Report
Share
Download to read offline
Recommended
Bahagian luar ikan hiasan

Ikan Hiasan - menceritakan ikan hiasan .
kbsr tahun 5, kemahiran hidup bersepadu.
More Related Content
What's hot
What's hot (16)
งานนำเสนอ หน่วยที่ 5 เรื่องที่ 2 5 ตัวดำเนินการขนาดใหญ่

งานนำเสนอ หน่วยที่ 5 เรื่องที่ 2 5 ตัวดำเนินการขนาดใหญ่
งานนำเสนอ หน่วยที่ 1 เรื่องที่ 3 การประมวลผลคำโดยใช้ microsoft word 2010

งานนำเสนอ หน่วยที่ 1 เรื่องที่ 3 การประมวลผลคำโดยใช้ microsoft word 2010
Viewers also liked
Bahagian luar ikan hiasan

Ikan Hiasan - menceritakan ikan hiasan .
kbsr tahun 5, kemahiran hidup bersepadu.
Using Google effectively for 6th form research

How to use Google effectively, aimed at Years 12 and 13 (Key Stage 5).
Correlation between corrosion inhibitive effect and quantum molecular structu...

Correlation between corrosion inhibitive effect and quantum molecular structure of Schiff bases for iron in acidic and alkaline media
Quantum chemical calculations using the density functional theory (DFT) have been applied to the five kinds of polydentate Schiff base compounds (PSCs), act as inhibitors for iron in aerated 2.0 M HNO3 and 2.0 M NaOH media. The structural parameters, such as the frontier molecular orbital energy HOMO (highest occupied molecular orbital), LUMO (lowest unoccupied molecular orbital), energy gap ΔE (ELUMO - EHOMO), the charge distribution, the absolute electronegativity (χ), the fraction of electrons transfer (ΔN) from inhibitors to iron, the dipole moment (μ), the global hardness (η) and the total energy (Etotal) were also calculated and correlated with their inhibition efficiencies (%IE). The inhibition effects of (PSCs) may be explained in terms of electronic properties. The results showed that the (%IE) of PSCs increased with the increase in EHOMO and decrease in ELUMO - EHOMO. The inhibitor molecules were first adsorbed on the iron surface and blocking the reaction sites available for corrosive attack; and the areas containing N and O atoms are most possible sites for bonding by donating electrons to the iron surface through interaction with π-electrons of the aromatic rings, and the azo methine group. Also, the adsorbed Schiff base molecules interact with iron ions in the corrosive media leading to neutral and cationic iron-Schiff base complexes. Adsorption process is spontaneous, exothermic and obeyed Temkin isotherm and regarded as physical as chemical mechanism. The polarization studied indicated that the inhibitors act as a mixed type inhibitor in HNO3; cause only inhibition of the cathode process in NaOH, and the magnitude of the displacement of the Tafel plot is proportional to its concentration. Models for the inhibition corrosion behaviour were developed based upon statistical analyses of the experimental data. Some thermodynamic and kinetic parameters (Kads, ΔGads) were estimated. Both experimental and quantum theoretical calculations are in excellent agreement. The inhibition efficiency increase in the order of: PSC_1 > PSC_2 > PSC_3 > PSC_4>PSC_5. Thus, DFT study gave further insight into the mechanism of inhibition action of PSCs. This research might provide a theoretical inhibition performance evaluation approach for homologous inhibitors.
064 و تكونون لي شهوداً القس كرم لامعى

14أَنْتُمْ نُورُ الْعَالَمِ. لاَ يُمْكِنُ أَنْ تُخْفَى مَدِينَةٌ مَوْضُوعَةٌ عَلَى جَبَل، 15وَلاَ يُوقِدُونَ سِرَاجًا وَيَضَعُونَهُ تَحْتَ الْمِكْيَالِ، بَلْ عَلَى الْمَنَارَةِ فَيُضِيءُ لِجَمِيعِ الَّذِينَ فِي الْبَيْتِ. 16فَلْيُضِئْ نُورُكُمْ هكَذَا قُدَّامَ النَّاسِ، لِكَيْ يَرَوْا أَعْمَالَكُمُ الْحَسَنَةَ، وَيُمَجِّدُوا أَبَاكُمُ الَّذِي فِي السَّمَاوَاتِ
Besparen met LED-TL en Ahlight

DENK GROEN, DENK AAN HET MILEU
MAAK UW ORGANISTATIE VERANTWOORDELIJK
EN STAP OVER OP LED -TL MET AHLight
Viewers also liked (19)
Correlation between corrosion inhibitive effect and quantum molecular structu...

Correlation between corrosion inhibitive effect and quantum molecular structu...
Best Practices for Complementing Live Chat with a Virtual Assistant

Best Practices for Complementing Live Chat with a Virtual Assistant
Similar to อุปกรณ์เครือข่ายคอมพิวเตอร์
Similar to อุปกรณ์เครือข่ายคอมพิวเตอร์ (20)
หลักสูตรที่ 2 หน่วยที่ 3: การวางแผนด้านวัสดุอุปกรณ์และระบบ

หลักสูตรที่ 2 หน่วยที่ 3: การวางแผนด้านวัสดุอุปกรณ์และระบบ
ใบความรู้เรื่อง การสื่อสารข้อมูลและเครือข่ายคอมพิวเตอร์ (1)

ใบความรู้เรื่อง การสื่อสารข้อมูลและเครือข่ายคอมพิวเตอร์ (1)
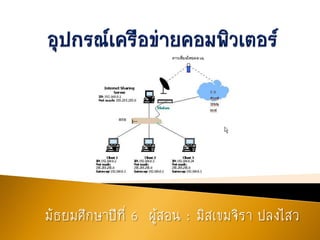
อุปกรณ์เครือข่ายคอมพิวเตอร์
- 1. มัธยมศึกษาปีที่ 6 ผู้สอน : มิสเขมจิรา ปลงไสว
- 2. การใช้คอมพิวเตอร์ช่วยส่งเสริมการทางานด้านต่าง ๆ นอกจากการเชื่อมต่ออุปกรณ์เพื่อส่งเสริมการทางาน เฉพาะด้านแล้ว... การเชื่อมต่อคอมพิวเตอร์ในรูปแบบของเครือข่าย คอมพิวเตอร์ (Network Computer) ก็ช่วยส่งเสริมการ ทางานของคอมพิวเตอร์ด้วยเช่นกัน
- 3. แล้วยังนิยมใช้สายรับและส่งสัญญาณ ข้อมูล ฮับ โมเด็ม และอุปกรณ์จัดเส้นทางช่วยในการ เชื่อมต่อเครือข่ายคอมพิวเตอร์
- 7. เป็นอุปกรณ์ที่ใช้เชื่อมต่อระหว่างคอมพิวเตอร์กับเครือข่ายขนาดใหญ่ที่ มีความซับซ้อน อุปกรณ์จัดเส้นทางจะทาหน้าที่ทวน แยกประเภท แปลง และจัดการกับสัญญาณข้อมูลให้สามารถใช้งานร่วมกันได้กับ คอมพิวเตอร์ทุกเครื่อง มีอุปกรณ์ 3 อย่าง ดังนี้ค่ะ
- 9. เหมาะสาหรับเชื่อมต่อระหว่างเครือข่าย 2 เครือข่ายที่มีขนาดหรือ มาตรฐานแตกต่างกัน
- 10. เหมาะสาหรับเชื่อมต่อกับเครือข่ายคอมพิวเตอร์หลายๆ เครือข่ายที่มี ความซับซ้อนกันมาก ๆ ซึ่งไม่สามารถจัดเส้นทางได้ด้วยบริดจ์และเรา เตอร์
