1) There are two main types of intravenous (IV) fluids - crystalloids and colloids. Crystalloids include normal saline, lactated Ringer's, and dextrose solutions. Colloids include albumin and starch solutions.
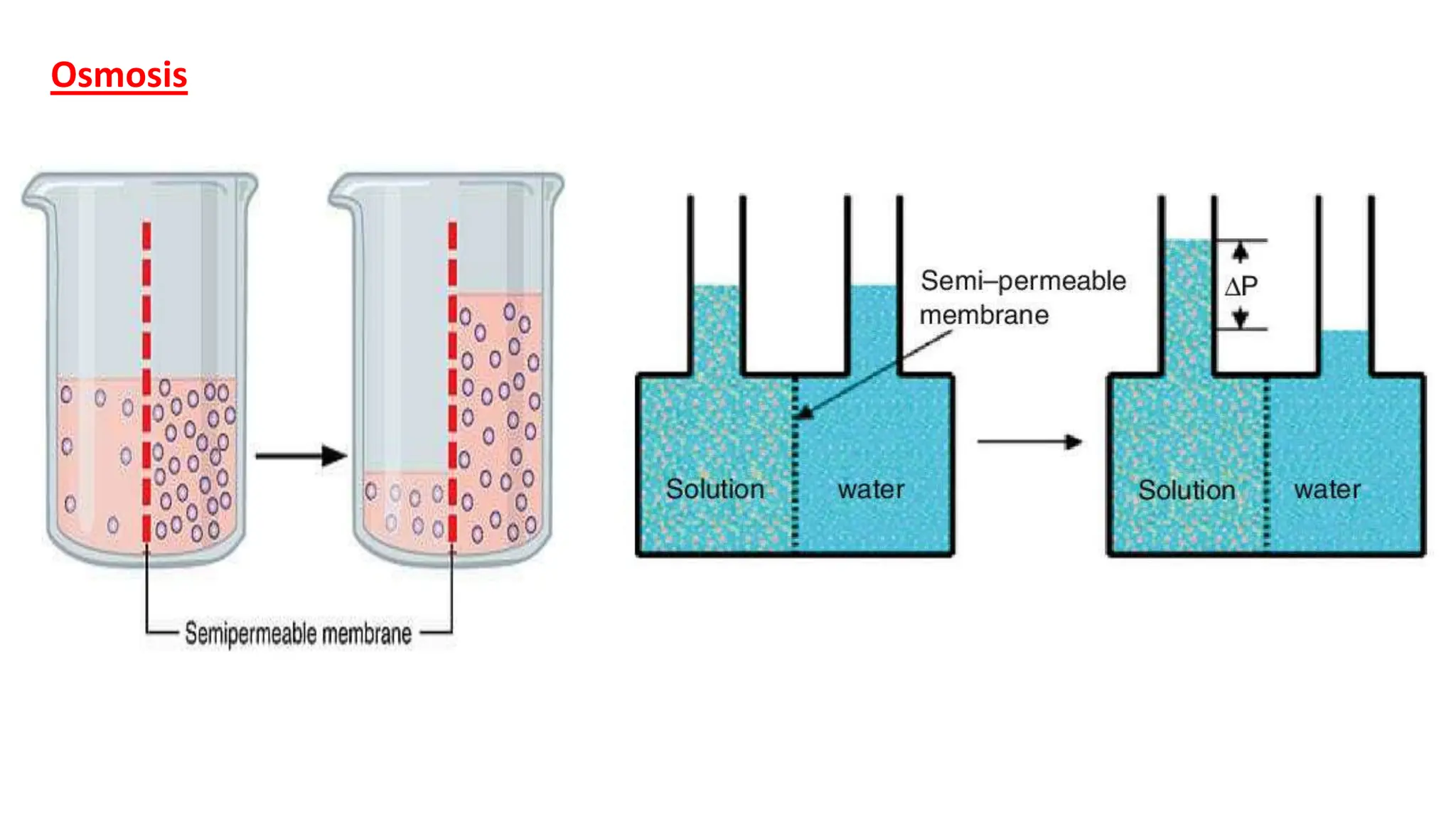
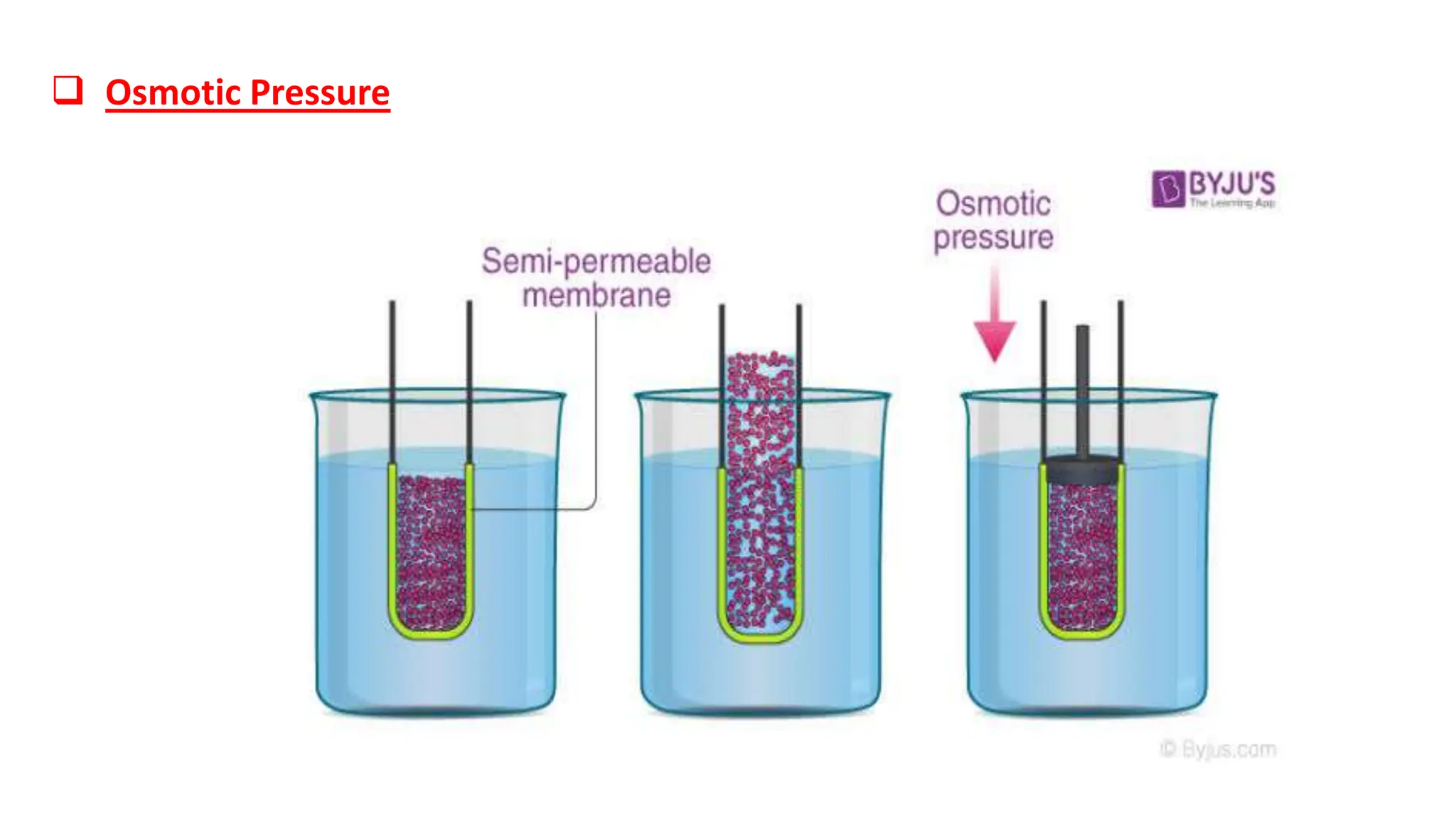
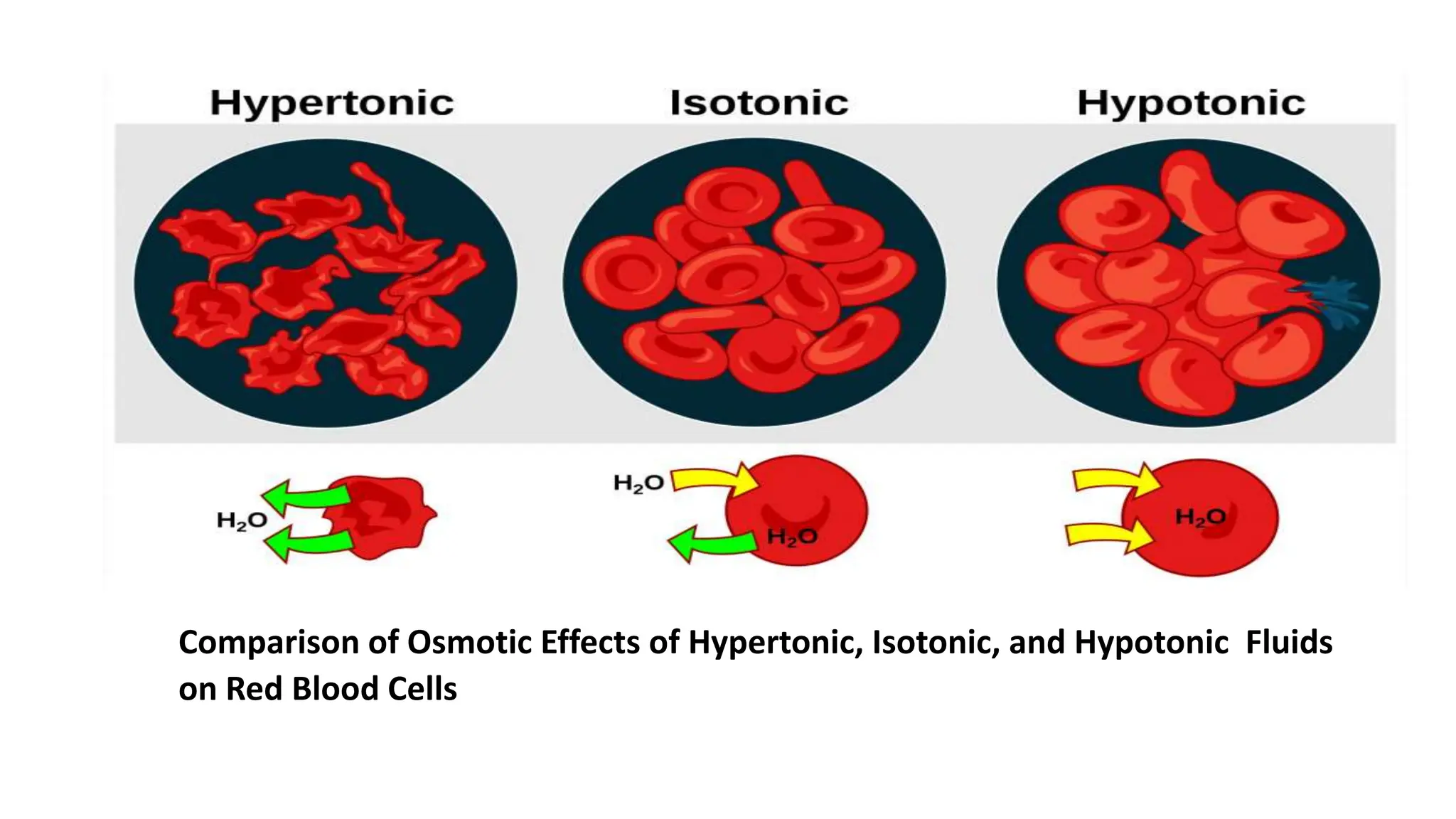
2) IV fluids can be isotonic, hypotonic, or hypertonic depending on their solute concentration compared to cells. Isotonic fluids like normal saline do not cause water movement between cells. Hypotonic fluids cause water to move into cells, and are used to treat conditions like hypernatremia. Hypertonic fluids cause water to move out of cells.
3) Common isotonic crystalloid solutions include normal