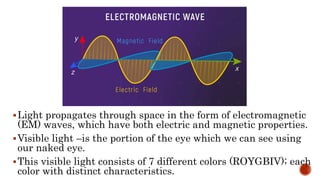
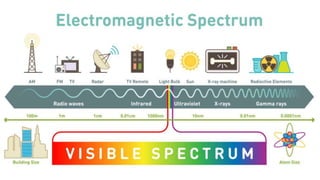
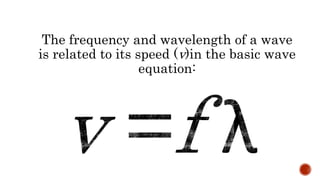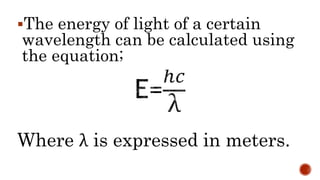This document discusses the nature of light and quanta. It explains that light propagates as electromagnetic waves that can be described by amplitude, wavelength, and frequency. Max Planck proposed the quantum theory which holds that light energy is quantized in multiples of Planck's constant times the frequency of light. This means light exists as both waves and particles called photons, whose energy is calculated using Planck's constant and the frequency of light.