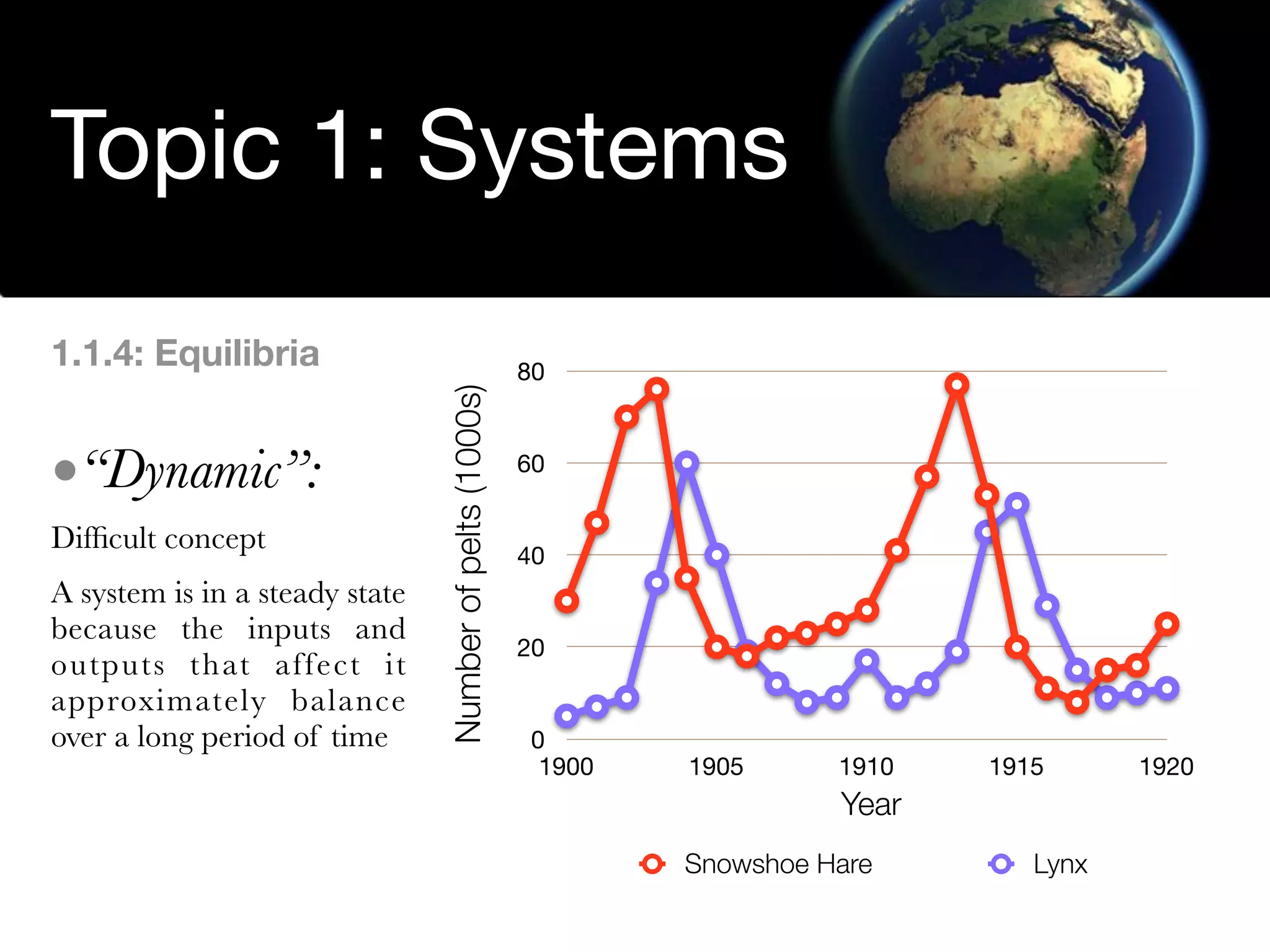

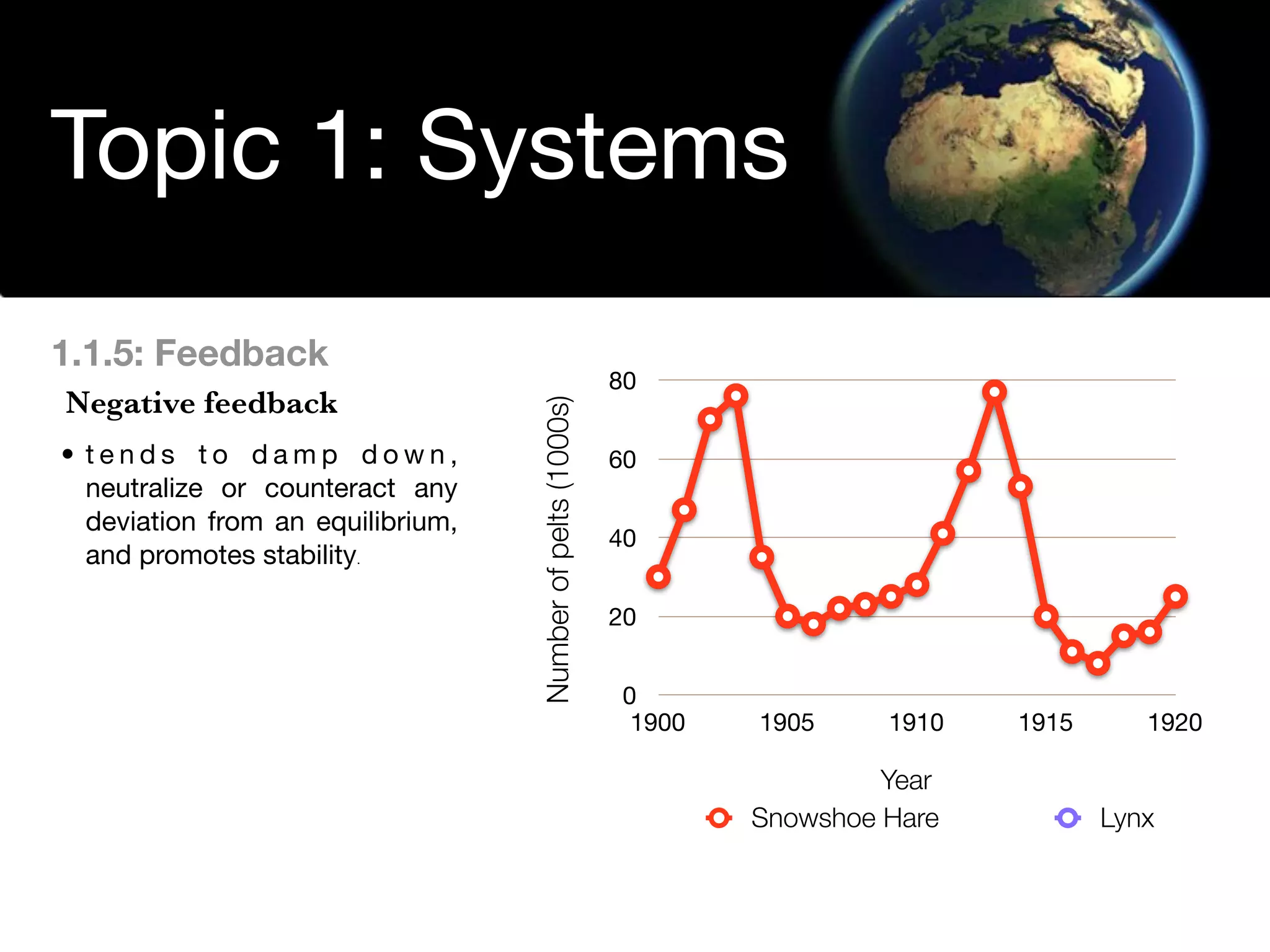
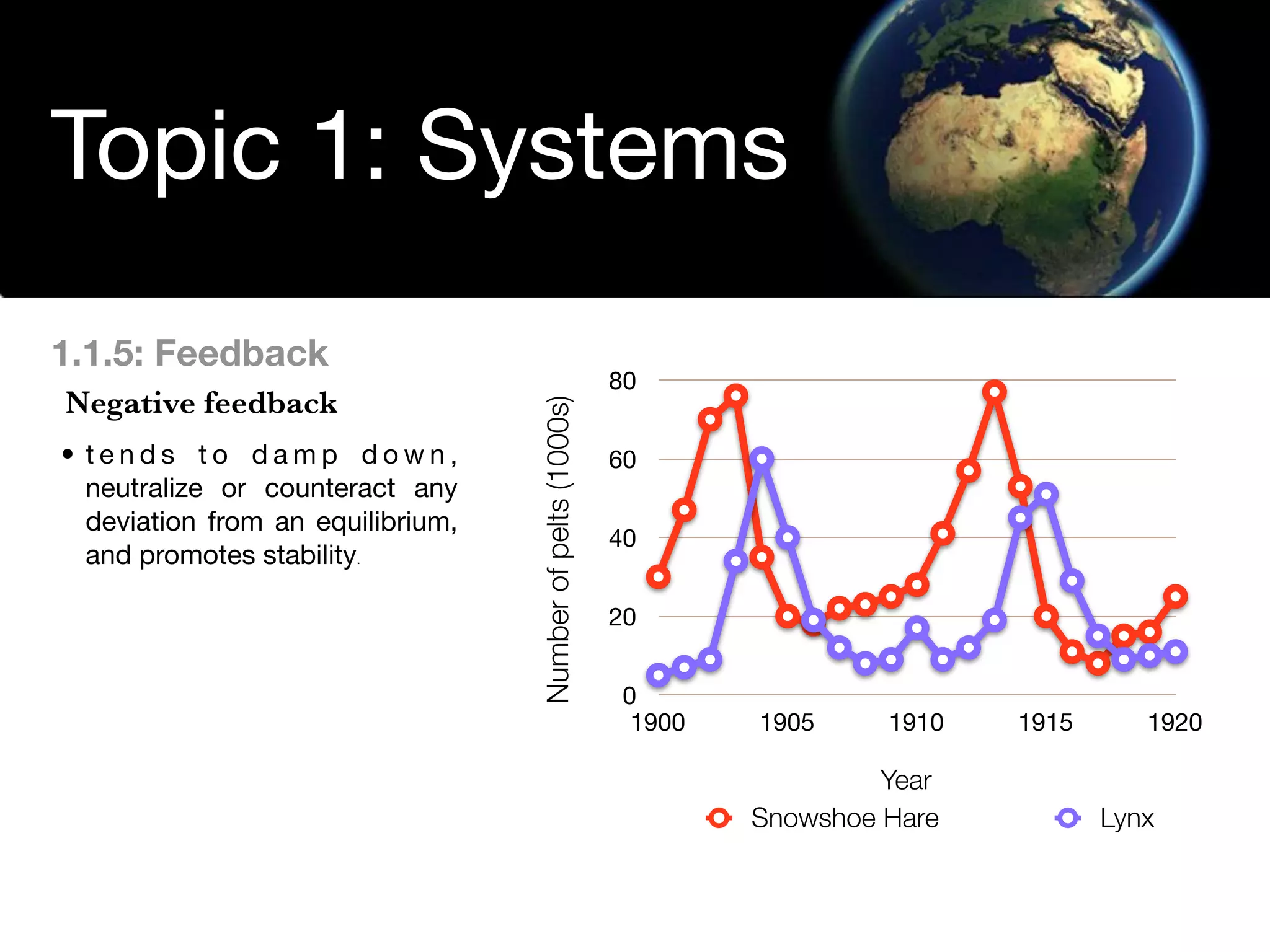
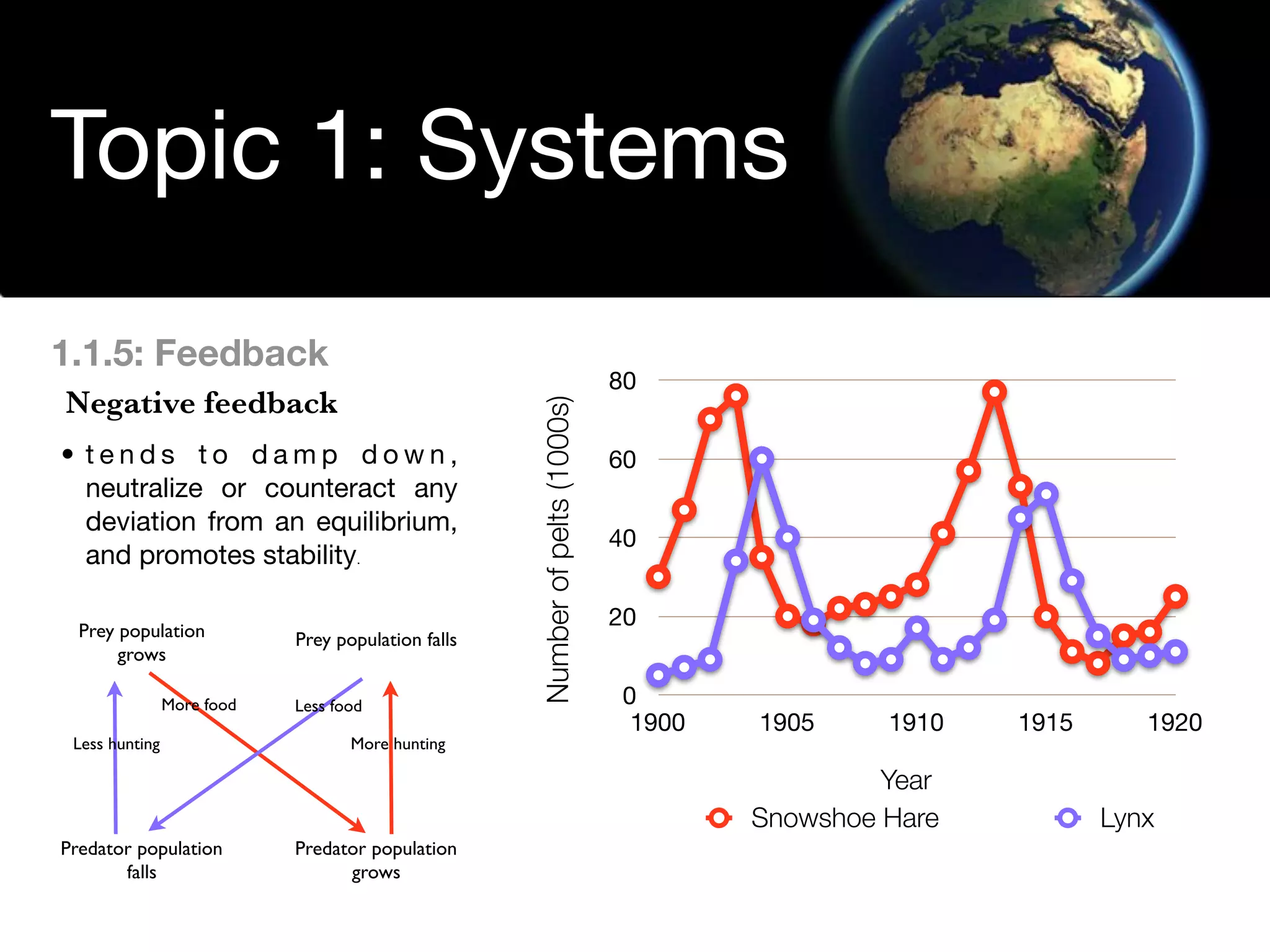





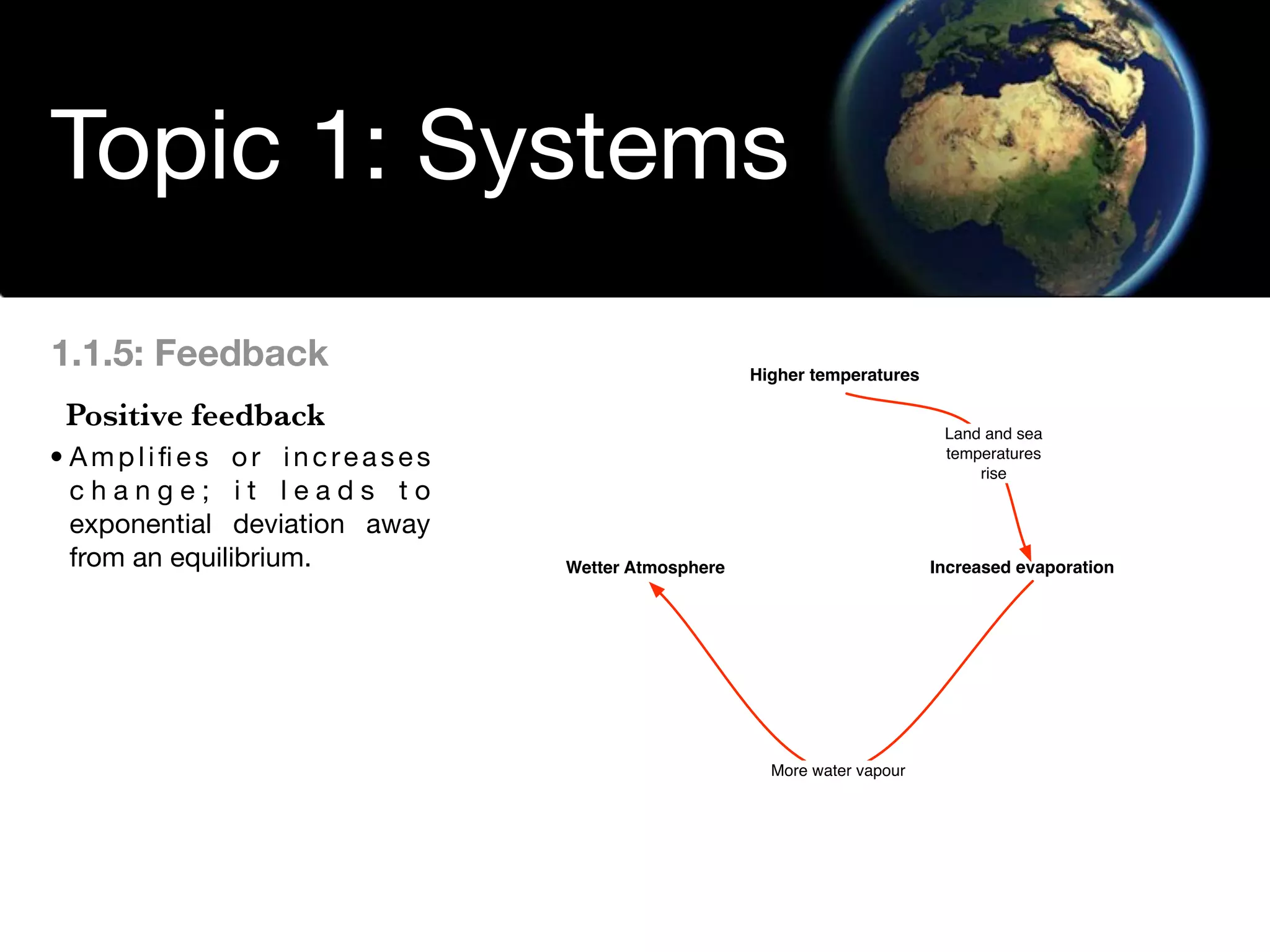
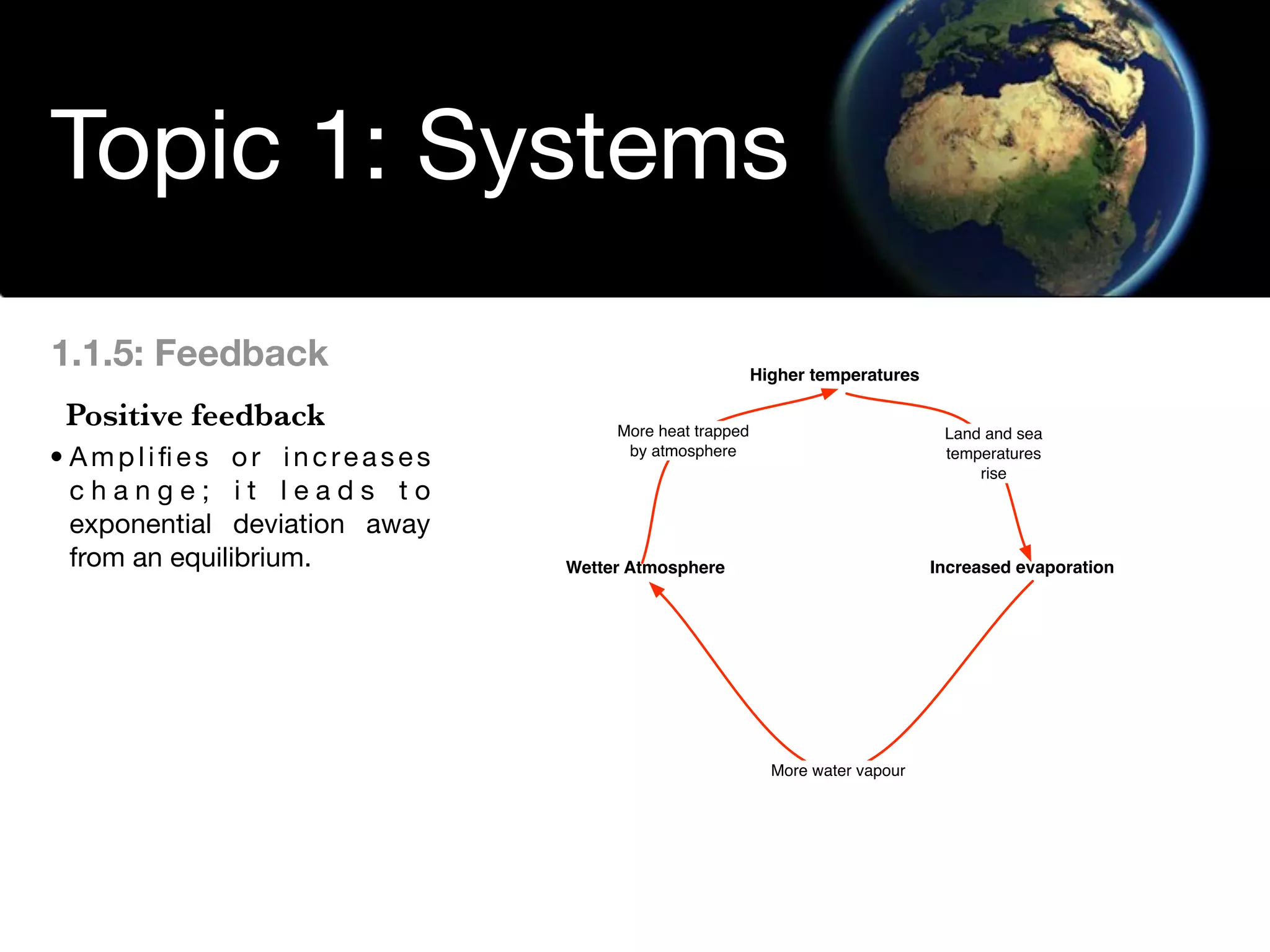
















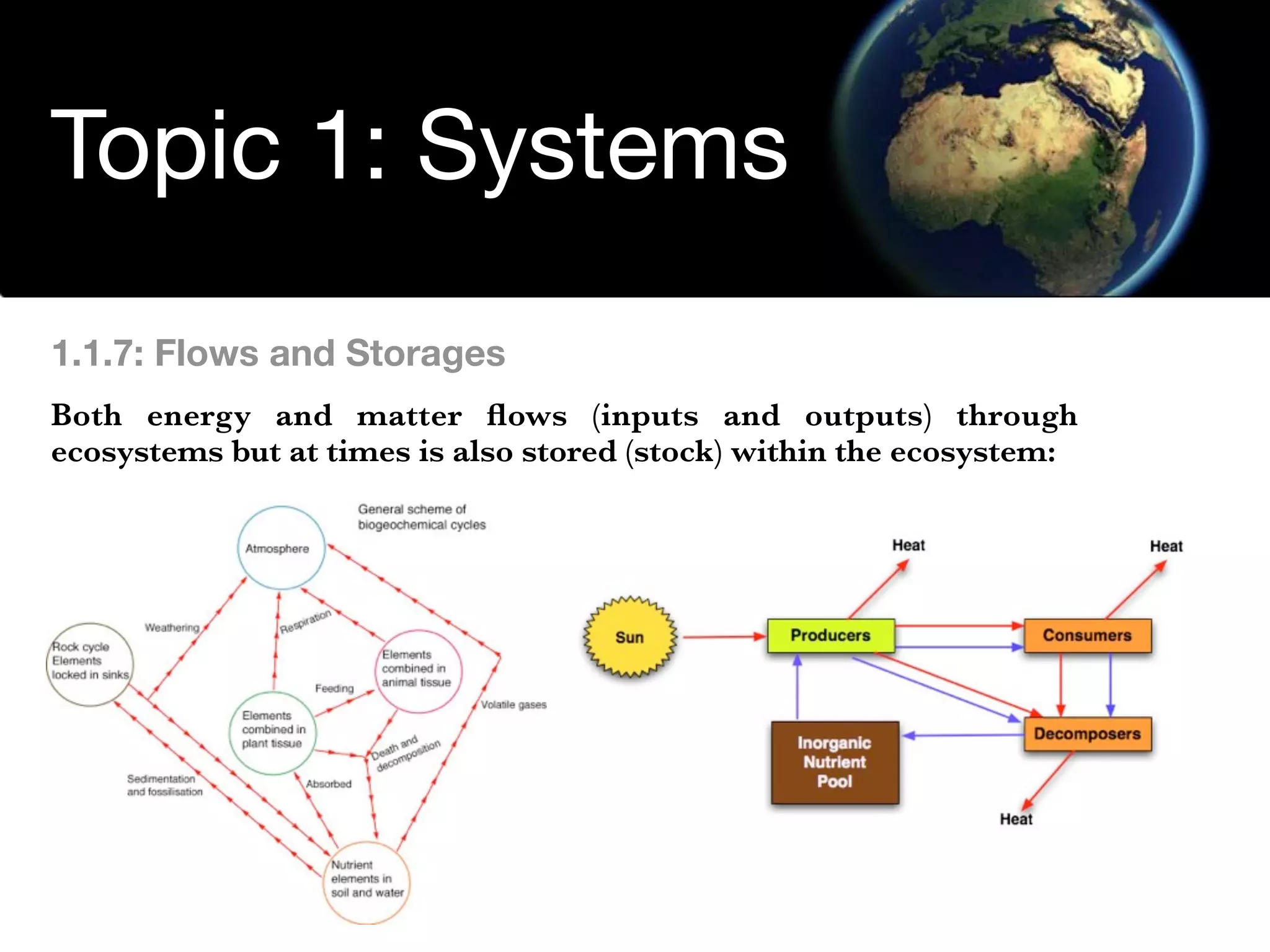

The document discusses the concept of systems, describing their components as inputs, processes, and outputs, and categorizes them into open, closed, and isolated systems. It further explains the laws of thermodynamics as they apply to energy within systems, covering the first and second laws which address energy conservation and efficiency. Lastly, it indicates how feedback loops influence system balance and dynamics, including negative and positive feedback mechanisms.






































































