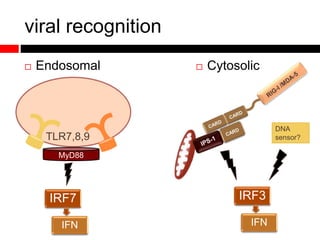
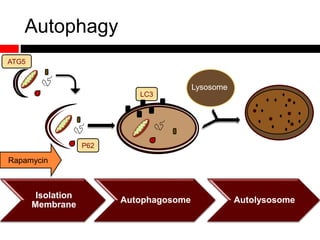
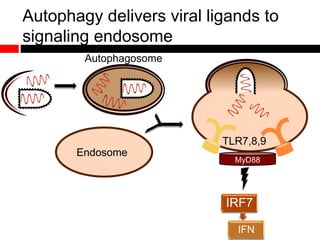
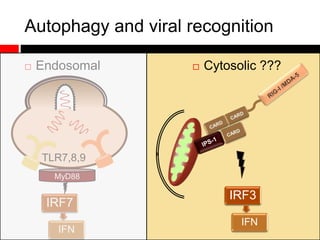
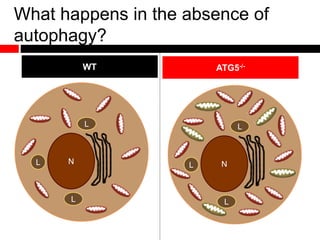
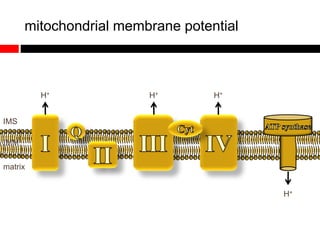
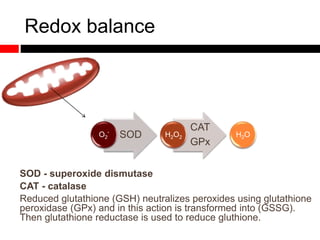
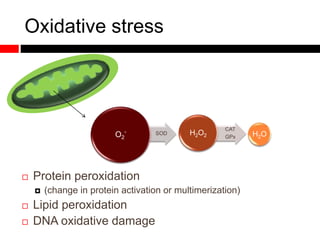
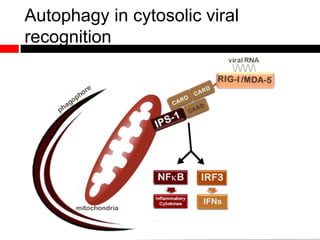
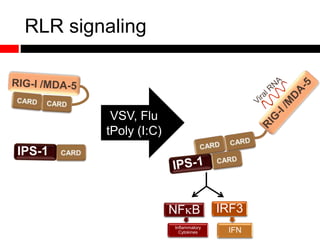
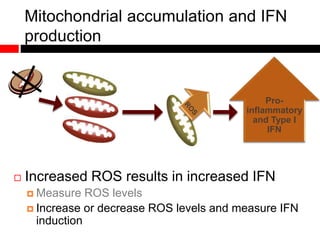
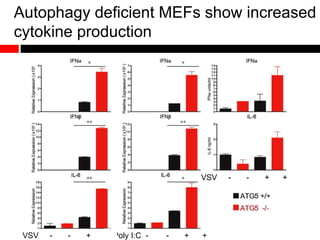
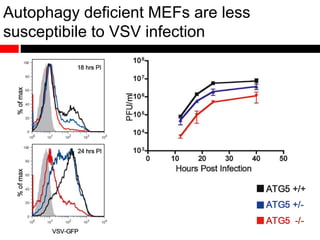
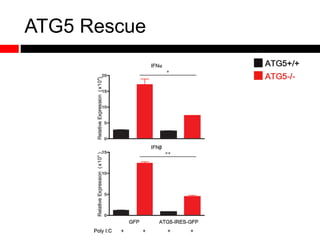
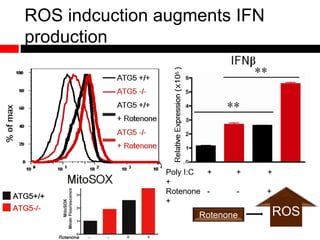
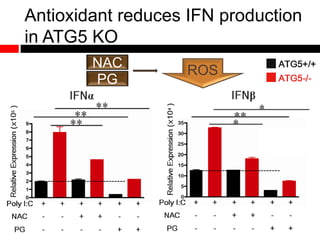
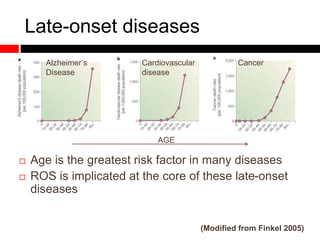
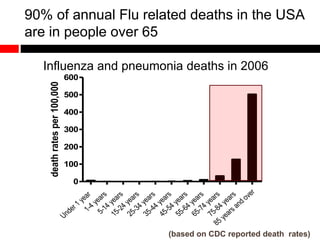
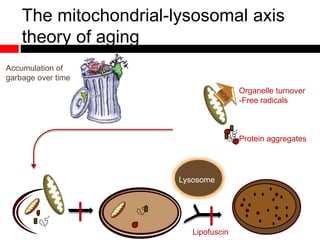
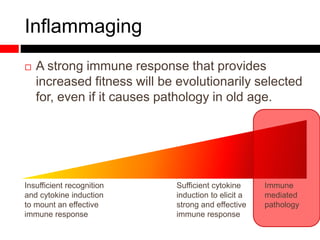
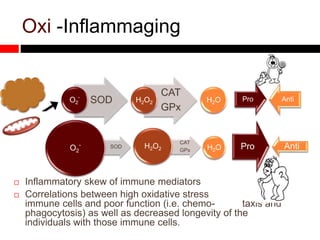
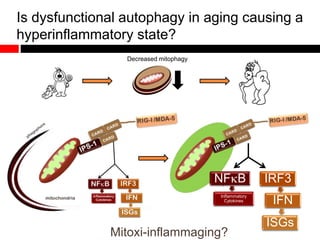
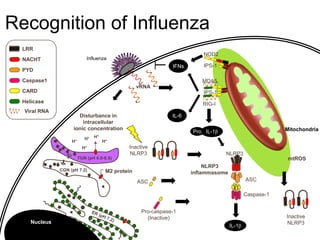
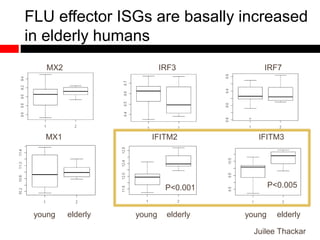
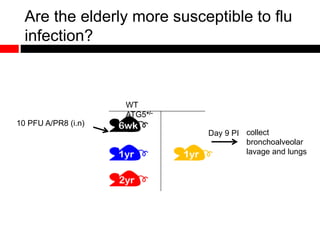
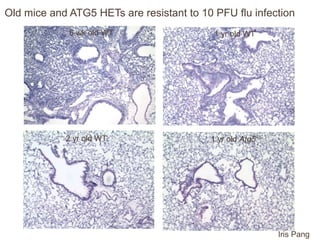
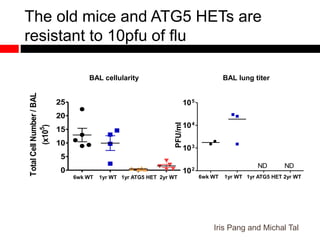
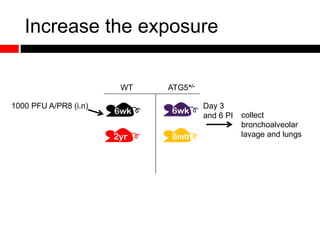
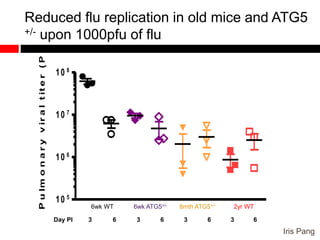
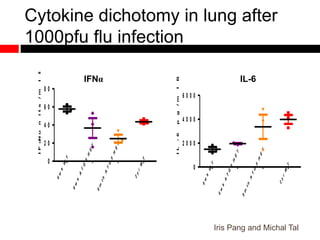
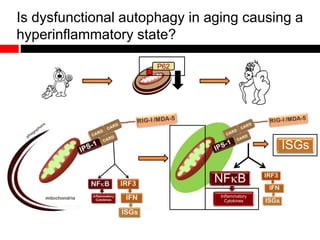
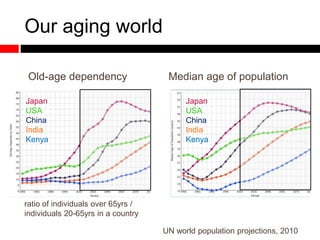
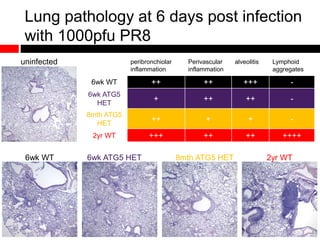
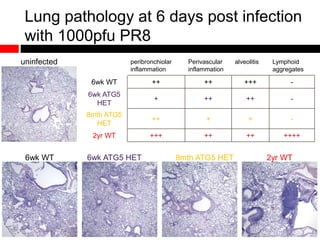
Mitochondrial accumulation and increased ROS levels in the absence of autophagy modulates cytosolic antiviral signaling. Decreased autophagy with age impacts ROS levels and antiviral signaling. Specifically:
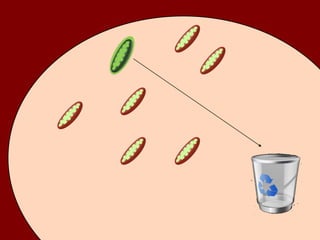
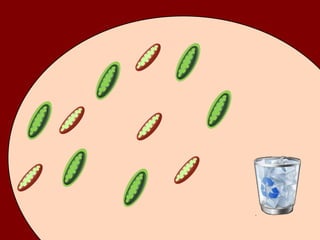
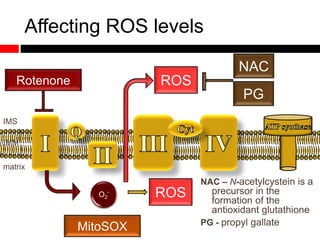
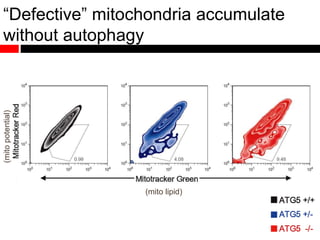
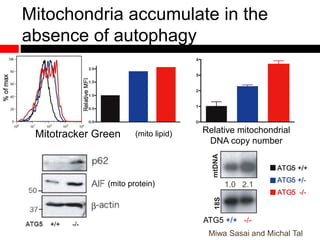
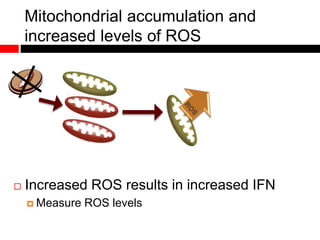
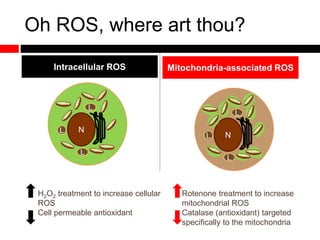
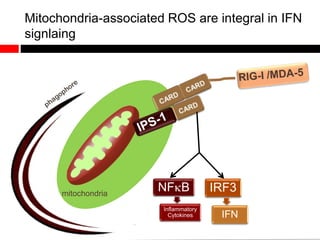
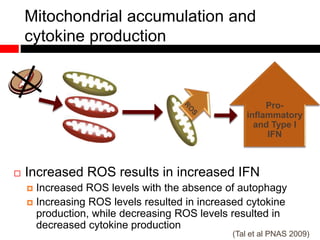
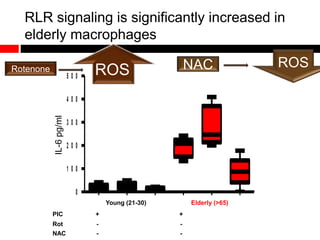
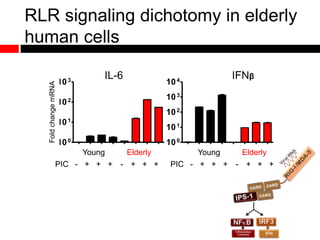
1) In the absence of autophagy, mitochondria accumulate and ROS levels increase. This leads to increased cytosolic antiviral signaling and cytokine production.
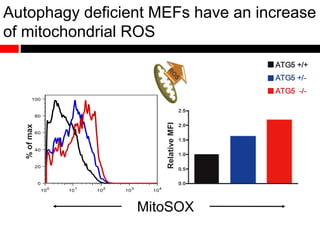
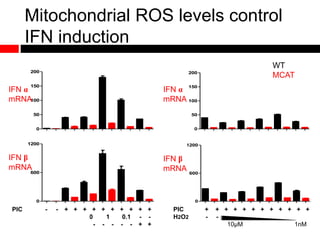
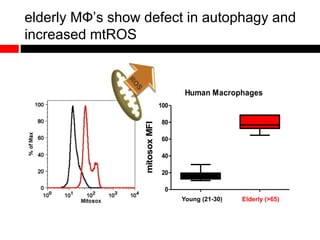
2) Autophagy deficient cells have increased mitochondrial ROS, which modulates cytosolic antiviral signaling pathways and results in increased type I interferon and inflammatory cytokine production.
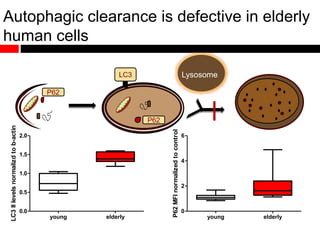
3) Decreased autophagy with age impacts mitochondrial and ROS homeostasis, which affects antiviral signaling. This may contribute to increased susceptibility to viral infection