Microencapsulation is a technique that involves surrounding tiny droplets or particles of substances with a polymeric material to form microcapsules, which can have various core materials and coating types. The process can provide benefits such as sustained drug release, protection from environmental factors, and improved bioavailability, but also has disadvantages like high costs and potential degradation issues. Different methods including physical, physico-chemical, and chemical techniques are employed to manufacture microcapsules, with applications ranging from drug delivery to food preservation.


![Unit II
Microencapsulation
Akanksha Patel
Asst. prof.
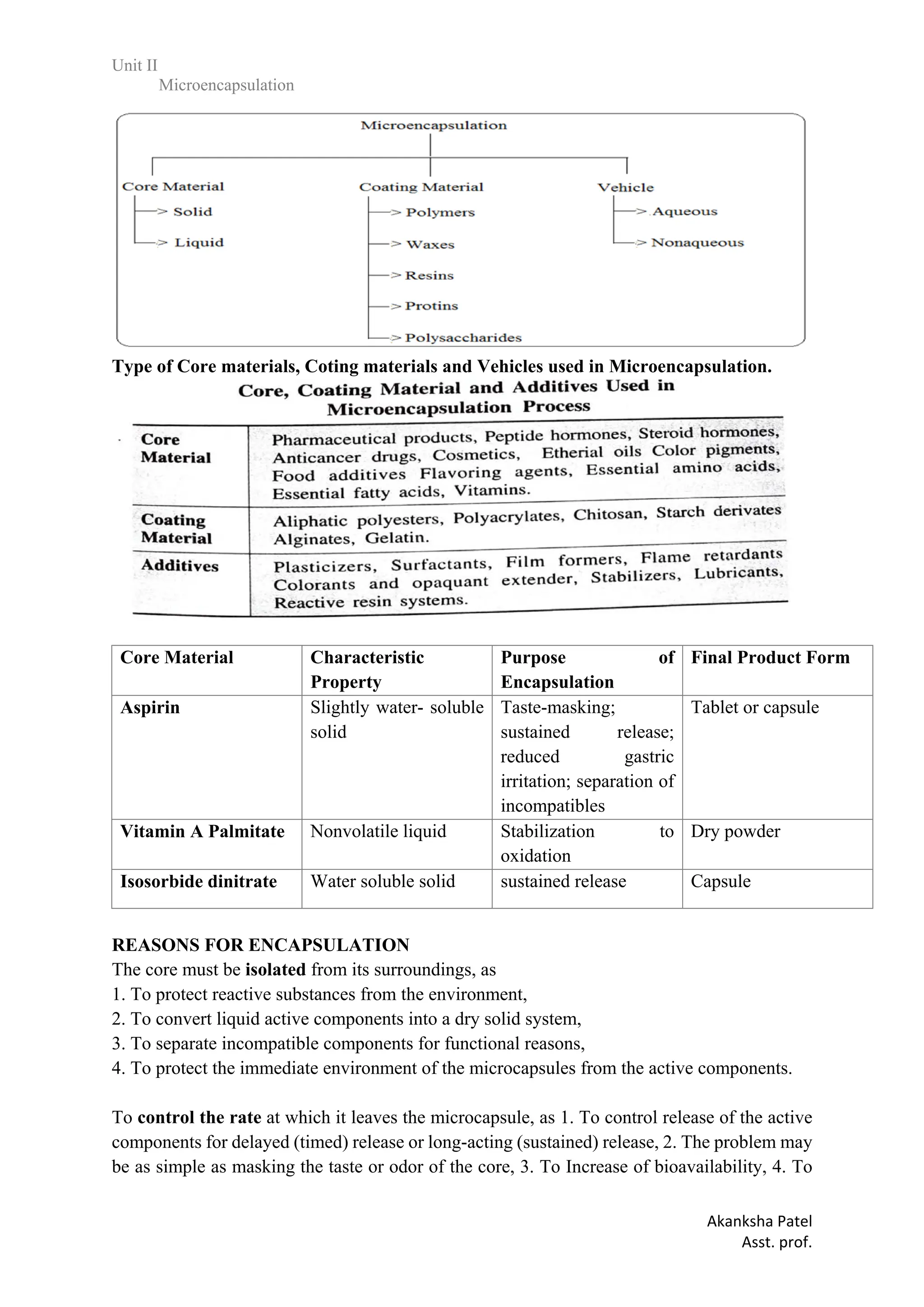
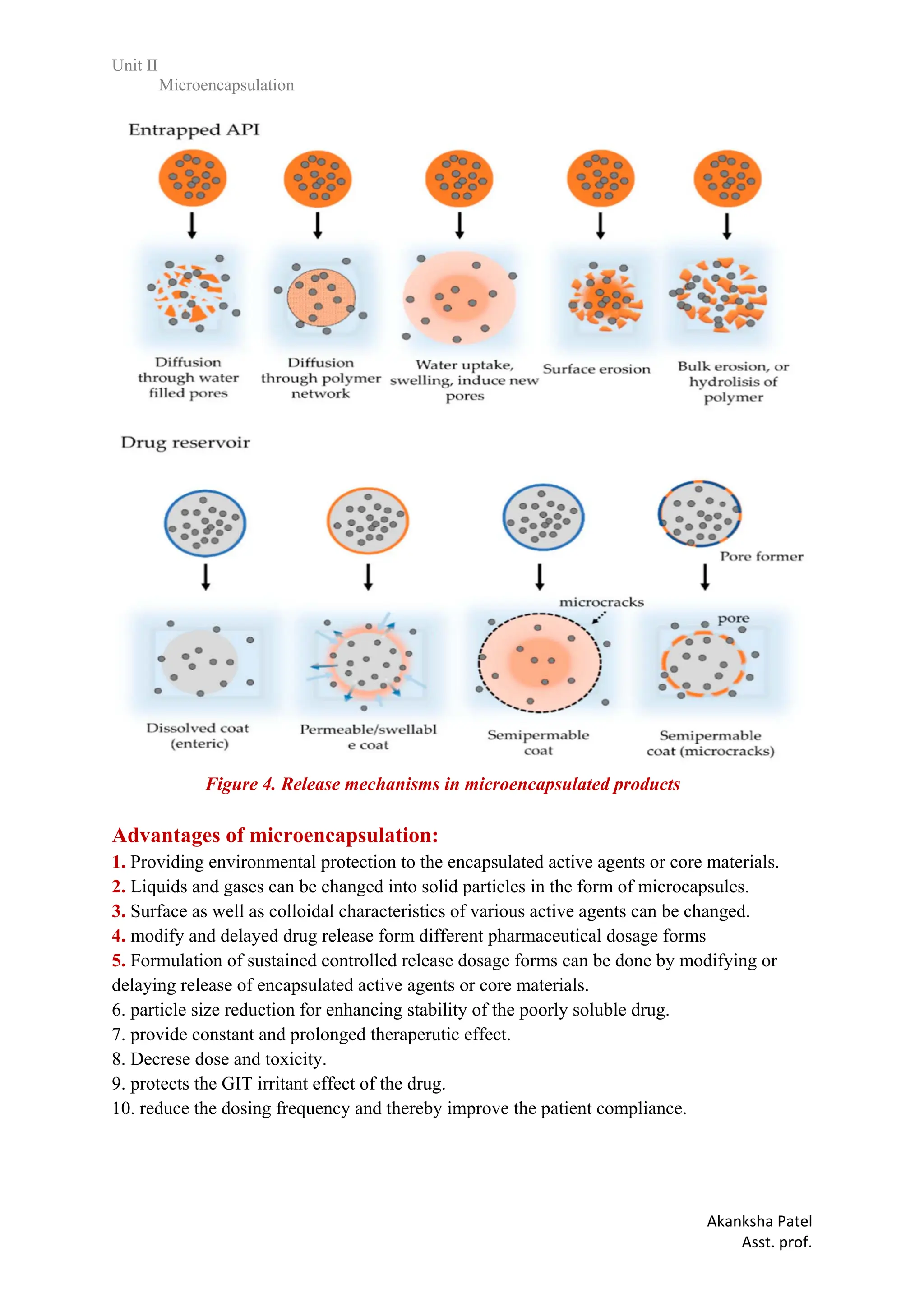
produce a targeted drug delivery, 5. Protects the GIT from irritant effects of the drug, 6.
Extension of duration of activity for an equal level of active.agent. 7. To protect the immediate
environment of the microcapsules from the active components.
Mechanism and kinetics of drug release-
Diffusion
Diffusion is the most commonly involved mechanism wherein the dissolution fluid penetrates
the shell, dissolves the core and leak out through the interstitial channels or pores. Thus, the
overall release depends on, (a) the rate at which dissolution fluid penetrates the wall of
microcapsules, (b) the rate at which drug dissolves in the dissolution fluid, and (c) the rate at
which the dissolved drug leak out and disperse from the surface. The kinetics of such drug
release obeys Higuchi‟s equation as below:
Q = [D/J (2A - ε CS) CS t]1/2
Where, Q is the amount of drug released per unit area of exposed surface in time t; D is the
diffusion coefficient of the solute in the solution; A is the total amount of drug per unit volume;
CS is the solubility of drug in permeating dissolution fluid; ε is the porosity of the wall of
microcapsule; J is the tortuosity of the capillary system in the wall. The above equation can be
simplified to Q = vt where, v is the apparent release rate.
Dissolution
Dissolution rate of polymer coat determines the release rate of drug from the microcapsule
when the coat is soluble in the dissolution fluid. Thickness of coat and its solubility in the
dissolution fluid influence the release rate.
Osmosis
The polymer coat of microcapsule acts as semi permeable membrane and allows the creation
of an osmotic pressure difference between the inside and the outside of the microcapsule and
drives drug solution out of the microcapsule through small pores in the coat.
Erosion
Erosion of coat due to pH and/or enzymatic hydrolysis causes drug release with certain coat
materials like glyceryl monostearate, bees wax and stearyl alcohol.](https://image.slidesharecdn.com/2-240213173542-1720d24a/75/unit-2-various-approaches-on-Microencapsulation-pdf-4-2048.jpg)

![Unit II
Microencapsulation
Akanksha Patel
Asst. prof.
I] Physical or Physico-mechanical methods
22
1.Air-suspension coating- Inventions of Professor Dale E. Wurster,
Basically the wurster process consists of the dispersing of solid, particulate core materials in a
supporting air stream and the spray-coating of the air suspended particles.
Equipment ranging in capacities from one pound to 990 pounds.
Micron or submicron particles can be effectively encapsulated by air suspension techniques.
Disadvantage- Agglomeration of the particles to some larger size is normally achieved.
Processing variables for efficient, effective encapsulation by air suspension techniques:
1.Density, surface area, melting point, solubility, friability, volatility, Crystallinity, and flow-
ability of core the core material.
2.Coating material concentration (or melting point if not a solution).
3.Coating material application rate.
4.Volume of air required to support and fluidizes the core material.
5.Amount of coating material required.
6.Inlet and outlet operating temperatures.](https://image.slidesharecdn.com/2-240213173542-1720d24a/75/unit-2-various-approaches-on-Microencapsulation-pdf-7-2048.jpg)


![Unit II
Microencapsulation
Akanksha Patel
Asst. prof.
5. Vibrational Nozzle-
The process works very well for generating droplets between 100–5,000 μm
Units are deployed in industries and research mostly with capacities of 1–10,000 kg
per hour at working temperatures of 20–1500 °C.
Nozzles heads are available from one up to several hundred thousand are available.
II] Physico-chemical methods 39
1.Ionotropic gelation-
In this technique, the preparation of drug containing microparticles is based on the
principle of coalescence of colloidal polymer particles. Ionotropic gelation of the
anionic polysaccharide sodium alginate with oppositely charged calcium ions forms
microparticles.
Verapamil hydrochloride causes gastric irritation on sudden release. It is usually
administered as conventional tablets containing 40-120 mg, 3 times a day. Due to its
ready solubility in water and shorter half-life. Microparticulate system of verapamil
hydrochloride is prepared for prolonged release delivery system.](https://image.slidesharecdn.com/2-240213173542-1720d24a/75/unit-2-various-approaches-on-Microencapsulation-pdf-11-2048.jpg)
![Unit II
Microencapsulation
Akanksha Patel
Asst. prof.
2. Coacervation-Phase Separation
The term coacervation is derived from Latin word acervus, which means a heap or aggregation.
The term coacervation was introduced in 1929 by Bungenberg de Jong and Kruyt, for a process
of phase separation in liquids containing colloidal solute.
Simple coacervation generally uses one colloid, which is precipitated out by salts or non
solvents or by increase or decrease in temperature.
Complex coacervation involves two or more than two colloids and salting out is carried out
by oppositely charged polymers, for example, Acaia and Gelatin
3 steps under continues agitation-
1) Formation of three immiscible chemical phases (Liquid phase, coating and core material
phase) 2)Deposition of the coating 3)Rigidization of the coating.
•Formation of three immiscible chemical Phases (Phase seperation)
• Temperature Change
•Incompatible Polymer addition
•Non Solvent addition
•Salt addition
•Polymer Polymer interaction
Schematic representation of the coacervation process.
(a) Core material dispersion in solution of shell polymer;
(b) separation of coacervate from solution;
(c) coating of core material by microdroplets of coacervate;
(d) coalescence of coacervate to form continuous shell around core particles.
III] Chemical process 45
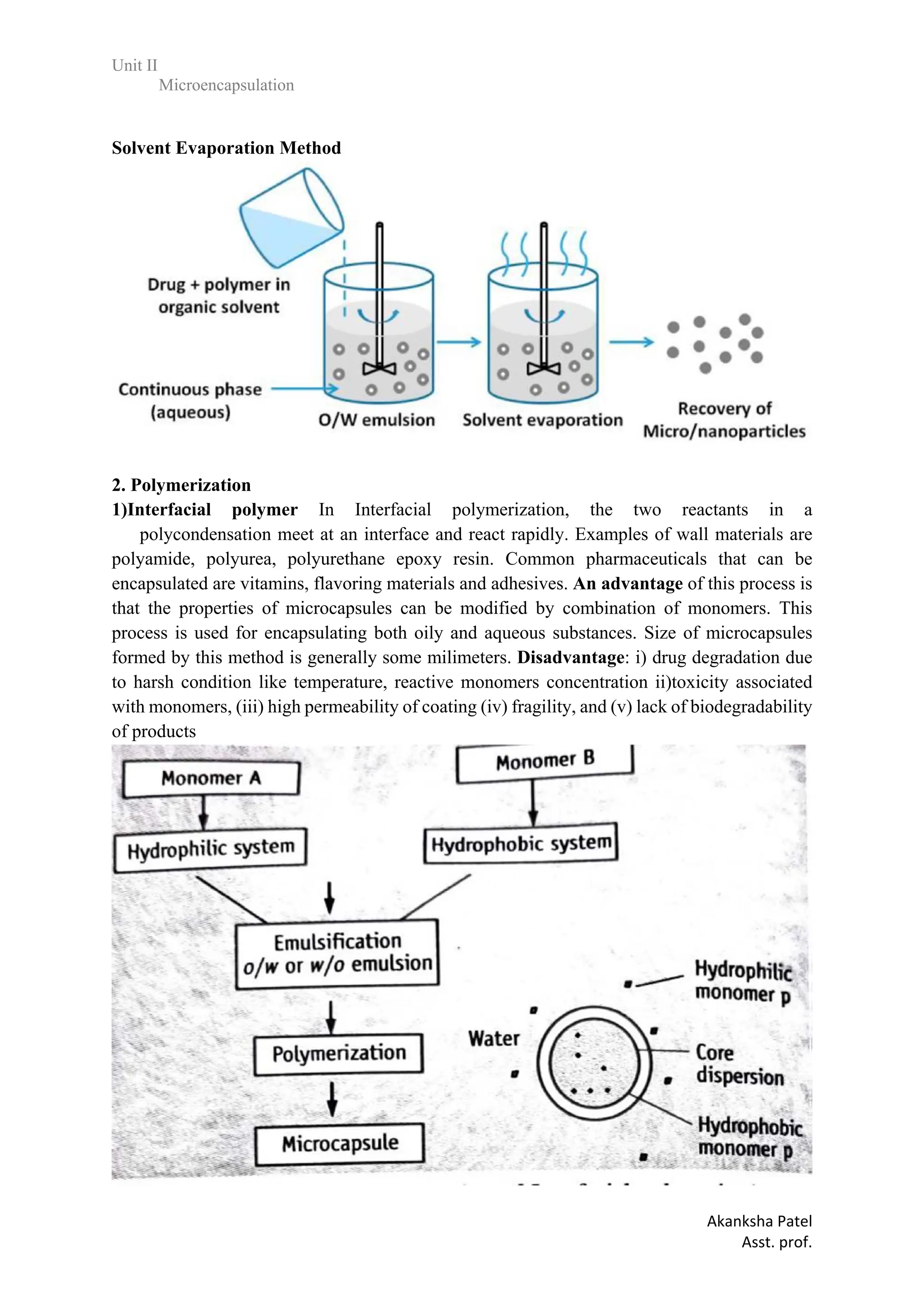
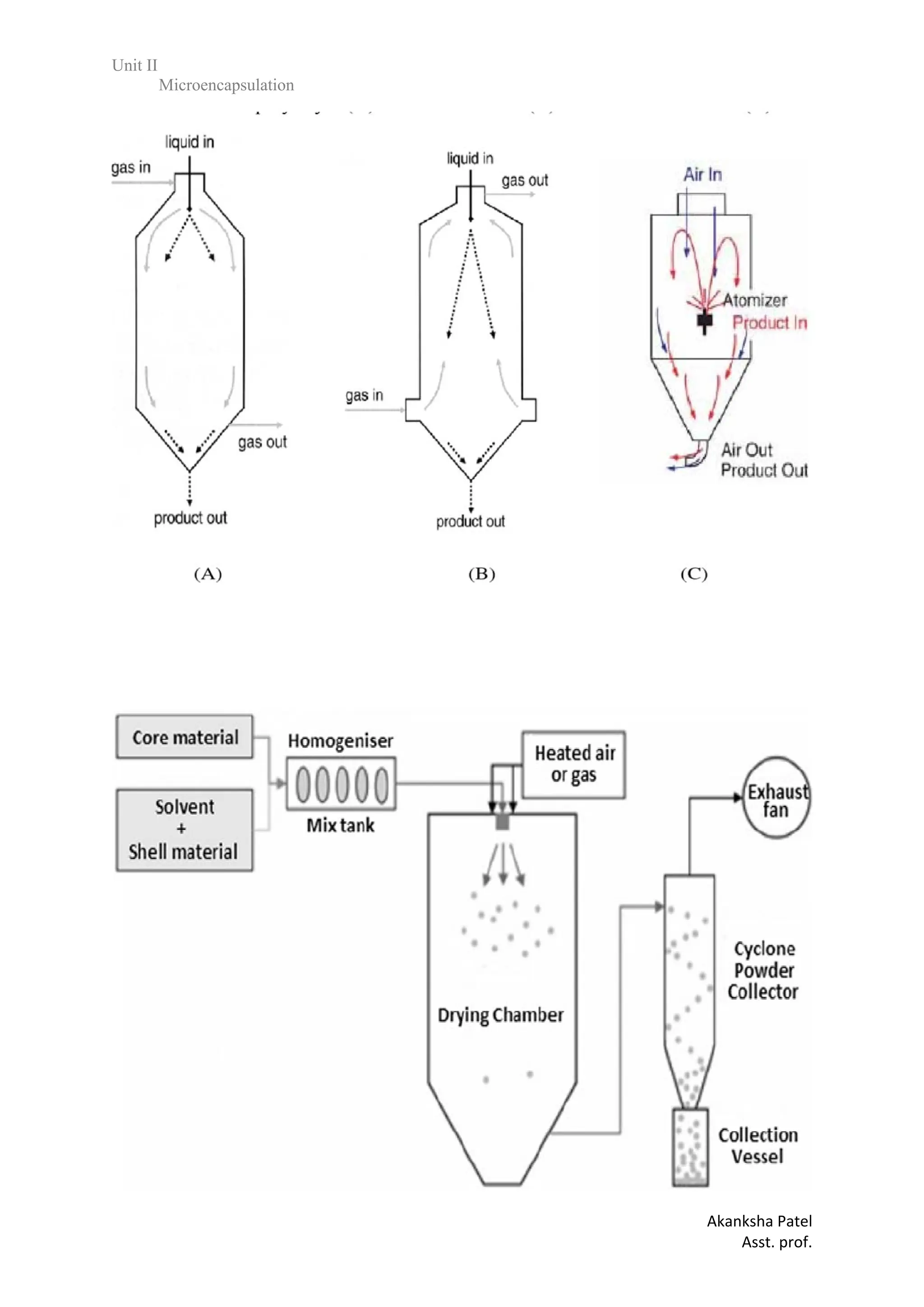
1.Solvent Evaporation
In the case in which the core material is dispersed in the polymer solution, polymer shrinks
around the core. In the case in which core material is dissolved in the coating polymer solution,
a matrix - type microcapsule is formed.
The core materials may be either water - soluble or water - insoluble materials.
A variety of film - forming polymers can be used as coatings.](https://image.slidesharecdn.com/2-240213173542-1720d24a/75/unit-2-various-approaches-on-Microencapsulation-pdf-12-2048.jpg)