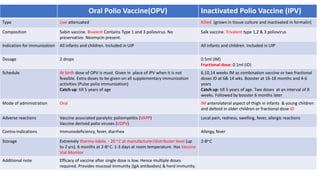
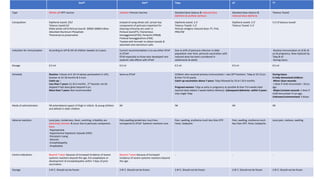
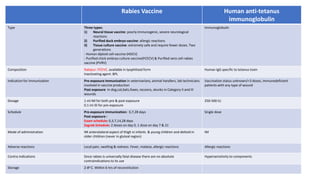
This document summarizes key information about various vaccines and immunization schedules in India. It provides details on vaccines for BCG, oral polio, hepatitis B, Hib, DPT, pneumococcal, hepatitis A, typhoid and rotavirus. For each vaccine, it outlines the type, composition, indications, dosage, schedule, administration method, adverse reactions, contraindications, storage and additional notes. The document is intended to serve as a reference for healthcare professionals on the national immunization schedule and guidelines in India.