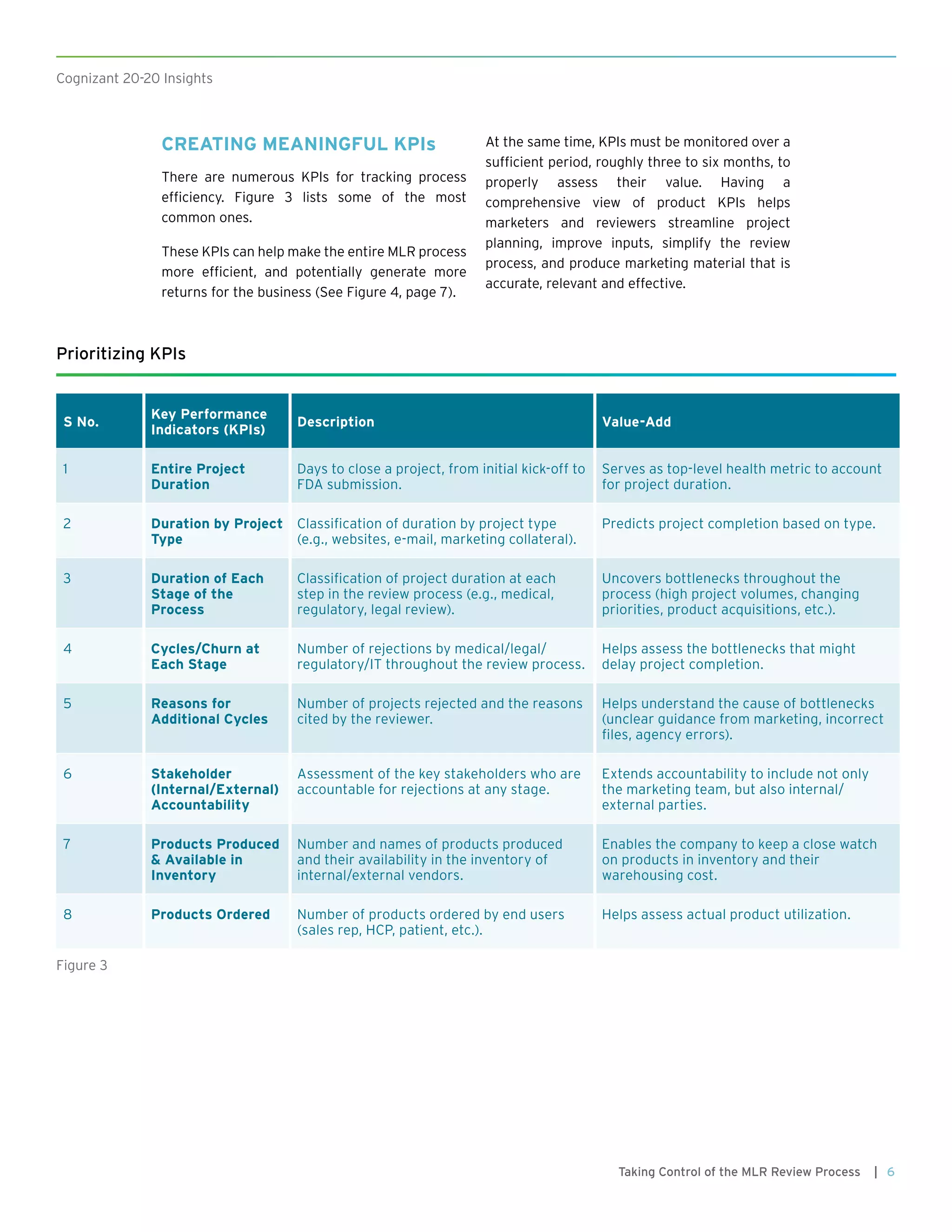
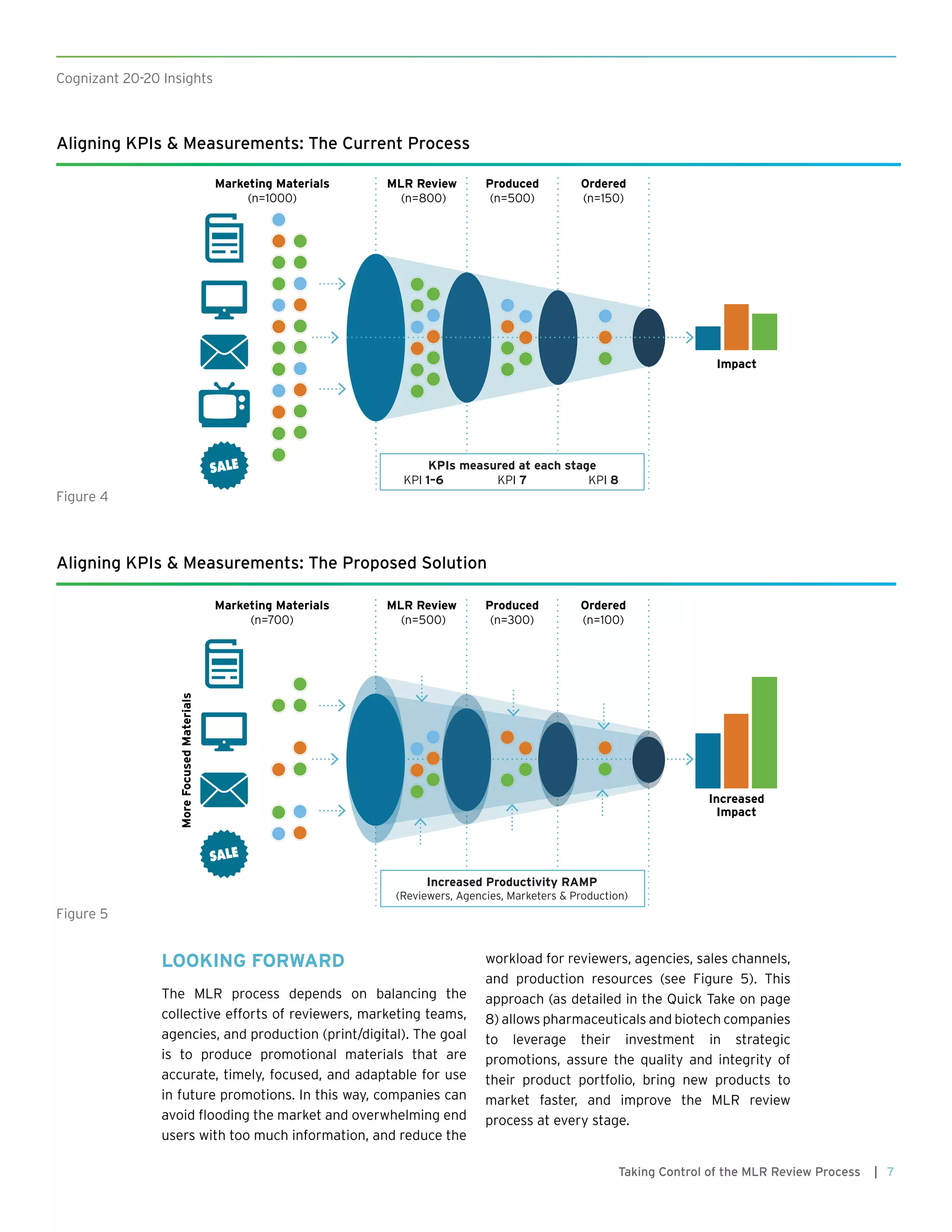
This document discusses how pharmaceutical companies can improve the efficiency and effectiveness of their medical, legal, and regulatory (MLR) review process for promotional materials. It recommends that companies define key performance indicators (KPIs) to track the progress of materials through the review stages and identify bottlenecks. Analytics can be used to analyze historical review data and monitor KPIs over time to optimize the process, reduce review times, and ensure compliance.