Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering Effective Care With BCMA Antibodies and Cellular Therapy
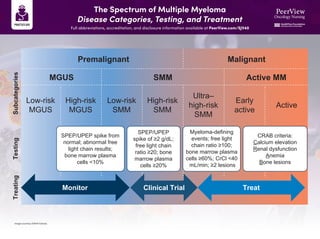
- 1. The Spectrum of Multiple Myeloma Disease Categories, Testing, and Treatment Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 Image courtesy of Beth Faiman. Premalignant Malignant MGUS SMM Active MM Low-risk MGUS High-risk MGUS Low-risk SMM High-risk SMM Ultra– high-risk SMM Early active Active Treating Testing Subcategories SPEP/UPEP spike from normal; abnormal free light chain results; bone marrow plasma cells <10% SPEP/UPEP spike of ≥2 g/dL; free light chain ratio ≥20; bone marrow plasma cells ≥20% Myeloma-defining events; free light chain ratio ≥100; bone marrow plasma cells ≥60%; CrCl <40 mL/min; ≥2 lesions CRAB criteria: Calcium elevation Renal dysfunction Anemia Bone lesions Monitor Clinical Trial Treat
- 2. Nursing Tools, Strategies, and Take-Homes for Multiple Myeloma Patient Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 Bone health changes • Monitor for changes in bone health • Supplement with vitamin D and calcium • Determine if bone-modifying drugs are needed • Schedule regular dental examinations if patients are on bone-modifying therapies • Determine if patients are at high risk for infection (eg, patients with hypogammaglobulinemia) • Monitor for neutropenia and myelosuppression • Administer shingles prophylaxis when treating with proteasome inhibitors, monoclonal antibodies, or cellular therapies • Administer IVIG for recurrent infections • Assess for diarrhea when treating with bortezomib and lenalidomide • Assess for constipation when treating with thalidomide and doxorubicin • Administer an antiemetic prior to treating with carfilzomib, selinexor, doxorubicin, or proteasome inhibitors • Monitor for PN when treating with bortezomib and thalidomide; promptly intervene to prevent irreversible PN • Avoid renal-toxic agents • Administer 24-hour urine albumin (bone-modifying drugs) • Dose reduce (lenalidomide, melphalan, opioids, acyclovir) • Recommend healthy eating, moderating alcohol intake, and exercising • Remind patients to keep checkup appointments • Recommend patients stop smoking • Recommend regular eye exams, especially if patients are on corticosteroids (because of the risk of cataracts) or belantamab • Communicate with and educate patients on: financial expectations, psychosocial issues, treatment options, adherence to appointments, drugs, physical activity, and patient resources • Perform monthly disease assessments and imaging as indicated Infectious diseases GI toxicities Neurologic effects Renal impairment Disease monitoring Health maintenance Survivorship care General Nursing Considerations to Address for All Patients1
- 3. Nursing Tools, Strategies, and Take-Homes for Multiple Myeloma Patient Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 Predictor Acronym Score Immunomodulatory drug Body mass index ≥25 kg/m2 Pelvic, hip, or femur fracture Erythropoiesis-stimulating agent Doxorubicin Dexamethasone High dose Low dose 4 2 Ethnicity/race of Asian/Pacific Islander History of VTE before MM Tunneled line/central venous catheter Existing thromboprophylaxis: therapeutic LMWH or warfarin I M P E D 4 1 4 1 3 E V T E -3 5 2 -4 Existing thromboprophylaxis: prophylactic LMWH or aspirin -3 IMPEDE VTE Risk Scoring Can Be Useful Prior to Deciding On Therapy2 VTE Risk Scores <3: low risk 3-6: intermediate risk >6: high risk 1. NCCN Clinical Practice Guidelines in Oncology. Multiple Myeloma. Version 3.2023. https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf. 2. Covut F et al. Br J Haematol. 2021;193:1213-1219.
- 4. Take-Homes on BCMA CAR-T Constructs in Multiple Myeloma Treatment Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 1. Carvykti (ciltacabtagene autoleucel) Prescribing Information. https://www.fda.gov/media/156560/download. 2. Abecma (idecabtagene vicleucel) Prescribing Information. https://www.fda.gov/media/147055/download. REMS Cilta-Cel1 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD Recommended Dose Range 0.5-1.0 × 106 CAR-positive viable T cells (maximum dose of 1 × 108 CAR-positive viable T cells per single-dose infusion) Recommended Dose Range 300-460 × 106 CAR-positive viable T cells Ide-Cel2 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD General Principles for CAR-T Therapy CAR-T Cell Therapy: Indications and Dosing Referral to a certified healthcare facility is required for collection of patient’s cells and administration of CAR-T therapy Avoid prophylactic use of dexamethasone or other systemic corticosteroids Premedicate with acetaminophen and an H1 antihistamine Monitor for CRS and ICANS and confirm tocilizumab availability before infusion Ide-cel and cilta-cel are available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) Monitor for neurologic events, hemophagocytic lymphohistiocytosis/ macrophage activation syndrome, and cytopenias Administer a lymphodepleting chemotherapy regimen of cyclophosphamide and fludarabine before CAR-T infusion Do not use a leukodepleting filter when administering
- 5. Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 Regulatory Status Dosing Select Considerations Phase 2 study in patients with RRMM and no prior BCMA- targeted treatment2 Patients received 76 mg weekly with a two-step-up priming regimen per the MagnetisMM-3 trial3 The most common AEs were hematologic events and CRS; the two-step-up priming regimen is intended to help mitigate the rate and severity of CRS3 Elranatamab BCMA x CD3 Phase 1 study in patients with RRMM In early-phase testing, dose escalation was 0.025-120 mg and dose expansion was 60 mg The most common TEAEs were neutropenia, anemia, CRS, and fatigue ABBV-3831 BCMA x CD3 Therapy/Target Phase 1/2 study in patients with RRMM4 200 mg5 The most common grade 3/4 TEAEs were hematologic; the most frequent TEAEs were fatigue and CRS (most CRS events were grade 1)4 Linvoseltamab BCMA x CD3
- 6. Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 a Currently available under accelerated approval. 1. D'Souza A et al. J Clin Oncol. 2022;40:3576-3586. 2. https://clinicaltrials.gov/ct2/show/NCT04649359. 3. Lesokhin A et al. ASCO 2022. Abstract 8006. 4. Zonder JA et al. EHA 2022. Abstract S189. 5. Bumma N et al. ASH 2022. Abstract 4555. 6. Tecvayli (teclistamab-cqyv) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761291s000lbl.pdf. 7. Moreau P et al. ASH 2021. Abstract 896. 8. Wong SW et al. ASH 2022. Abstract 162. Although the BCMA antibody–drug conjugate belamaf received FDA Accelerated Approval in 2020, the manufacturer has initiated the process for withdrawal of the US marketing authorization based on findings from the DREAMM-3 phase 3 confirmatory trial Other bispecific BCMA and non-BCMA agents are also in rapid development, including TNB383b, talquetamab, cevostamab, and HPN 217 Other new developments: Alnuctamab is an emerging BCMA x CD3 antibody that has demonstrated clinical activity in patients with RRMM who have been treated with ≥3 prior lines of therapy8 • Approved in RRMM in patients who have received ≥4 lines of prior therapy, including an IMiD, a proteasome inhibitor, and an anti-CD38 antibody (FDA); also approved by the European Medicines Agency(see PI for more information) • Day 1: 0.06 mg/kg SC injection • Day 4: 0.3 mg/kg • Day 7: 1.5 mg/kg • Followed by 1.5 mg/ kg once weekly until PD or unacceptable toxicity The most common AEs were hematologic events and CRS. Note: ICANS events occurred in 4 patients but were all grade 1/2 and resolved without discontinuation7 Teclistamab6,a BCMA x CD3 Regulatory Status Dosing Select Considerations Therapy/Target
- 7. Resources for Encouraging Patient Engagement in Care From HealthTree Foundation for Multiple Myeloma Full abbreviations, accreditation, and disclosure information available at PeerView.com/SJV40 HealthTree Foundation for Multiple Myeloma offers a wide range of resources that can be used to encourage patients to learn more about their disease or a planned course of treatment Patient education and navigation tools Programs that create patient connections and build community A living, real-world evidence data portal that delivers researchers the data they need to accelerate a cure Scan to visit: healthtree.org/myeloma Scan to visit: healthtree.org/myeloma/coach Free one-on-one help from experienced patients and caregivers Help patients get answers to basic questions about myeloma Scan to visit: healthtree.org/myeloma/university Selected Resources
