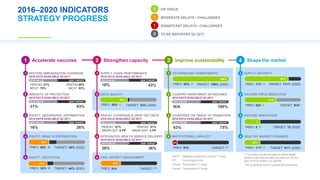
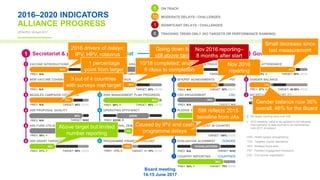
The GAVI Board meeting on June 14-15, 2017, reviewed the progress of the 2016-2020 strategy, highlighting data-driven insights and performance indicators. Key achievements included immunizing over 65 million children and a commitment to improve vaccine delivery and equity. The meeting emphasized ongoing challenges in vaccine introductions and the need for enhanced data quality and sub-national analysis for better vaccination strategies.